

Chin J Plan Ecolo ›› 2016, Vol. 40 ›› Issue (8): 834-846.DOI: 10.17521/cjpe.2016.0118
• Reviews • Previous Articles Next Articles
Ying JIN, Chuan-Kuan WANG*( ), Zheng-Hu ZHOU
), Zheng-Hu ZHOU
Online:2016-08-10
Published:2016-08-23
Contact:
Chuan-Kuan WANG
Ying JIN, Chuan-Kuan WANG, Zheng-Hu ZHOU. Mechanisms of xylem embolism repair in woody plants: Research progress and questions[J]. Chin J Plan Ecolo, 2016, 40(8): 834-846.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.17521/cjpe.2016.0118
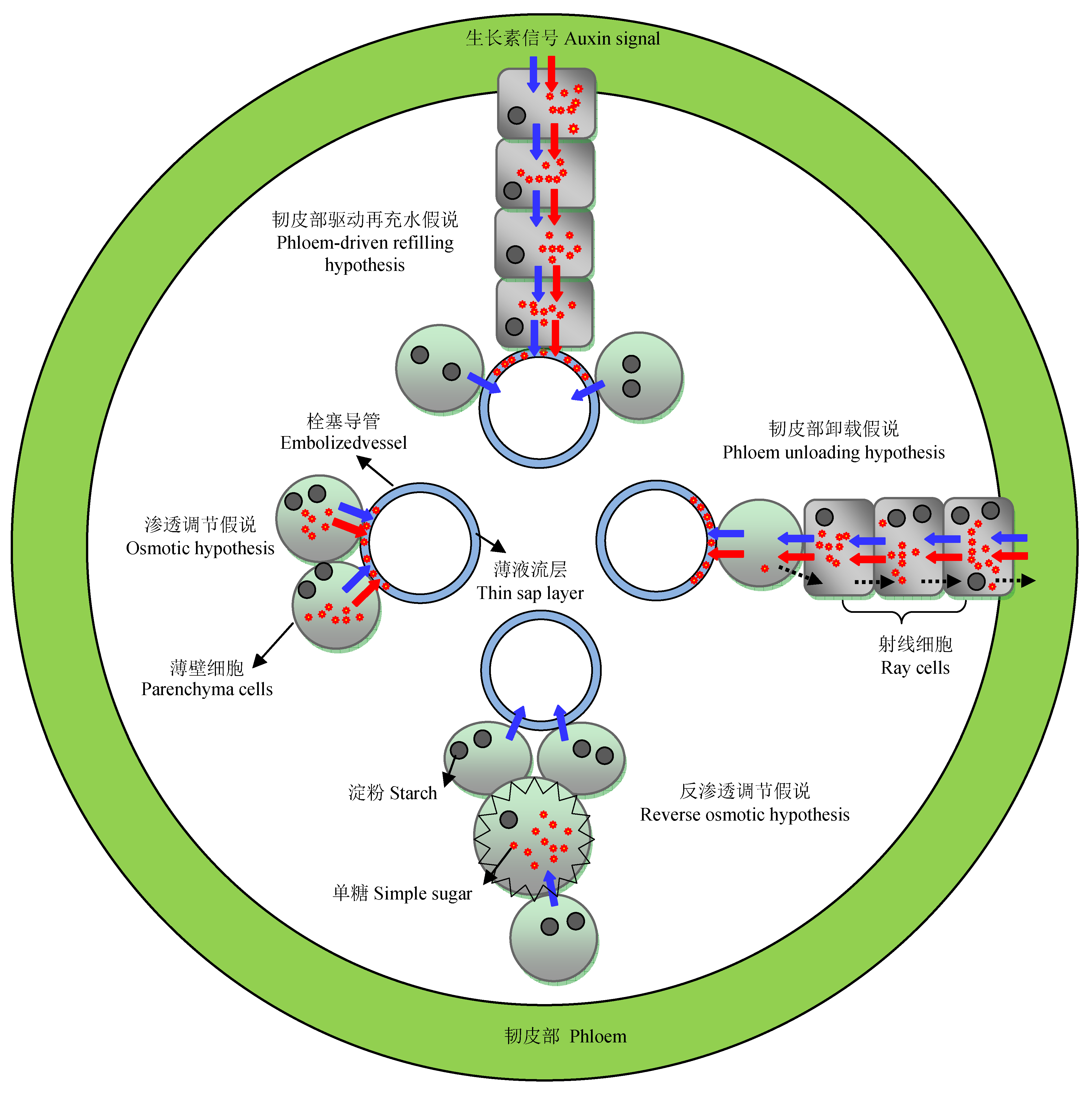
Fig. 1 Four hypotheses for the mechanisms of the novel refilling in xylem embolism repair. Red and blue arrows represent solutes and water flow, respectively; the dash arrows represent the signal from parenchyma cells.
