Chinese Journal of Plant Ecology >
Relationships among xylem transport, anatomical structure and mechanical strength in stems and roots of three Podocarpaceae species
- 1State Key Laboratory of Subtropical Silviculture, Zhejiang A&F University, Hangzhou 311300, China
2College of Forestry and Biotechnology, Zhejiang A&F University, Hangzhou 311300, China
3State Key Laboratory of Desert and Oasis Ecology, Xinjiang Institute of Ecology and Geography, Chinese Academy of Sciences, Ürümqi 830011, China
Received date: 2020-12-04
Accepted date: 2021-03-17
Online published: 2021-06-04
Supported by
National Natural Science Foundation of China(31770651);National Natural Science Foundation of China(41730638);National Natural Science Foundation of China(31901280)
Abstract
Aims Hydraulic failure is one of the primary cause of plant mortality during drought. Thus, quantitative analysis on inter-specific and inter-organ variance in hydraulic traits can help us to predict the response and even survivability of species under climate change.
Methods Here, three Podocarpaceae species (Podocarpus macrophyllus, P. macrophyllusvar. maki and Nageia nagi) grown in a mesic common garden were studied, with xylem hydraulic function (specific hydraulic conductivity (Ks); embolism resistance (P50)), anatomical structure (tracheid diameter (Dt); hydraulic diameter (Dh); tracheid wall thickness (Tw); tracheid density (Nt); pit membrane diameter (Dp); pit density (Np)) and mechanical strength (wood density (WD); tracheid thickness to span ratio ((t/b)2)) measured. Then, we analyzed hydraulic traits variance at the organ level (stem and root) in three Podocarpaceae species, and investigated the relationships among xylem hydraulic traits, anatomical structure and mechanical strength in stems and roots.
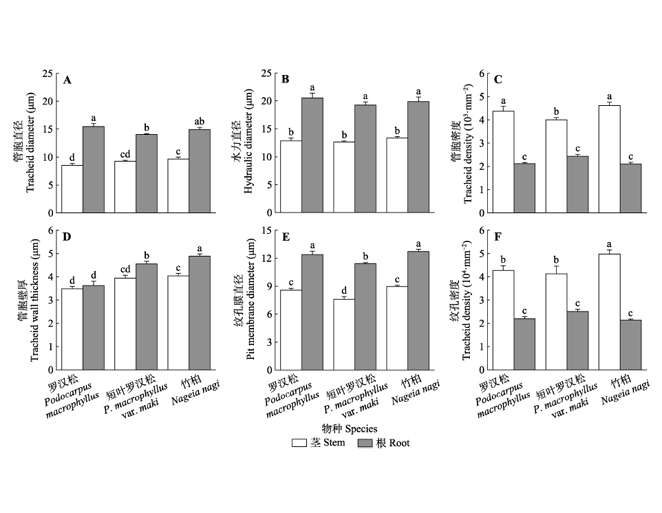
Important findings We found that: 1) The stem xylem in three Podocarpaceae species exhibited no safety- efficiency trade-off. In contrast, the root xylem exhibited safety-efficiency trade-off. 2) For stems, Ks was positively correlated with Dp, but decoupled with stem WD and (t/b)2; Stem P50 was negatively correlated with Dp, but not correlated with WD and (t/b)2. 3) For roots, Ks was positively correlated with hydraulic diameter, but negatively correlated with root Tw and (t/b)2; Root P50 was positively correlated with Tw, (t/b)2 and WD. Root xylem traits exhibited strong relationships with both Ks and P50, demonstrating its cause-and-effect basis for the safety-efficiency tradeoff. On the other hand, the absence of safety-efficiency tradeoff in stems may be attributed to the overbuilt xylem of Podocarpaceae. More experimental evidence on the overbuilding of xylem is desired in the future study.
Key words: embolism; hydraulic conductivity; vulnerability curve; tracheid; Podocarpaceae
Cite this article
LU Shi-Tong, CHEN Sen, LI Yan, WANG Zhong-Yuan, PAN Tian-Tian, YE Lin-Feng, XIE Jiang-Bo . Relationships among xylem transport, anatomical structure and mechanical strength in stems and roots of three Podocarpaceae species[J]. Chinese Journal of Plant Ecology, 2021 , 45(6) : 659 -669 . DOI: 10.17521/cjpe.2020.0402
References
| [1] | Adams HD, Zeppel MJB, Anderegg WRL, Hartmann H, Landhäusser SM, Tissue DT, Huxman TE, Hudson PJ, Franz TE, Allen CD, Anderegg LDL, Barron-Gafford GA, Beerling DJ, Breshears DD, Brodribb TJ, et al. (2017). A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nature Ecology & Evolution, 1, 1285-1291. |
| [2] | Allen CD (2009). Climate-induced forest dieback: an escalating global phenomenon. Unasylva, 60, 43-49. |
| [3] | Anderegg WRL, Klein T, Bartlett M, Sack L, Pellegrini AFA, Choat B, Jansen S (2016). Meta-analysis reveals that hydraulic traits explain cross-species patterns of drought- induced tree mortality across the globe. Proceedings of the National Academy of Sciences of the United States of America, 113, 5024-5029. |
| [4] | Baas P, Ewers FW, Davis SD, Wheeler EA (2004). Evolution of xylem physiology//Hemsley AR, Poole I. The Evolution of Plant Physiology. Elsevier, Oxford, UK. 273-295. |
| [5] | Brodribb TJ (2011). A functional analysis of podocarp ecology. Smithsonian Contributions to Botany, 95, 165-173. |
| [6] | Brodribb TJ, Feild TS (2000). Stem hydraulic supply is linked to leaf photosynthetic capacity: evidence from New Caledonian and Tasmanian rainforests. Plant, Cell & Environment, 23, 1381-1388. |
| [7] | Brodribb T, Hill RS (1999). The importance of xylem constraints in the distribution of conifer species. New Phytologist, 143, 365-372. |
| [8] | Choat B, Brodribb TJ, Brodersen CR, Duursma RA, López R, Medlyn BE (2018). Triggers of tree mortality under drought. Nature, 558, 531-539. |
| [9] | Choat B, Jansen S, Brodribb TJ, Cochard H, Delzon S, Bhaskar R, Bucci SJ, Feild TS, Gleason SM, Hacke UG, Jacobsen AL, Lens F, Maherali H, Martínez-Vilalta J, Mayr S, et al. (2012). Global convergence in the vulnerability of forests to drought. Nature, 491, 752-755. |
| [10] | Choat B, Medek DE, Stuart SA, Pasquet-Kok J, Egerton JJ, Salari H, Sack L, Ball MC (2011). Xylem traits mediate a trade-off between resistance to freeze-thaw-induced embolism and photosynthetic capacity in overwintering evergreens. New Phytologist, 191, 996-1005. |
| [11] | Dietrich L, Delzon S, Hoch G, Kahmen A (2019). No role for xylem embolism or carbohydrate shortage in temperate trees during the severe 2015 drought. Journal of Ecology, 107, 334-349. |
| [12] | Domec JC, Lachenbruch B, Meinzer FC (2006). Bordered pit structure and function determine spatial patterns of air-seeding thresholds in xylem of Douglas-fir (Pseudotsuga menziesii; Pinaceae) trees. American Journal of Botany, 93, 1588-1600. |
| [13] | Duursma R, Choat B (2017). Fitplc—An R package to fit hydraulic vulnerability curves. Journal of Plant Hydraulics, 4, e002. DOI: 10.20870/jph.2017.e002. |
| [14] | Ennajeh M, Simões F, Khemira H, Cochard H (2011). How reliable is the double-ended pressure sleeve technique for assessing xylem vulnerability to cavitation in woody angiosperms? Physiologia Plantarum, 142, 205-210. |
| [15] | Fan ZX, Sterck F, Zhang SB, Fu PL, Hao GY (2017). Tradeoff between stem hydraulic efficiency and mechanical strength affects leaf-stem allometry in 28 Ficus tree species. Frontiers in Plant Science, 8, 1619. DOI: 10.3389/fpls.2017.01619. |
| [16] | Fichot R, Barigah TS, Chamaillard S, Le Thiec D, Laurans F, Cochard H, Brignolas F (2010). Common trade-offs between xylem resistance to cavitation and other physiological traits do not hold among unrelated Populus deltoides × Populus nigra hybrids. Plant, Cell & Environment, 33, 1553-1568. |
| [17] | Gleason SM, Westoby M, Jansen S, Choat B, Hacke UG, Pratt RB, Bhaskar R, Brodribb TJ, Bucci SJ, Cao KF, Cochard H, Delzon S, Domec JC, Fan ZX, Field TS, et al. (2016). Weak tradeoff between xylem safety and xylem-specific hydraulic efficiency across the world’s woody plant species. New Phytologist, 211, 123-136. |
| [18] | Hacke UG, Jansen S (2009). Embolism resistance of three boreal conifer species varies with pit structure. New Phytologist, 182, 675-686. |
| [19] | Hacke UG, Sperry JS, Pittermann J (2000). Drought experience and cavitation resistance in six shrubs from the Great Basin, Utah. Basic and Applied Ecology, 1, 31-41. |
| [20] | Hacke UG, Sperry JS, Pittermann J (2004). Analysis of circular bordered pit function II. Gymnosperm tracheids with torus- margo pit membranes. American Journal of Botany, 91, 386-400. |
| [21] | Hacke UG, Sperry JS, Pockman WT, Davis SD, McCulloh KA (2001). Trends in wood density and structure are linked to prevention of xylem implosion by negative pressure. Oecologia, 126, 457-461. |
| [22] | Hacke UG, Sperry JS, Wheeler JK, Castro L (2006). Scaling of angiosperm xylem structure with safety and efficiency. Tree Physiology, 26, 689-701. |
| [23] | Hacke UG, Spicer R, Schreiber SG, Plavcová L (2017). An ecophysiological and developmental perspective on variation in vessel diameter. Plant, Cell & Environment, 40, 831-845. |
| [24] | Hajek P, Kurjak D, von Wühlisch G, Delzon S, Schuldt B (2016). Intraspecific variation in wood anatomical, hydraulic, and foliar traits in ten European beech provenances differing in growth yield. Frontiers in Plant Science, 7, 791. DOI: 10.3389/fpls.2016.00791. |
| [25] | IPCC (2013). Climate Change 2013: the Physical Science Basis: Working Group I Contribution to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK.. |
| [26] | Jansen S, Choat B, Pletsers A (2009). Morphological variation of intervessel pit membranes and implications to xylem function in angiosperms. American Journal of Botany, 96, 409-419. |
| [27] | Johnson DM, Wortemann R, McCulloh KA, Jordan-Meille L, Ward E, Warren JM, Palmroth S, Domec JC (2016). A test of the hydraulic vulnerability segmentation hypothesis in angiosperm and conifer tree species. Tree Physiology, 36, 983-993. |
| [28] | Lachenbruch B, McCulloh KA (2014). Traits, properties, and performance: How woody plants combine hydraulic and mechanical functions in a cell, tissue, or whole plant. New Phytologist, 204, 747-764. |
| [29] | Larter M, Pfautsch S, Domec JC, Trueba S, Nagalingum N, Delzon S (2017). Aridity drove the evolution of extreme embolism resistance and the radiation of conifer genus Callitris. New Phytologist, 215, 97-112. |
| [30] | Lens F, Sperry JS, Christman MA, Choat B, Rabaey D, Jansen S (2011). Testing hypotheses that link wood anatomy to cavitation resistance and hydraulic conductivity in the genus Acer. New Phytologist, 190, 709-723. |
| [31] | Lewis AM (1992). Measuring the hydraulic diameter of a pore or conduit. American Journal of Botany, 79, 1158-1161. |
| [32] | Li S, Lens F, Espino S, Karimi Z, Klepsch M, Schenk HJ, Schmitt M, Schuldt B, Jansen S (2016). Intervessel pit membrane thickness as a key determinant of embolism resistance in angiosperm xylem. IAWA Journal, 37, 152-171. |
| [33] | Liu CH, Li YY, Chen WY(2014). Hydraulic architecture of three typical woody plants in Ziwuling forest zone on the Loess Plateau. Acta Botanica Boreali-Occidentalia Sinica, 34, 835-842. |
| [33] | [ 刘存海, 李秧秧, 陈伟月(2014). 子午岭林区3种典型树木的水力结构特性比较. 西北植物学报, 34, 835-842.] |
| [34] | Loepfe L, Martinez-Vilalta J, Piñol J, Mencuccini M (2007). The relevance of xylem network structure for plant hydraulic efficiency and safety. Journal of Theoretical Biology, 247, 788-803. |
| [35] | Maherali H, Moura CF, Caldeira MC, Willson CJ, Jackson RB (2006). Functional coordination between leaf gas exchange and vulnerability to xylem cavitation in temperate forest trees. Plant, Cell & Environment, 29, 571-583. |
| [36] | Mackay DS, Savoy PR, Grossiord C, Tai XN, Pleban JR, Wang D, McDowell NG, Adams HD, Sperry JS (2020). Conifers depend on established roots during drought: results from a coupled model of carbon allocation and hydraulics. New Phytologist, 225, 679-692. |
| [37] | Martin-StPaul N, Delzon S, Cochard H (2017). Plant resistance to drought depends on timely stomatal closure. Ecology Letters, 20, 1437-1447. |
| [38] | Mayr S, Cochard H (2003). A new method for vulnerability analysis of small xylem areas reveals that compression wood of Norway spruce has lower hydraulic safety than opposite wood. Plant, Cell & Environment, 26, 1365-1371. |
| [39] | McCulloh KA, Domec JC, Johnson DM, Smith DD, Meinzer FC (2019). A dynamic yet vulnerable pipeline: integration and coordination of hydraulic traits across whole plants. Plant, Cell & Environment, 42, 2789-2807. |
| [40] | McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, Yepez EA (2008). Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytologist, 178, 719-739. |
| [41] | McElrone AJ, Pockman WT, Martínez-Vilalta J, Jackson RB (2004). Variation in xylem structure and function in stems and roots of trees to 20 m depth. New Phytologist, 163, 507-517. |
| [42] | Ogle K, Barber JJ, Willson C, Thompson B (2009). Hierarchical statistical modeling of xylem vulnerability to cavitation. New Phytologist, 182, 541-554. |
| [43] | Peters JMR, Gauthey A, Lopez R, Carins-Murphy MR, Brodribb TJ, Choat B (2020). Non-invasive imaging reveals convergence in root and stem vulnerability to cavitation across five tree species. Journal of Experimental Botany, 71, 6623-6637. |
| [44] | Pittermann J, Choat B, Jansen S, Stuart SA, Lynn L, Dawson TE (2010). The relationships between xylem safety and hydraulic efficiency in the Cupressaceae: the evolution of pit membrane form and function. Plant Physiology, 153, 1919-1931. |
| [45] | Pittermann J, Sperry JS (2006). Analysis of freeze-thaw embolism in conifers. The interaction between cavitation pressure and tracheid size. Plant Physiology, 140, 374-382. |
| [46] | Pittermann J, Sperry J (2003). Tracheid diameter is the key trait determining the extent of freezing-induced embolism in conifers. Tree Physiology, 23, 907-914. |
| [47] | Pittermann J, Sperry JS, Wheeler JK, Hacke UG, Sikkema EH (2006a). Mechanical reinforcement of tracheids compromises the hydraulic efficiency of conifer xylem. Plant, Cell & Environment, 29, 1618-1628. |
| [48] | Pittermann J, Sperry JS, Hacke UG, Wheeler JK, Sikkema EH (2006b). Inter-tracheid pitting and the hydraulic efficiency of conifer wood: the role of tracheid allometry and cavitation protection. American Journal of Botany, 93, 1265-1273. |
| [49] | Pratt RB, Jacobsen AL, Ewers FW, Davis SD (2007a). Relationships among xylem transport, biomechanics and storage in stems and roots of nine Rhamnaceae species of the California chaparral. New Phytologist, 174, 787-798. |
| [50] | Pratt RB, Jacobsen AL, Golgotiu KA, Sperry JS, Ewers FW, Davis SD (2007b). Life history type and water stress tolerance in nine California chaparral species (Rhamnaceae). Ecological Monographs, 77, 239-253. |
| [51] | Rowland L, da Costa ACL, Galbraith DR, Oliveira RS, Binks OJ, Oliveira AAR, Pullen AM, Doughty CE, Metcalfe DB, Vasconcelos SS, Ferreira LV, Malhi Y, Grace J, Mencuccini M, Meir P (2015). Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature, 528, 119-122. |
| [52] | Rodriguez-Dominguez CM, Carins Murphy MR, Lucani C, Brodribb TJ (2018). Mapping xylem failure in disparate organs of whole plants reveals extreme resistance in olive roots. New Phytologist, 218, 1025-1035. |
| [53] | Rodriguez-Dominguez CM, Brodribb TJ (2020). Declining root water transport drives stomatal closure in olive under moderate water stress. New Phytologist, 225, 126-134. |
| [54] | Rosas T, Mencuccini M, Barba J, Cochard H, Saura-Mas S, Martínez-Vilalta J (2019). Adjustments and coordination of hydraulic, leaf and stem traits along a water availability gradient. New Phytologist, 223, 632-646. |
| [55] | Schoonmaker AL, Hacke UG, Landhäusser SM, Lieffers VJ, Tyree MT (2010). Hydraulic acclimation to shading in boreal conifers of varying shade tolerance. Plant, Cell & Environment, 33, 382-393. |
| [56] | Schumann K, Leuschner C, Schuldt B (2019). Xylem hydraulic safety and efficiency in relation to leaf and wood traits in three temperate Acer species differing in habitat preferences. Trees, 33, 1475-1490. |
| [57] | Schulte PJ, Hacke UG, Schoonmaker AL (2015). Pit membrane structure is highly variable and accounts for a major resistance to water flow through tracheid pits in stems and roots of two boreal conifer species. New Phytologist, 208, 102-113. |
| [58] | Schuldt B, Knutzen F, Delzon S, Jansen S, Müller-Haubold H, Burlett R, Clough Y, Leuschner C (2016). How adaptable is the hydraulic system of European beech in the face of climate change-related precipitation reduction? New Phytologist, 210, 443-458. |
| [59] | Sperry JS, Hacke UG, Pittermann J (2006). Size and function in conifer tracheids and angiosperm vessels. American Journal of Botany, 93, 1490-1500. |
| [60] | Tyree MT, Davis SD, Cochard H (1994). Biophysical perspectives of xylem evolution: Is there a tradeoff of hydraulic efficiency for vulnerability to dysfunction? IAWA Journal, 15, 335-360. |
| [61] | Tyree MT, Zimmermann MH (2002). Xylem Structure and the Ascent of Sap. Springer, Berlin. 45-56. |
| [62] | van der Willigen C, Sherwin HW, Pammenter NW (2000). Xylem hydraulic characteristics of subtropical trees from contrasting habitats grown under identical environmental conditions. New Phytologist, 145, 51-59. |
| [63] | Zhang JZ, Gou XH, Zhao ZQ, Liu WH, Zhang F, Cao ZY, Zhou FF(2013). Improved method of obtaining micro- core paraffin sections in dendroecological research. Chinese Journal of Plant Ecology, 37, 972-977. |
| [63] | [ 张军周, 勾晓华, 赵志千, 刘文火, 张芬, 曹宗英, 周非飞(2013). 树轮生态学研究中微树芯石蜡切片制作的方法探讨. 植物生态学报, 37, 972-977.] |
| [64] | Zhou HH, Li WH, Ayup M, Xu Q(2012). Xylem hydraulic conductivity and embolism properties of desert riparian forest plants and its response to drought stress. Chinese Journal of Plant Ecology, 36, 19-29. |
| [64] | [ 周洪华, 李卫红, 木巴热克•阿尤普, 徐茜(2012). 荒漠河岸林植物木质部导水与栓塞特征及其对干旱胁迫的响应. 植物生态学报, 36, 19-29.] |
| [65] | Zhu SD, Cao KF (2009). Hydraulic properties and photosynthetic rates in co-occurring lianas and trees in a seasonal tropical rainforest in southwestern China. Plant Ecology, 204, 295-304. |