

植物生态学报 ›› 2009, Vol. 33 ›› Issue (3): 425-432.DOI: 10.3773/j.issn.1005-264x.2009.03.001
• 研究论文 • 下一篇
收稿日期:2008-05-30
接受日期:2008-07-25
出版日期:2009-05-30
发布日期:2009-05-31
通讯作者:
王效科
作者简介:*E-mail: Wangxk@rcees.ac.cn基金资助:
ZHANG Wei-Wei, ZHENG Fei-Xiang, WANG Xiao-Ke( ), FENG Zhao-Zhong, OUYANG Zhi-Yun
), FENG Zhao-Zhong, OUYANG Zhi-Yun
Received:2008-05-30
Accepted:2008-07-25
Online:2009-05-30
Published:2009-05-31
Contact:
WANG Xiao-Ke
摘要:
采用旋转布气法开顶式气室 (Open top chambers, OTCs) 装置, 研究4种臭氧 (O3) 浓度水平 (过滤大气, O3浓度20nl·L-1;环境大气, O3浓度40nl·L-1;中等O3浓度处理, O3浓度为75nl·L-1;高浓度处理, O3浓度为150nl·L-1) 下水稻 (Oryzasativa) 根系中根系活力、可溶性蛋白含量、膜脂过氧化程度与抗氧化系统的变化差异。主要结果表明与过滤大气处理相比, O3浓度升高 (75和150nl·L-1) 使植株根系活力显著降低, 根系大幅度、过早地衰退;根系可溶性蛋白质含量显著下降;根系MDA含量显著升高, 膜脂过氧化程度加剧;SOD活性呈先升高后下降的变化趋势根系中H2O2含量大幅度显著上升, 并随着O3处理浓度升高和暴露时间延长变化幅度增大;CAT与POD活性则表现出升高趋势, 但处理后期升高幅度略微降低;整个处理期间根系ASA含量无显著变化。环境大气处理与过滤大气处理植株各个指标变化趋势基本一致并略微下降, 随着处理时间延长根系活力与蛋白质含量出现显著下降, 其他指标无显著差异。试验结果表明O3浓度升高会对植物地下部分根系产生影响;随着O3胁迫时间的延长, 植物将面临着缺乏强有力的根系生理代谢活力支持。
张巍巍, 郑飞翔, 王效科, 冯兆忠, 欧阳志云. 臭氧对水稻根系活力、可溶性蛋白含量与抗氧化系统的影响. 植物生态学报, 2009, 33(3): 425-432. DOI: 10.3773/j.issn.1005-264x.2009.03.001
ZHANG Wei-Wei, ZHENG Fei-Xiang, WANG Xiao-Ke, FENG Zhao-Zhong, OUYANG Zhi-Yun. EFFECTS OF OZONE ON ROOT ACTIVITY, SOLUBLE PROTEIN CONTENT AND ANTIOXIDANT SYSTEM IN ORYZA SATIVA ROOTS. Chinese Journal of Plant Ecology, 2009, 33(3): 425-432. DOI: 10.3773/j.issn.1005-264x.2009.03.001
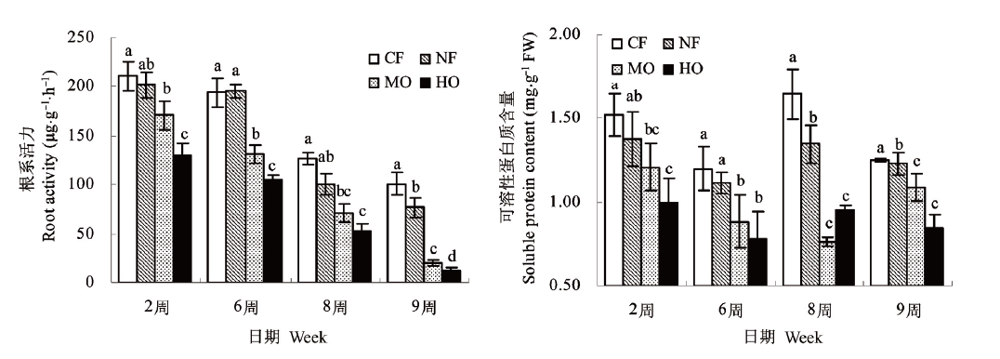
图1 不同O3浓度处 理下水稻根系活性力与可溶性蛋白质含量的变化 CF:过滤大气处理Charcoal-filtered treatment NF:环境大气处理Non-filtered treatment MO:中等O3浓度处理Moderate ozone treatment HO:高O3浓 度处理High ozone treatment小写字母表示Duncan’s检验中差异性达到0.05显著水平Values within the same column with different small letters are significantly different at the 0.05 probability level using Duncan’s multiple range test
Fig.1 Changes of different ozone concentrations on the root activity and soluble protein content in rice roots
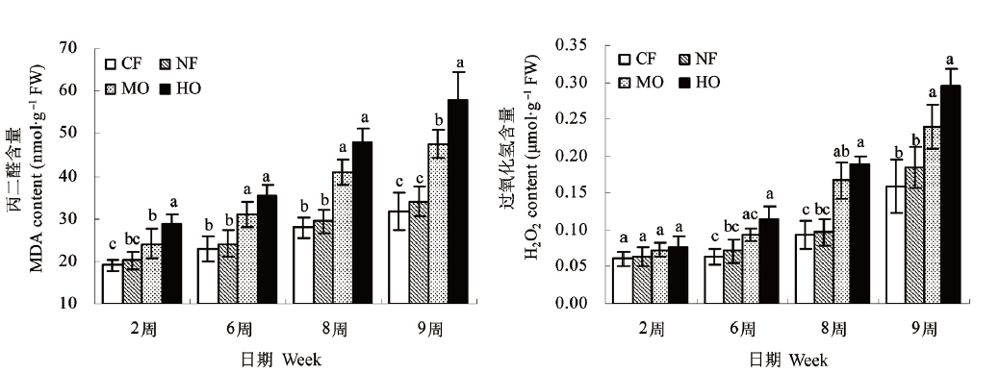
图2 不同O3浓度处理下水稻根系MDA含量与H2O2含量的变化 图注同图1
Fig.2 Changes of different ozone concentrations on the MDA content and H2O2 content in rice roots Notes see Fig.1
| [1] | Aebi H (1984). Catalase in vitro. Methods in Enzymology, 105,121-126. |
| [2] | Asada K (1992). Ascorbate peroxidase:a hydrogen perox-ide-scavenging enzyme in plants. Physiologia Planta-rum, 85,235-241. |
| [3] |
Asada K (1999). The water-water cycle in chloroplasts:scavenging of active oxygens and dissipation of excess photons. Annual Review of Plant Physiology and Plant Molecular Biology, 50,601-639.
PMID |
| [4] |
Beffa R, Martin HV, Pilet PE (1990). In vitro oxidation of indoleacetic acid by soluble auxin-oxidases and per-oxidases from maize roots. Plant Physiology, 94,485-491.
DOI URL |
| [5] |
Beyer WF Jr, Fridovich I (1987). Assaying for superoxide dismutase activity:some large consequences of minor changes in conditions. Analytical Biochemistry, 161,559-566.
DOI URL |
| [6] |
Bradford MM (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utiliz-ing the principle of protein-dye binding. Analytical Biochemistry, 72,248-254.
DOI URL |
| [7] |
Burkey KO, Eason G, Fiscus EL (2003). Factors that affect leaf extra cellular ascorbic acid content and redox status. Physiologia Plantarum, 117,51-57.
DOI URL |
| [8] |
Cooley RD, Manning WJ (1988). Ozone effects on growth and assimilate partitioning in alfalfa, Medicago sativa L. Environmental Pollution, 49,19-36.
DOI URL |
| [9] |
de Souza IRP, MacAdam JW (1998). A transient increase in apoplastic peroxidase activity precedes decrease in elongation rate of B73maize (Zea mays) leaf blades. Physiologia Plantarum, 104,556-562.
DOI URL |
| [10] | Dizengremel P, Petrini M (1994). Effects of air pollutants on the pathways of carbohydrate breakdown.In:Al-scher RG, Wellburn AR eds.Plant Response to the Gaseous Environment.Molecular, Metabolic and Physiological Aspects. Chapman and Hall, London,255-278. |
| [11] | Heath RL, Packer L (1968). Photoperoxidation in isolated chloroplast.I.Kinetics and stoichiometry of fatty acid peroxidation. Archives of Biochemistry and Biophysics, 25,189-198. |
| [12] | Heath RL, Taylor GE (1997). Physiological processes and plant responses to ozone exposure.In:Sandermann H, Wellburn AL, Heath RL eds.Forest Decline and Ozone, Ecological Studies, Vol.127. Springer-Verlag, Berlin,317-368. |
| [13] |
Hodges DM, Delong JM, Forney CF (1999). Improving the thiobarbituric acid-reactive-substances assay for esti-mating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta, 207,604-611.
DOI URL |
| [14] |
Ito O, Okano K, kuroiwa M (1985). Effects of NO2and O3alone or in combination on kidney bean plant (Phase-olus vulgaris L) :growth, partitioning of assimilates and root activities. Journal of Experimental Botany, 36,652-662.
DOI URL |
| [15] |
Karnosky DF, Pregitzer KS, Zak DR, Kubiske ME, Hendrey GR, Weinstein D, Nosal M, Percy KE (2005). Scaling ozone responses of forest trees to the ecosystem level in a changing climate. Plant, Cell and Environment, 28,965-981.
DOI URL |
| [16] |
Karpinski S, Escobar C, Karpinska B, Creissen G, Nul-lineaux PM (1997). Photosynthetic electron transport regulates the expression of cytosolic ascorbate peroxi-dase genes in Arabidopsis during excess light stress. Plant Cell, 9,627-640.
PMID |
| [17] | Kasurinen A, Helmisaari HS, Holopainen T (1999). The influence of elevated CO2, and O3on ozone roots and mycorrhizas of naturally growing young Scots pine trees during three exposure years. Global Change Bi-ology, 5,771-780. |
| [18] |
King JS, Pregitzer KS, Zak DR, Sober J, Isebrands JG, Dickson RE, Hendrey GR, Karnosky DF (2001). Fine-root biomass and fluxes of soil carbon in young stands of paper birch and trembling aspen as affected by elevated atmospheric CO2and tropospheric O3. Oecologia, 128,237-250.
DOI PMID |
| [19] | Krivosheeva A, Tao DL, Ottande C (1996). Cold acclimation and photoinhibition of photosynthesis in Scopine. Planta, 200,296-305. |
| [20] | Law MY, Charles SA, Halliwell B (1983). Glutathione andascorbic acid in spinach (Spinacia oleracea) chloro-plasts, the effect of hydrogen peroxide and of paraquat. Biochemistry, 210,899-903. |
| [21] |
Lyons TM, Barnes JD (1998). Influence of plant age on ozone resistance in Plantago major. New Phytologist, 138,83-89.
DOI URL |
| [22] | Matyssek R, Bytnerowicz A, Karlsson PE, Paoletti E, Sanz M, Schaub M, Wieser G (2007). Promoting the O3flux concept for European forest trees. Environmental Pol-lution, 146,587-607. |
| [23] | Mudd JB (1997). Biochemical basis for the toxicity ofozone.In:Yunus M, Iqbal M eds.Plant Response toAir Pollution. Wiley, New York,267-284. |
| [24] |
Mukherjee SP, Choudhuri MA (1983). Implications of water stress-induced changes in the levels of endogenouascorbic acid and hydrogen peroxide in Vigna seedlings. Physiologia Plantarum, 58,166-170.
DOI URL |
| [25] | Neill SJ, Desikan R, Clarke A, Hurst RD, Hancock JT (2002). Hydrogen peroxide and nitric oxide as signal-ing molecules in plants. Journal of Experimental Bot-any, 53,1237-1247. |
| [26] |
Noormets A, Sober A, Pell EJ, Dickson RE, Podila GK, Sober J, Isebrands JG, Karnosky DF (2001). Stomatal and non-stomatal limitation to photosynthesis in two trembling aspen (Populus tremuloides Michx.) clonesexposed to elevated CO2and/or O3. Plant, Cell and Environment, 24,327-336.
DOI URL |
| [27] | Nouchi I, Ito O, Harazono Y, Kouchi H (1995). Accelera-tion of13C-labelled photosynthate partitioning from leaves to panicles in rice plants exposed to chronic ozone at the reproductive stage. Environmental Pollu-tion, 88,253-260. |
| [28] |
Pearson S, Davison AW, Reiling K, Ashenden T, Oller-enshaw JH (1996). The effects of different ozone ex-posures on three contrasting populations of Plantago major. New Phytologist, 132,493-502.
DOI PMID |
| [29] |
Pell EJ, Sinn JP, Brendley BW, Samuelson L, Vinten-Johansen C, Tien M, Skillman J (1999). Differential response of four tree species to ozone-induced acceleration of foliar senescence. Plant, Cell and Environment, 22,779-790.
DOI URL |
| [30] |
Puckette MC, Weng H, Mahalingam R (2007). Physiological and biochemical responses to acute ozone-induced oxidative stress in Medicago truncatula. Plant Physiology and Biochemistry, 45,70-79.
PMID |
| [31] | Reich (1983). Effects of low concentrations of O3on netphotosynthesis, dark respiration, and chlorophyllcontents in aging hybrid poplar leaves. PlantPhysiology, 73,291-296. |
| [32] |
Robinson JM, Sicher RC (2004). Antioxidant levels decrease in primary leaves of barley during growth at ambient and elevated carbon dioxide levels. International Journal of Plant Sciences, 165,965-973.
DOI URL |
| [33] |
Sakaki T, Kondo N, Sugahara K (1983). Breakdown of photosynthetic pigments and lipids in spinach leaves with ozone fumigation:role of active oxygens. Physiologia Plantarum, 59,28-34.
DOI URL |
| [34] |
Spence RD, Rykiel EJ, Sharpe PJH (1990). Ozone alters carbon allocation in loblolly pine:assessment with carbon-11labeling. Environmental Pollution, 64,93-106.
DOI URL |
| [35] | Tingey DT, Heck WW, Reinert RA (1971). Effect of low concentrations of ozone and sulfur dioxide on foliage, growth and yield of radish. Journal of the American Society for Horticultural Science, 96,369-371. |
| [36] | Torsethaugen G, Pell EJ, Assmann SM (1999). Ozone inhibits guard cell K+channels implicated in stomatal opening. Proceedings of the National Academy of Sciences of the United States of America, 96,13577-13582. |
| [37] | United States Environmental Protection Agency (USEPA) (1996). Air Quality Criteria for Ozone and Related Photochemical Oxidants Vol.2.USEPA, Office of Research and Development, Washington DC. |
| [38] | Wei DZ (魏智道), Ning SJ (宁书菊), Lin WX (林文雄) (2004). Relationship between wheat root activity and leaf senescence. Chinese Journal of Applied Ecology (应用生态学报), 15,1565-1569. (in Chinese with English abstract) |
| [39] | Willekens H, Langebartels C, TiréC, Van Montagu M, InzéD, Van Camp W (1994). Differential expression of catalase genes in Nicotiana plumbaginifolia (L.). Proceedings of the National Academy of Sciences of the United States of America, 91,10450-10454. |
| [40] | Zhao TH (赵天宏), Shi Y (史奕), Huang GH (黄国宏) (2003). Effect of doubled CO2and O3concentration and their interactions on ultrastructure of soybean chloroplast. Chinese Journal of Applied Ecology (应用生态学报), 14,2229-2232. (in Chinese with English abstract) |
| [1] | 马艳泽, 杨熙来, 徐彦森, 冯兆忠. 四种常见树木叶片光合模型关键参数对臭氧浓度升高的响应[J]. 植物生态学报, 2022, 46(3): 321-329. |
| [2] | 李景, 王欣, 王振华, 王斌, 王成章, 邓美凤, 刘玲莉. 臭氧和气溶胶复合污染对杨树叶片光合作用的影响[J]. 植物生态学报, 2020, 44(8): 854-863. |
| [3] | 冯兆忠, 袁相洋, 李品, 尚博, 平琴, 胡廷剑, 刘硕. 地表臭氧浓度升高对陆地生态系统影响的研究进展[J]. 植物生态学报, 2020, 44(5): 526-542. |
| [4] | 冯兆忠, 徐彦森, 尚博. FACE实验技术和方法回顾及其在全球变化研究中的应用[J]. 植物生态学报, 2020, 44(4): 340-349. |
| [5] | 周慧敏, 李品, 冯兆忠, 张殷波. 地表臭氧浓度升高与干旱交互作用对杨树非结构性碳水化合物积累和叶根分配的短期影响[J]. 植物生态学报, 2019, 43(4): 296-304. |
| [6] | 高峰, 李品, 冯兆忠. 臭氧与干旱对植物复合影响的研究进展[J]. 植物生态学报, 2017, 41(2): 252-268. |
| [7] | 胡俊靖, 陈双林, 郭子武, 陈卫军, 杨清平, 李迎春. 美丽箬竹水分生理整合的分株比例效应——基于叶片抗氧化系统与光合色素[J]. 植物生态学报, 2015, 39(7): 762-772. |
| [8] | 熊淑萍, 王静, 王小纯, 丁世杰, 马新明. 耕作方式及施氮量对砂姜黑土区小麦氮代谢及籽粒产量和蛋白质含量的影响[J]. 植物生态学报, 2014, 38(7): 767-775. |
| [9] | 沙依然·外力,李秉柏,张佳华,杨沈斌. 水稻模拟模型在高温敏感性研究中的应用[J]. 植物生态学报, 2014, 38(5): 515-528. |
| [10] | 张佳蕾,李向东,杨传婷,高芳,张凤,王媛媛,孙莲强. 两种品质类型花生品质形成与子叶细胞超微结构的关系[J]. 植物生态学报, 2013, 37(8): 768-776. |
| [11] | 邹媛媛, 刘琳, 刘洋, 赵亮, 邓启云, 吴俊, 庄文, 宋未. 不同水稻品种种子固有细菌群落的多样性[J]. 植物生态学报, 2012, 36(8): 880-890. |
| [12] | 隗溟,廖学群,李冬霞,段海龙. 水稻分蘖节位生产力比较[J]. 植物生态学报, 2012, 36(4): 324-332. |
| [13] | 朱治林,孙晓敏,赵风华,温学发,唐新斋,袁国富. 鲁西北平原冬小麦田臭氧浓度变化特征及对产量的潜在影响和机理分析[J]. 植物生态学报, 2012, 36(4): 313-323. |
| [14] | 陈娟, 曾青, 朱建国, 刘钢, 曹际玲, 谢祖彬, 唐昊冶, 小林和彦. 施氮肥缓解臭氧对小麦光合作用和产量的影响[J]. 植物生态学报, 2011, 35(5): 523-530. |
| [15] | 顾东祥, 汤亮, 徐其军, 雷晓俊, 曹卫星, 朱艳. 水氮处理下不同品种水稻根系生长分布特征[J]. 植物生态学报, 2011, 35(5): 558-566. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2022 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19