| [1] | Ackerly DD, Reich PB (1999). Convergence and correlations among leaf size and function in seed plants: A comparative test using independent contrasts. American Journal of Botany, 86, 1272-1281. |
| [2] | Améglio T, Decourteix M, Alves G, Valentin V, Sakr S, Julien JL, Petel G, Guilliot A, Lacointe A (2004). Temperature effects on xylem sap osmolarity in walnut trees: Evidence for a vitalistic model of winter embolism repair. Tree Physiology, 24, 785-793. |
| [3] | Brodersen CR, McElrone AJ (2013). Maintenance of xylem network transport capacity: A review of embolism repair in vascular plants. Frontiers in Plant Science, 4, 1-11. |
| [4] | Brodersen CR, McElrone AJ, Choat B, Lee EF, Shackel KA, Matthews MA (2013). In vivo visualizations of drought- induced embolism spread in Vitis vinifera. Plant Physiology, 161, 1820-1829. |
| [5] | Brodersen CR, McElrone AJ, Choat B, Matthews MA, Shackel KA (2010). The dynamics of embolism repair in xylem: In vivo visualizations using high-resolution computed tomography. Plant Physiology, 154, 1088-1095. |
| [6] | Brodribb TJ, Bowman DJMS, Nichols S, Delzon S, Burlett R (2010). Xylem function and growth rate interact to determine recovery rates after exposure to extreme water deficit. New Phytologist, 188, 533-542. |
| [7] | Brodribb TJ, Cochard H (2009). Hydraulic failure defines the recovery and point of death in water-stressed conifers. Plant Physiology, 149, 575-584. |
| [8] | Brodribb TJ, Pittermann J, Coomes DA (2012). Elegance versus speed: Examining the competition between conifer and angiosperm trees. International Journal of Plant Sciences, 173, 673-694. |
| [9] | Bucci SJ, Scholz FG, Goldstein G, Meinzer FC, Sternberg LDASL (2003). Dynamic changes in hydraulic conduc- tivity in petioles of two savanna tree species: Factors and mechanisms contributing to the refilling of embolized vessels, Plant, Cell & Environment, 26, 1633-1645. |
| [10] | Canny MJ (1993). Transfusion tissue of pine needles as a site of retrieval of solutes from the transpiration stream. New Phytologist, 123, 227-232. |
| [11] | Carnicer J, Barbeta A, Sperlich D, Coll M, Penuelas J (2013). Contrasting trait syndromes in angiosperms and conifers are associated with different responses of tree growth to temperature on a large scale. Front Plant Science, 4, 409. |
| [12] | Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009). Towards a worldwide wood economics spectrum. Ecology Letters, 12, 351-366. |
| [13] | Choat B, Cobb AR, Jansen S (2008). Structure and function of bordered pits: New discoveries and impacts on whole-plant hydraulic function. New Phytologist, 177, 608-626. |
| [14] | Choat B, Jansen S, Brodribb TJ, Cochard H, Delzon S, Bhaskar R, Bucci SJ, Feild TS, Gleason SM, Hacke UG, Jacobsen AL, Lens F, Maherali H, Martinez-Vilalta JM, Mayr S, Mencuccini M, Mitchell PJ, Nardini A, Pittermann J, Pratt RB, Sperry JS, Westoby M, Wright IJ, Zanne AE (2012). Global convergence in the vulnerability of forests to drought. Nature, 491, 752-755. |
| [15] | Choat B, Lahr EC, Melcher PJ, Zwieniecki MA, Holbrook NM (2005). The spatial pattern of air seeding thresholds in mature sugar maple trees. Plant, Cell & Environment, 28, 1082-1089. |
| [16] | Christman MA, Sperry JS, Smith DD (2012). Rare pits, large vessels and extreme vulnerability to cavitation in a ring-porous tree species. New Phytologist, 193, 713-720. |
| [17] | Cochard H, Badel E, Herbette S, Delzon S, Choat B, Jansen S (2013). Methods for measuring plant vulnerability to cavitation: A critical review. Journal of Experimental Botany, ert193. |
| [18] | Cochard H, Delzon S (2013). Hydraulic failure and repair are not routine in trees. Annals of Forest Science, 70, 659-661. |
| [19] | Cochard H, Delzon S, Badel E (2014). X-ray microtomography (micro-CT): A reference technology for high-resolution quantification of xylem embolism in trees. Plant, Cell & Environment, 38, 201-206. |
| [20] | Cochard H, Holtta T, Herbette S, Delzon S, Mencuccini M (2009). New insights into the mechanisms of water-stress- induced cavitation in conifers. Plant Physiology, 151, 949-954. |
| [21] | Comstock JP (1999). Why Canny’s theory doesn’t hold water. American Journal of Botany, 86, 1077-1081. |
| [22] | Davis SD, Sperry JS, Hacke UG (1999). The relationship between xylem conduit diameter and cavitation caused by freezing. American Journal of Botany, 86, 1367-1372. |
| [23] | Ewers FW, Améglio T, Cochard H, Beaujard F, Martignac M, Vandame M, Bodet C, Cruiziat AP (2001). Seasonal variation in xylem pressure of walnut trees: Root and stem pressures. Tree Physiology, 21, 1123-1132. |
| [24] | Hacke U, Sperry JS (2003). Limits to xylem refilling under negative pressure in Laurus nobilis and Acer negundo. Plant, Cell & Environment, 26, 303-311. |
| [25] | Hacke UG, Stiller V, Sperry JS, Pittermann J, McCulloh KA (2001). Cavitation fatigue. Embolism and refilling cycles can weaken the cavitation resistance of xylem. Plant Physiology, 125, 779-786. |
| [26] | Holbrook NM, Ahrens ET, Burns MJ, Zwieniecki MA (2001). In vivo observation of cavitation and embolism repair using magnetic resonance imaging. Plant Physiology, 126, 27-31. |
| [27] | Holbrook NM, Zwieniecki MA (1999). Embolism repair and xylem tension: Do we need a miracle? Plant Physiology, 120, 7-10. |
| [28] | Hölttä T, Vesala T, Perämäki M, Nikinmaa E (2006). Refilling of embolised conduits as a consequence of ‘Munch water’ circulation. Functional Plant Biology, 33, 949-959. |
| [29] | Ishida A, Nakano T, Yazaki K, Matsuki S, Koike N, Lauenstein DL, Shimizu M, Yamashita N (2008). Coordination between leaf and stem traits related to leaf carbon gain and hydraulics across 32 drought-tolerant angiosperms. Oecologia, 156, 193-202. |
| [30] | Jansen S, Choat B, Pletsers A (2009). Morphological variation of intervessel pit membranes and implications to xylem function in angiosperms. American Journal of Botany, 96, 409-419. |
| [31] | Johnson DM, McCulloh KA, Woodruff DR, Meinzer FC (2012). Hydraulic safety margins and embolism reversal in stems and leaves: Why are conifers and angiosperms so different? Plant Science, 195, 48-53. |
| [32] | Johnson DM, Woodruff DR, McCulloh KA, Meinzer FC (2009). Leaf hydraulic conductance, measured in situ, declines and recovers daily: Leaf hydraulics, water potential and stomatal conductance in four temperate and three tropical tree species. Tree Physiology, 29, 879-887. |
| [33] | Knipfer T, Cuneo I, Brodersen C, McElrone AJ (2016). In-situ visualization of the dynamics in xylem embolism formation and removal in the absence of root pressure: A study on excised grapevine stems. Plant Physiology, doi: 10.1104/pp.16.00136. |
| [34] | Knipfer T, Eustis A, Brodersen C, Walker AM, McElrone AJ (2015). Grapevine species from varied native habitats exhibit differences in embolism formation/repair associated with leaf gas exchange and root pressure. Plant, Cell & Environment, 38, 1503-1513. |
| [35] | Laur J, Hacke UG (2013). Transpirational demand affects aquaporin expression in poplar roots. Journal of Experimental Botany, 64, 2283-2293. |
| [36] | Leng HN (2012). Cloning and Expression of PIPs Gene in Populus and Aquaporins Role in Embolism Recovery. PhD dissertation, Chinese Academy of Forestry, Beijing. (in Chinese).[冷华妮 (2012). 植物栓塞修复机制与质膜内在水通道蛋白基因的克隆、表达和转基因研究. 博士学位论文, 中国林业科学研究院, 北京.] |
| [37] | Li R, Jiang ZM, Zhang SX, Cai J (2015). A review of new research progress on the vulnerability of xylem embolism of woody plants. Chinese Journal of Plant Ecology, 39, 838-848. (in Chinese with English abstract)[李荣, 姜在民, 张硕新, 蔡靖 (2015). 木本植物木质部栓塞脆弱性研究新进展. 植物生态学报, 39, 838-848.] |
| [38] | Maherali H, Moura CF, Caldeira MC, Willson CJ, Jackson RB (2006). Functional coordination between leaf gas exchange and vulnerability to xylem cavitation in temperate forest trees. Plant, Cell & Environment, 29, 571-583. |
| [39] | McCulloh K, Sperry JS, Lachenbruch B, Meinzer FC, Reich PB (2010). Moving water well: Comparing hydraulic efficiency in twigs and trunks of coniferous, ring-porous, and diffuse-porous saplings from temperate and tropical forests. New Phytologist, 186, 439-450. |
| [40] | McCulloh KA, Meinzer FC (2015). Further evidence that some plants can lose and regain hydraulic function daily. Tree Physiology, 35, 691-693. |
| [41] | McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, Yepez EA (2008). Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytologist, 178, 719-739. |
| [42] | McElrone AJ, Brodersen CR, Alsina MM, Drayton WM, Matthews MA, Shackel KA, Wada H, Zufferey V, Choat B (2012). Centrifuge technique consistently overestimates vulnerability to water stress-induced cavitation in grapevines as confirmed with high-resolution computed tomography. New Phytologist, 196, 661-665. |
| [43] | Meinzer FC, Johnson DM, Lachenbruch B, McCulloh KA, Woodruff DR (2009). Xylem hydraulic safety margins in woody plants: Coordination of stomatal control of xylem tension with hydraulic capacitance. Functional Ecology, 23, 922-930. |
| [44] | Meinzer FC, McCulloh KA (2013). Xylem recovery from drought-induced embolism: Where is the hydraulic point of no return? Tree Physiology, 33, 331-334. |
| [45] | Nardini A, Lo Gullo MA, Salleo S (2011). Refilling embolized xylem conduits: Is it a matter of phloem unloading? Plant Science, 180, 604-611. |
| [46] | O’Brien MJ, Leuzinger S, Philipson CD, Tay J, Hector A (2014). Drought survival of tropical tree seedlings enhanced by non-structural carbohydrate levels. Nature Climate Change, 4, 710-714. |
| [47] | Ogasa M, Miki NH, Murakami Y, Yoshikawa K (2013). Recovery performance in xylem hydraulic conductivity is correlated with cavitation resistance for temperate deciduous tree species. Tree Physiology, 33, 335-344. |
| [48] | Panshin AJ, Zeeuw CD (1980). Textbook of Wood Technology. McGraw-Hill, New York. |
| [49] | Perrone I, Pagliarani C, Lovisolo C, Chitarra W, Roman F, Schu-bert A (2012). Recovery from water stress affects grape leaf petiole transcriptome. Planta, 235, 1383-1396. |
| [50] | Pittermann J, Sperry J (2003). Tracheid diameter is the key trait determining the extent of freezing-induced embolism in conifers. Tree Physiology, 23, 907-914. |
| [51] | Pittermann J, Sperry JS, Hacke UG, Wheeler JK, Sikkema EH (2005). Torus-margo pits help conifers compete with angiosperms. Science, 310, 1924-1924. |
| [52] | Plavcová L, Jansen S, Klepsch M, Hacke U (2012). Nobody’s perfect: Can irregularities in pit structure influence vulnerability to cavitation? Frontiers in Plant Science, 4, 453-453. |
| [53] | Poorter L, McDonald I, Alarcón A, Fichtler E, Licona JC, Peña-Claros M, Sterck F, Villegas Z, Sass-Klaassen U (2010). The importance of wood traits and hydraulic con- ductance for the performance and life history strategies of 42 rainforest tree species. New Phytologist, 185, 481-492. |
| [54] | Pou A, Medrano H, Flexas J, Tyerman SD (2013). A putative role for TIP and PIP aquaporins in dynamics of leaf hydraulic and stomatal conductances in grapevine under water stress and re-watering. Plant, Cell & Environment, 36, 828-843. |
| [55] | Rolland V, Dana MB, Lenné T, Bryant G, Chen H, Wolfe J, Holbrook NM, Stanton DE, Ball MC (2015). Easy come, easy go: Capillary forces enable rapid refilling of embolized primary xylem vessels. Plant Physiology, 168, 1636-1647. |
| [56] | Salleo S, Lo Gullo MA, de Paoli D, Zippo M (1996). Xylem recovery from cavitation-induced embolism in young plants of Laurus nobilis: A possibile mechanism. New Phytologist, 132, 47-56. |
| [57] | Salleo S, Lo Gullo MA, Trifilò P, Nardini A (2004). New evidence for a role of vessel-associated cells and phloem in the rapid xylem refilling of cavitated stems of Laurus nobilis L. Plant, Cell & Environment, 27, 1065-1076. |
| [58] | Salleo S, Nardini A, Pitt F, Lo Gullo MA (2000). Xylem cavitation and hydraulic control of stomatal conductance in laurel (Laurus nobilis L.). Plant, Cell & Environment, 23, 71-79. |
| [59] | Salleo S, Trifilò P, Esposito S, Nardini A, Lo Gullo MA (2009). Starch-to-sugar conversion in wood parenchyma of field-growing Laurus nobilis plants: A component of the signal pathway for embolism repair? Functional Plant Biology, 36, 815-825. |
| [60] | Salleo S, Trifilò P, Lo Gullo MA (2006). Phloem as a possible major determinant of rapid cavitation reversal in stems of Laurus nobilis (laurel). Functional Plant Biology, 33, 1063-1074. |
| [61] | Scheenen TWJ, Vergeldt FJ, Heemskerk AM, van As H (2007). Intact plant magnetic resonance imaging to study dynamics in long-distance sap flow and flow-conducting surface area. Plant Physiology, 144, 1157-1165. |
| [62] | Scoffoni C, Sack L (2014). Are leaves “freewheelin”? Testing for a Wheeler-type effect in leaf xylem hydraulic decline. Plant, Cell & Environment, 38, 534-543. |
| [63] | Secchi F, Gilbert ME, Zwieniecki MA (2011). Transcriptome response to embolism formation in stems of Populus trichocarpa provides insight into signaling and the biology of refilling. Plant Physiology, 157, 1419-1429. |
| [64] | Secchi F, Zwieniecki MA (2010). Patterns of PIP gene expression in Populus trichocarpa during recovery from xylem embolism suggest a major role for the PIP1 aquaporin subfamily as moderators of refilling process. Plant, Cell & Environment, 33, 1285-1297. |
| [65] | Secchi F, Zwieniecki MA (2011). Sensing embolism in xylem vessels: The role of sucrose as a trigger for refilling. Plant, Cell & Environment, 34, 514-524. |
| [66] | Secchi F, Zwieniecki MA (2012). Analysis of xylem sap from functional (nonembolized) and nonfunctional (embolized) vessels of Populus nigra: Chemistry of refilling. Plant Physiology, 160, 955-964. |
| [67] | Secchi F, Zwieniecki MA (2014). Down-regulation of plasma intrinsic protein1 aquaporin in poplar trees is detrimental to recovery from embolism. Plant Physiology, 164, 1789-1799. |
| [68] | Sperry JS (2013). Cutting-edge research or cutting-edge arte- fact? An overdue control experiment complicates the xy- lem refilling story. Plant, Cell & Environment, 11, 35-40. |
| [69] | Sperry JS, Donnelly JR, Tyree MT (1988). A method for measuring hydraulic conductivity and embolism in xylem. Plant, Cell & Environment, 11, 35-45. |
| [70] | Sperry JS, Hacke UG (2004). Analysis of circular bordered pit function I. Angiosperm vessels with homogenous pit membranes. American Journal of Botany, 91, 369-385. |
| [71] | Sperry JS, Holbrook NM, Zimmermann MH, Tyree MT (1987). Spring filling of xylem vessels in wild grapevine. Plant Physiology, 83, 414-417. |
| [72] | Sperry JS, Tyree MT (1990). Water-stress-induced xylem embolism in three species of conifers. Plant, Cell & Environment, 13, 427-436. |
| [73] | Stiller V, Sperry JS (2002). Cavitation fatigue and its reversal in sunflower (Helianthus annuus L.). Journal of Experimental Botany, 53, 1155-1161. |
| [74] | Torres-Ruiz JM, Jansen S, Choat B, McElrone AJ, Cochard H, Brodribb TJ, Badel E, Burlett R, Bouche PS, Brodersen CR, Li S, Morris H, Delzon S (2015). Direct X-ray microtomography observation confirms the induction of embolism upon xylem cutting under tension. Plant Physiology, 167, 40-43. |
| [75] | Trifilò P, Nardini A, Lo Gullo MA, Barbera PM, Savi T, Raimondo F (2015). Diurnal changes in embolism rate in nine dry forest trees: Relationships with species-specific xylem vulnerability, hydraulic strategy and wood traits. Tree Physiology, 35, 694-705. |
| [76] | Trifilò P, Raimondo F, Lo Gullo MA, Barbera PM, Salleo S, Nardini A (2014). Relax and refill: Xylem rehydration prior to hydraulic measurements favours embolism repair in stems and generates artificially low PLC values. Plant, Cell & Environment, 37, 2491-2499. |
| [77] | Tyree MT, Salleo S, Nardini A, Lo Gullo MA, Mosca R (1999). Refilling of embolized vessels in young stems of laurel. Do we need a new paradigm? Plant Physiology, 120, 11-21. |
| [78] | Urli M, Porté A, Cochard H, Guengant Y, Burlett R, Delzon S (2013). Xylem embolism threshold for catastrophic hydraulic failure in angiosperm trees. Tree Physiology, 33, 672-683. |
| [79] | Venturas MD, Mackinnon ED, Jacobsen AL, Pratt RB (2015). Excising stem samples underwater at native tension does not induce xylem cavitation. Plant, Cell & Environment, 38, 1060-1068. |
| [80] | Vesala T, Hölttä T, Perämäki M, Nikinmaa E (2003). Refilling of a hydraulically isolated embolized xylem vessel: Model calculations. Annuals of Botany, 91, 419-428. |
| [81] | Wheeler JK, Huggett BA, Tofte AN, Rockwell FE, Holbrook NM (2013). Cutting xylem under tension or supersaturated with gas can generate PLC and the appearance of rapid recovery from embolism. Plant, Cell & Environment, 36, 1938-1949. |
| [82] | Zimmermann MH (1983). Xylem Structure and the Ascent of Sap. Springer-Verlag, Berlin. |
| [83] | Zwieniecki MA, Holbrook NM (1998). Diurnal variation in xylem hydraulic conductivity in white ash (Fraxinus americana L.) red maple (Acer rubrum L.) and red spruce (Picea rubens Sarg.). Plant, Cell & Environment, 21, 1173-1180. |
| [84] | Zwieniecki MA, Holbrook NM (2000). Bordered pit structure and vessel wall surface properties. Implications for embolism repair. Plant Physiology, 123, 1015-1020. |
| [85] | Zwieniecki MA, Holbrook NM (2009). Confronting Maxwell’s demon: Biophysics of xylem embolism repair. Trends in Plant Science, 14, 530-534. |
| [86] | Zwieniecki MA, Hutyra L, Thompson MV, Holbrook NM (2000). Dynamic changes in petiole specific conductivity in red maple (Acer rubrum L.) tulip tree (Liriodendron tulipifera L.) and northern fox grape (Vitis labrusca L.). Plant, Cell & Environment, 23, 407-414. |
| [87] | Zwieniecki MA, Melcher PJ, Ahrens ET (2013). Analysis of spatial and temporal dynamics of xylem refilling in Acer rubrum L. using magnetic resonance imaging. Frontiers in Plant Science, 4, 265. |
| [1] | WU Min, TIAN Yu, FAN Da-Yong, ZHANG Xiang-Xue. Hydraulic regulation of Populus tomentosa and Acer truncatum under drought stress [J]. Chin J Plant Ecol, 2022, 46(9): 1086-1097. |
| [2] | LUO Dan-Dan, WANG Chuan-Kuan, JIN Ying. Response mechanisms of hydraulic systems of woody plants to drought stress [J]. Chin J Plant Ecol, 2021, 45(9): 925-941. |
| [3] | LI Zhi-Min, WANG Chuan-Kuan. Research progress on responses of xylem of woody plants to freeze-thaw embolism [J]. Chin J Plant Ecol, 2019, 43(8): 635-647. |
| [4] | Dan-Dan LUO, Chuan-Kuan WANG, Ying JIN. Plant water-regulation strategies: Isohydric versus anisohydric behavior [J]. Chin J Plan Ecolo, 2017, 41(9): 1020-1032. |
| [5] | LI Rong,JIANG Zai-Min,ZHANG Shuo-Xin,CAI Jing. A review of new research progress on the vulnerability of xylem embolism of woody plants [J]. Chin J Plan Ecolo, 2015, 39(8): 838-848. |
| [6] | HUANG Ju-Ying, YU Hai-Long, ZHANG Shuo-Xin. EFFECTS OF POTASSIUM ADDITION AND WATER SUPPLY ON XYLEM EMBOLISM IN ACER TRUNCATUMAND LIGUSTRUM LUCIDUM [J]. Chin J Plant Ecol, 2009, 33(6): 1199-1207. |
| [7] | HUANG Ju-Ying, CAI Jing, JIANG Zai-Min, ZHANG Shuo-Xin, YU Hai-Long. RESPONSE OF XYLEM EMBOLISM TO PHOSPHORUS ADDITION UNDER DIFFERENT WATER REGIMES IN TWO TREE SPECIES [J]. Chin J Plant Ecol, 2008, 32(1): 183-188. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2022 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn