

Chin J Plant Ecol ›› 2013, Vol. 37 ›› Issue (9): 851-860.DOI: 10.3724/SP.J.1258.2013.00089
• Research Articles • Previous Articles Next Articles
XIA Jiang-Bao1, ZHANG Shu-Yong2,*( ), ZHAO Zi-Guo1, ZHAO Yan-Yun1, Gao Yuan1, GU Guang-Yi1, SUN Jing-Kuan1
), ZHAO Zi-Guo1, ZHAO Yan-Yun1, Gao Yuan1, GU Guang-Yi1, SUN Jing-Kuan1
Received:2013-04-07
Accepted:2013-06-24
Online:2013-04-07
Published:2013-09-02
Contact:
ZHANG Shu-Yong
About author:*E-mail: zhsy@sdau.edu.cnXIA Jiang-Bao, ZHANG Shu-Yong, ZHAO Zi-Guo, ZHAO Yan-Yun, Gao Yuan, GU Guang-Yi, SUN Jing-Kuan. Critical effect of photosynthetic efficiency in Salix matsudana to soil moisture and its thres- hold grade in shell ridge island[J]. Chin J Plant Ecol, 2013, 37(9): 851-860.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.3724/SP.J.1258.2013.00089
| 土壤相对 含水量 Wr(%) | 表观量子效率 AQY | 光抑制项 β (10-4× m-2·s-1· pigment molecules-1) | 光饱和项 γ (10-4×m-2·s-1· pigment molecules-1) | 光补偿点 LCP (μmol·m-2·s-1) | 光饱和点 LSP (μmol·m-2·s-1) | 最大净光合速率 Pnmax (μmol·m-2·s-1) | 暗呼吸速率 Rd (μmol·m-2·s-1) |
|---|---|---|---|---|---|---|---|
| 93.2 | 0.030c | 2.70c | 5.14a | 38.9e | 1 367b | 14.06ef | 1.13cd |
| 88.0 | 0.037d | 2.43c | 7.77ab | 36.6e | 1 348b | 15.62f | 0.70b |
| 80.9 | 0.049e | 0.98a | 20.95d | 31.8d | 1 775d | 13.47e | 1.64e |
| 77.1 | 0.046e | 1.79b | 15.18c | 19.2b | 1 370b | 13.48e | 1.93f |
| 69.1 | 0.050e | 0.99a | 26.41e | 18.6ab | 1 614c | 11.56d | 1.30d |
| 61.2 | 0.023b | 1.81b | 8.67b | 16.4a | 1 619c | 10.06d | 1.03c |
| 46.8 | 0.017a | 2.86c | 4.32a | 18.8ab | 1 354b | 8.79c | 0.32a |
| 34.2 | 0.020ab | 2.97c | 16.52c | 25.6c | 946a | 4.76b | 0.79b |
| 23.4 | 0.021ab | 1.58b | 48.00f | 36.5e | 958a | 2.44a | 0.66b |
Table 1 Light response parameters of net photosynthetic rate in leaves of Salix matsudana under different soil water content conditions
| 土壤相对 含水量 Wr(%) | 表观量子效率 AQY | 光抑制项 β (10-4× m-2·s-1· pigment molecules-1) | 光饱和项 γ (10-4×m-2·s-1· pigment molecules-1) | 光补偿点 LCP (μmol·m-2·s-1) | 光饱和点 LSP (μmol·m-2·s-1) | 最大净光合速率 Pnmax (μmol·m-2·s-1) | 暗呼吸速率 Rd (μmol·m-2·s-1) |
|---|---|---|---|---|---|---|---|
| 93.2 | 0.030c | 2.70c | 5.14a | 38.9e | 1 367b | 14.06ef | 1.13cd |
| 88.0 | 0.037d | 2.43c | 7.77ab | 36.6e | 1 348b | 15.62f | 0.70b |
| 80.9 | 0.049e | 0.98a | 20.95d | 31.8d | 1 775d | 13.47e | 1.64e |
| 77.1 | 0.046e | 1.79b | 15.18c | 19.2b | 1 370b | 13.48e | 1.93f |
| 69.1 | 0.050e | 0.99a | 26.41e | 18.6ab | 1 614c | 11.56d | 1.30d |
| 61.2 | 0.023b | 1.81b | 8.67b | 16.4a | 1 619c | 10.06d | 1.03c |
| 46.8 | 0.017a | 2.86c | 4.32a | 18.8ab | 1 354b | 8.79c | 0.32a |
| 34.2 | 0.020ab | 2.97c | 16.52c | 25.6c | 946a | 4.76b | 0.79b |
| 23.4 | 0.021ab | 1.58b | 48.00f | 36.5e | 958a | 2.44a | 0.66b |
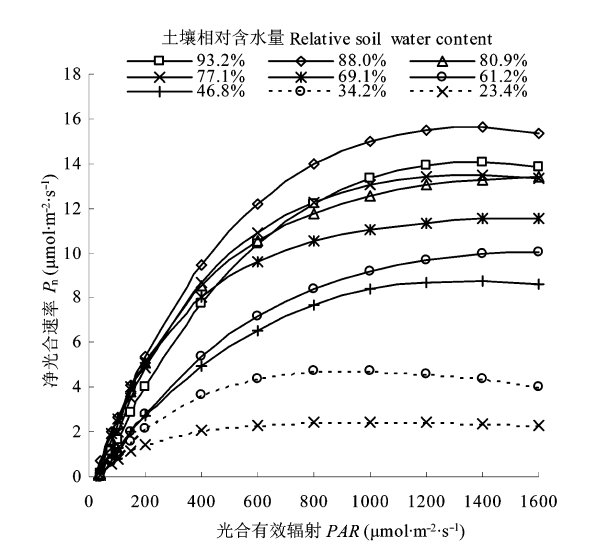
Fig. 1 Fitting response curves of net photosynthetic rate (Pn) in leaves of Salix matsudana to photosynthetically active radiation (PAR) under different relative water content conditions.
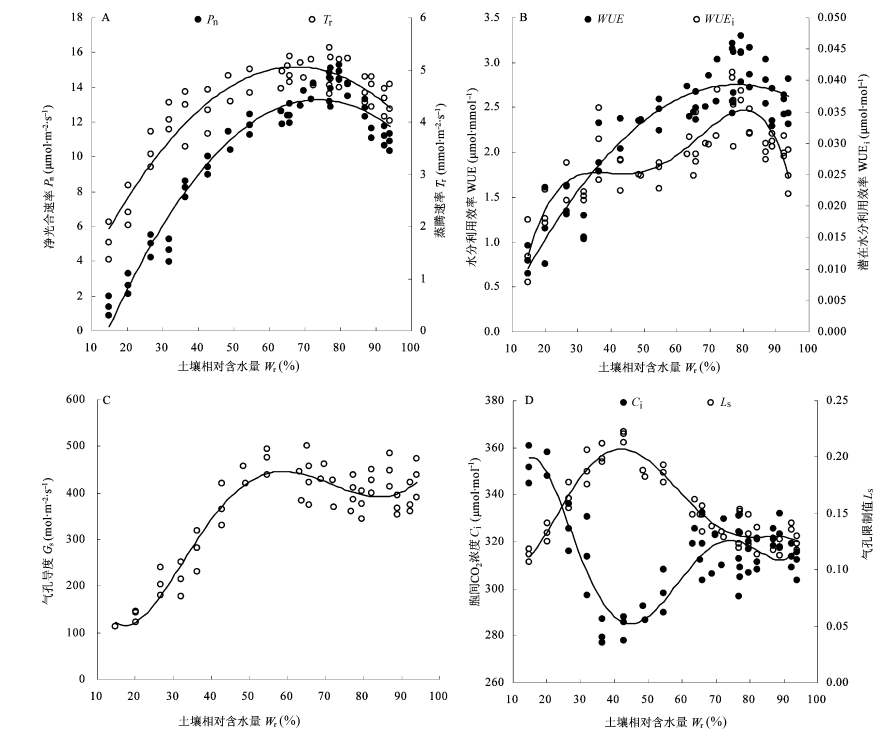
Fig. 2 Water response curves of net photosynthetic rate (Pn), transpiration rate(Tr) (A); water use efficiency (WUE), intrinsic water use efficiency (WUEi) (B); stomatal conductance (Gs) (C); intercellular CO2 concentration (Ci) and stomatal limitation value (Ls) (D) in leaves of Salix matsudana. Wr, relative soil water content.
| 土壤水分临界指标 Critical index of soil water1) | 土壤相对含水量临界点 Critical point of relative soil water content (Wr) | 土壤水分有效性分级 Grading of soil water availability2) | 土壤相对含水量阈值范围 Threshold grade of relative soil water content (Wr) |
|---|---|---|---|
| 净光合速率(Pn)水分补偿点 WCP Pn | 14.4% | 无产无效水 NPNEW | <14.4% |
| Pn水分气孔限制转折点 TP Pn | 42.9% | 低产低效水 LPLEW | 14.4%-48.1%; >93.2% |
| Pn水分饱和点 WS Pn | 73.1% | 中产低效水 MPLEW | 48.1%-50.4% |
| 水分利用效率(WUE)水分高效点 WSP WUE | 80.1% | 中产中效水 MPMEW | 50.4%-73.2% 80.1%-93.2% |
| Pn均值点 MVP Pn | 48.1%, 98.1% | ||
| WUE均值点 MVP WUE | 50.4%, 93.2% | 高产高效水 HPHEW | 73.1%-80.1% |
Table 2 Critical water point of photosynthetic efficiency and its threshold grade in leaves of Salix matsudana
| 土壤水分临界指标 Critical index of soil water1) | 土壤相对含水量临界点 Critical point of relative soil water content (Wr) | 土壤水分有效性分级 Grading of soil water availability2) | 土壤相对含水量阈值范围 Threshold grade of relative soil water content (Wr) |
|---|---|---|---|
| 净光合速率(Pn)水分补偿点 WCP Pn | 14.4% | 无产无效水 NPNEW | <14.4% |
| Pn水分气孔限制转折点 TP Pn | 42.9% | 低产低效水 LPLEW | 14.4%-48.1%; >93.2% |
| Pn水分饱和点 WS Pn | 73.1% | 中产低效水 MPLEW | 48.1%-50.4% |
| 水分利用效率(WUE)水分高效点 WSP WUE | 80.1% | 中产中效水 MPMEW | 50.4%-73.2% 80.1%-93.2% |
| Pn均值点 MVP Pn | 48.1%, 98.1% | ||
| WUE均值点 MVP WUE | 50.4%, 93.2% | 高产高效水 HPHEW | 73.1%-80.1% |
| [1] | Chen J, Zhang GC, Zhang SY, Wang MJ (2008). Response processes of Aralia elata photosynthesis and transpiration to light and soil moisture. Chinese Journal of Applied Ecology, 19, 1185-1190. (in Chinese with English abstract) |
| [陈建, 张光灿, 张淑勇, 王梦军 (2008). 辽东楤木光合和蒸腾作用对光照和土壤水分的响应过程. 应用生态学报, 19, 1185-1190.] | |
| [2] | Du N, Guo WH, Zhang XR, Wang RQ (2010). Morphological and physiological responses of Vitex negundo L. var. heterophylla (Franch.) Rehd. to drought stress. Acta Physiologiae Plantarum, 32, 839-848. |
| [3] | Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009). Plant drought stress: effects, mechanisms and management. Agronomy for Sustainable Development, 29, 185-212. |
| [4] | Farquhar GD, Sharkey TD (1982). Stomatal conductance and photosynthesis. Annual Review of Plant Physiology, 33, 317-345. |
| [5] | Gao S, Su PX, Yan QD, Ding SS (2010). Canopy and leaf gas exchange of Haloxylon ammodendron under different soil moisture regimes. Science in China (Series C: Life Sciences), 53, 718-728. |
| [6] |
Garadian Z, Tichý J, Vácha F (2011). Structure of PS I, PS II and antenna complexes from yellow-green alga Xanthonema debile. Photosynthesis Research, 108, 25-32.
URL PMID |
| [7] | Guo YQ, Almaz B, Gao QZ, Duan MJ, Ganzhu ZB, Wan YF, Li YE, Guo HB (2011). Photosynthetic characteristics of Stipa purpurea under irrigation in northern Tibet and its short-term response to temperature and CO2concentration. Chinese Journal of Plant Ecology, 35, 311-321. (in Chinese with English abstract) |
| [郭亚奇, 阿里穆斯, 高清竹, 段敏杰, 干珠扎布, 万运帆, 李玉娥, 郭红保 (2011). 灌溉条件下藏北紫花针茅光合特性及其对温度和CO2浓度的短期响应. 植物生态学报, 35, 311-321.] | |
| [8] | Jin X, Xu J, Bai KD, Feng JX, Zhang JS, Wan XC (2011). Comparison of drought strategies of three co-existing woody plants by their hydraulic structures. Journal of Beijing Forestry University, 33(6),135-141. (in Chinese with English abstract) |
| [靳欣, 徐洁, 白坤栋, 冯锦霞, 张劲松, 万贤崇 (2011). 从水力结构比较3种共存木本植物的抗旱策略. 北京林业大学学报, 33(6),135-141.] | |
| [9] | Julie CN, Spencer NB, Donald RY, Jarrod E, John EA (2010). Diurnal patterns of photosynthesis, chlorophyll fluorescence, and PRI to evaluate water stress in the invasive species, Elaeagnus umbellata Thunb. Trees, 24, 237-245. |
| [10] | Juvany M, Müller M, Munné-Bosch S (2013). Plant age-related changes in cytokinins, leaf growth and pigment accumulation in juvenile mastic trees. Environmental and Experimental Botany, 87, 10-18. |
| [11] | Mitchell PJ, Veneklaas EJ, Lambers H, Burgess SSO (2008). Leaf water relations during summer water deficit: differential responses in turgor maintenance and variation in leaf structure among different plant communities in south-western Australia. Plant, Cell & Environment, 31, 1791-1802. |
| [12] | Radwan UA (2007). Photosynthetic and leaf anatomical characteristics of the drought-resistant Balanites aegyptiaca (L.) Del. seedlings. American-Eurasian Journal of Agricultural and Environmental Sciences, 6, 680-688. |
| [13] | Sofo A, Dichio B, Montanaro G, Xiliyannis C (2009). Photosynthetic performance and light response of two olive cultivars under different water and light regimes. Photosynthetica, 47, 602-608. |
| [14] | Su H, Li YG, Su BY, Sun JX (2012). Effects of groundwater decline on photosynthetic characteristics and stress tolerance of Ulmus pumila in Hunshandake Sandy Land, China. Chinese Journal of Plant Ecology, 36, 177-186. (in Chinese with English abstract) |
| [苏华, 李永庚, 苏本营, 孙建新 (2012). 地下水位下降对浑善达克沙地榆树光合及抗逆性的影响. 植物生态学报, 36, 177-186.] | |
| [15] | Sun DB, Wang QS (2012). Effects of water on the photosynthetic characteristics of alfalfa (Medicago sativa). Chinese Journal of Plant Ecology, 36, 72-80. (in Chinese with English abstract) |
| [孙东宝, 王庆锁 (2012). 水分对苜蓿叶片光合特性的影响. 植物生态学报, 36, 72-80.] | |
| [16] | Wang H, Zhou GS, Jiang YL, Shi YH, Xu ZZ (2012). Interactive effects of changing precipitation and elevated CO2 concentration on photosynthetic parameters of Stipa breviflora. Chinese Journal of Plant Ecology, 36, 597-606. (in Chinese with English abstract) |
| [王慧, 周广胜, 蒋延玲, 石耀辉, 许振柱 (2012). 降水与CO2浓度协同作用对短花针茅光合特性的影响. 植物生态学报, 36, 597-606.] | |
| [17] | Wang RR, Xia JB, Yang JH, Zhao YY, Liu JT, Sun JK (2013). Comparison of light response models of photosynthesis in leaves of Periploca sepium under drought stress in sand habitat formed from seashells. Chinese Journal of Plant Ecology, 37, 111-121. (in Chinese with English abstract) |
| [王荣荣, 夏江宝, 杨吉华, 赵艳云, 刘京涛, 孙景宽 (2013). 贝壳砂生境干旱胁迫下杠柳叶片光合光响应模型比较. 植物生态学报, 37, 111-121.] | |
| [18] | Wang ZX, Wei H, Li CX, Lü Q, Zhou J, Gao W, Chen W (2012). Effect of soil moisture variations on photosynthetic characteristics of slash pine seedlings. Acta Botanica Boreali-Occidentalia Sinica, 32, 980-987. (in Chinese with English abstract) |
| [王振夏, 魏虹, 李昌晓, 吕茜, 周珺, 高伟, 陈伟 (2012). 土壤水分交替变化对湿地松幼苗光合特性的影响. 西北植物学报, 32, 980-987.] | |
| [19] |
Xia JB, Zhang GC, Liu G, Han W, Chen J, Liu X (2007). Light response of Wisteria sinensis leaves physiological parameters under different soil moisture conditions. Chinese Journal of Applied Ecology, 18, 30-34. (in Chinese with English abstract)
URL PMID |
|
[夏江宝, 张光灿, 刘刚, 韩炜, 陈建, 刘霞 (2007). 不同土壤水分条件下紫藤叶片生理参数的光响应. 应用生态学报, 18, 30-34.]
PMID |
|
| [20] | Xia JB, Zhang GC, Sun JK, Liu X (2011). Threshold effects of photosynthetic and physiological parameters in Prunus sibirica to soil moisture and light intensity. Chinese Journal of Plant Ecology, 35, 322-329. (in Chinese with English abstract) |
| [夏江宝, 张光灿, 孙景宽, 刘霞 (2011). 山杏叶片光合生理参数对土壤水分和光照强度的阈值效应. 植物生态学报, 35, 322-329.] | |
| [21] | Xia JB, Zhang SY, Zhang GC, Xie WJ, Lu ZH (2011). Critical responses of photosynthetic efficiency in Campsis radicans (L.) Seem to soil water and light intensities. African Journal of Biotechnology, 10, 17748-17754. |
| [22] | Ye ZP (2007). A new model for relationship between irradiance and the rate of photosynthesis in Oryza sativa. Phtosynthetica, 45, 637-640. |
| [23] | Ye ZP, Kang HJ (2012). Study on biological significance of coefficients in modified model of photosynthesis- irradiance. Journal of Yangzhou University (Agricultural and Life Science Edition), 33, 51-57. (in Chinese with English abstract) |
| [叶子飘, 康华靖 (2012). 植物光响应修正模型中系数的生物学意义研究. 扬州大学学报(农业与生命科学版), 33, 51-57.] | |
| [24] | Zhang GC, Xia JB, Shao HB, Zhang SY (2012). Grading woodland soil water productivity and soil bioavailability in the semi-arid Loess Plateau of China. Clean-Soil, Air, Water, 40, 148-153. |
| [25] | Zhang SY, Zhang GC, Gu SY, Xia JB, Zhao JK (2010). Critical responses of photosynthetic efficiency of Goldspur apple tree to soil water variation in semiarid loess hilly area. Photosynthetica, 48, 589-595. |
| [26] | Zhang SY, Zhou ZF, Xia JB, Zhang GC (2007). The responses of Euonymus fortunei var. radicans Sieb. leaf photosynthesis to light in different soil moisture. Acta Botanica Boreali-Occidentalia Sinica, 27, 2514-2521. (in Chinese with English abstract) |
| [张淑勇, 周泽福, 夏江宝, 张光灿 (2007). 不同土壤水分条件下小叶扶芳藤叶片光合作用对光的响应. 西北植物学报, 27, 2514-2521.] | |
| [27] | Zhang ZK, Zhang GC, Liu SS, Pei B, Xu ZQ, Xu P, Liu X (2012). Effects of soil moisture on photosynthesis diurnal changes of Prunus sibirica L. Science of Soil and Water Conservation, 10, 99-104. (in Chinese with English abstract) |
| [张征坤, 张光灿, 刘顺生, 裴斌, 徐志强, 徐萍, 刘霞 (2012). 土壤水分对山杏光合作用日变化过程的影响. 中国水土保持科学, 10, 99-104.] |
| [1] | LI Wei-Bin, ZHANG Hong-Xia, ZHANG Yu-Shu, CHEN Ni-Na. Influence of diurnal asymmetric warming on carbon sink capacity in a broadleaf Korean pine forest in Changbai Mountains, China [J]. Chin J Plant Ecol, 2023, 47(9): 1225-1233. |
| [2] | SHEN Jian, HE Zong-Ming, DONG Qiang, GAO Shi-Lei, LIN Yu. Effects of mild fire on soil respiration rate and abiotic factors in coastal sandy plantation [J]. Chin J Plant Ecol, 2023, 47(7): 1032-1042. |
| [3] | YANG Li-Lin, XING Wan-Qiu, WANG Wei-Guang, CAO Ming-Zhu. Variation of sap flow rate of Cunninghamia lanceolata and its response to environmental factors in the source area of Xinʼanjiang River [J]. Chin J Plant Ecol, 2023, 47(4): 571-583. |
| [4] | DU Ying-Dong, YUAN Xiang-Yang, FENG Zhao-Zhong. Effects of different nitrogen forms on photosynthesis characteristics and growth of poplar [J]. Chin J Plant Ecol, 2023, 47(3): 348-360. |
| [5] | LI Xue, DONG Jie, HAN Guang-Xuan, ZHANG Qi-Qi, XIE Bao-Hua, LI Pei-Guang, ZHAO Ming-Liang, CHEN Ke-Long, SONG Wei-Min. Response of soil CO2 and CH4 emissions to changes in moisture and salinity at a typical coastal salt marsh of Yellow River Delta [J]. Chin J Plant Ecol, 2023, 47(3): 434-446. |
| [6] | ZHENG Zhou-Tao, ZHANG Yang-Jian. Variation in ecosystem water use efficiency and its attribution analysis during 1982-2018 in Qingzang Plateau [J]. Chin J Plant Ecol, 2022, 46(12): 1486-1496. |
| [7] | HAN Lu, YANG Fei, WU Ying-Ming, NIU Yun-Ming, ZENG Yi-Ming, CHEN Li-Xin. Responses of short-term water use efficiency to environmental factors in typical trees and shrubs of the loess area in West Shanxi, China [J]. Chin J Plant Ecol, 2021, 45(12): 1350-1364. |
| [8] | ZHOU Xiong, SUN Peng-Sen, ZHANG Ming-Fang, LIU Shi-Rong. Spatio-temporal characteristics of vegetation water use efficiency and their relationships with climatic factors in alpine and subalpine area of southwestern China [J]. Chin J Plant Ecol, 2020, 44(6): 628-641. |
| [9] | FENG Zhao-Zhong, LI Pin, ZHANG Guo-You, LI Zheng-Zhen, PING Qin, PENG Jin-Long, LIU Shuo. Impacts of elevated carbon dioxide concentration on terrestrial ecosystems: problems and prospective [J]. Chin J Plant Ecol, 2020, 44(5): 461-474. |
| [10] | Aizezitiyuemaier MAIMAITI, Yusufujiang RUSULI, HE Hui, Baihetinisha ABUDUKERIMU. Spatio-temporal characteristics of vegetation water use efficiency and its relationship with climate factors in Tianshan Mountains in Xinjiang from 2000 to 2017 [J]. Chin J Plant Ecol, 2019, 43(6): 490-500. |
| [11] | LI Jian-Jun, LIU Lian, CHEN Di-Ma, XU Feng-Wei, CHENG Jun-Hui, BAI Yong-Fei. Effects of collar size and buried depth on the measurement of soil respiration in a typical steppe [J]. Chin J Plant Ecol, 2019, 43(2): 152-164. |
| [12] | LI Xin-Hao, YAN Hui-Juan, WEI Teng-Zhou, ZHOU Wen-Jun, JIA Xin, ZHA Tian-Shan. Relative changes of resource use efficiencies and their responses to environmental factors in Artemisia ordosica during growing season [J]. Chin J Plant Ecol, 2019, 43(10): 889-898. |
| [13] | Chao-Yang FENG, He-Song WANG, Jian-xin SUN. Temporal changes of vegetation water use efficiency and its influencing factors in Northern China [J]. Chin J Plant Ecol, 2018, 42(4): 453-465. |
| [14] | WANG Xiang, ZHU Ya-Qiong, ZHENG Wei, GUAN Zheng-Xuan, SHENG Jian-Dong. Soil respiration features of mountain meadows under four typical land use types in Zhaosu Basin [J]. Chin J Plant Ecol, 2018, 42(3): 382-396. |
| [15] | CHAI Xi, LI Ying-Nian, DUAN Cheng, ZHANG Tao, ZONG Ning, SHI Pei-Li, HE Yong-Tao, ZHANG Xian-Zhou. CO2 flux dynamics and its limiting factors in the alpine shrub-meadow and steppe-meadow on the Qinghai-Xizang Plateau [J]. Chin J Plant Ecol, 2018, 42(1): 6-19. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2022 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn