

Chin J Plan Ecolo ›› 2015, Vol. 39 ›› Issue (7): 753-761.DOI: 10.17521/cjpe.2015.0072
• Orginal Article • Previous Articles Next Articles
HUANG Yan-Bo1, WEI Yu-Kun1,*( ), WANG Qi1, XIAO Yue-E1, YE Xi-Yang2
), WANG Qi1, XIAO Yue-E1, YE Xi-Yang2
Online:2015-07-01
Published:2015-07-22
Contact:
Yu-Kun WEI
About author:# Co-first authors
HUANG Yan-Bo,WEI Yu-Kun,WANG Qi,XIAO Yue-E,YE Xi-Yang. Floral morphology and pollination mechanism of Salvia liguliloba, a narrow endemic species with degraded lever-like stamens[J]. Chin J Plan Ecolo, 2015, 39(7): 753-761.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.17521/cjpe.2015.0072

Fig. 1 Photographic images of Salvia liguliloba and its visiting insects. A, Habitat. B, Plant. C, Lateral view of flower. D, Longitudinal section of flower. E, Pistil. F, Front view of flower. G, Stamens. H, Fruits. I, Bombus trifasciatus (pollinator). J, Ventral visiting by a species of Halictidae. K, Dorsal visiting by a species of Halictidae. L, A visitor of Sphingidae species. Bar = 2 mm.
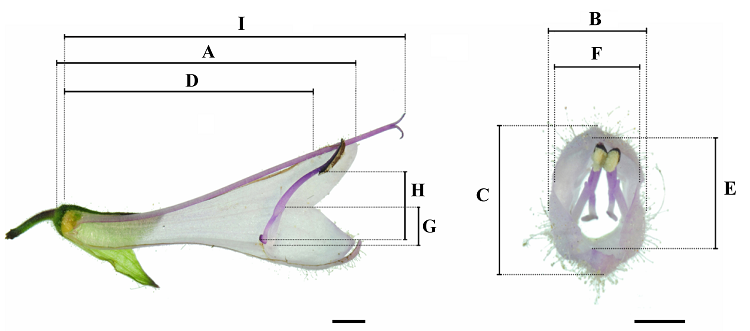
Fig. 2 Diagram of Salvia liguliloba flower measurement. A, Corolla length. B, Corolla width. C, Corolla height. D, Tube length. E, Entrance height. F, Entrance width. G, Filament length. H, Connective length. I, Pistil length. Bar = 2 mm.

Fig. 3 Comparisons of morphometric data between Salvia liguliloba (A to D) and S. digitaloides (E to H). A, E, Lateral view of a flower. B, F, Longitudinal section of a flower. C, G, Front view of a flower. D, H, Stamens. Bar = 5 mm.
| 花冠长 Corolla Length | 花冠宽 Corolla width | 花冠高 Corolla height | 冠筒长 Tube Length | 冠口高 Entrance height | 冠口宽 Entrance width | 花丝长 Filament length | 药隔长 Connective length | 雌蕊长 Pistil Length | |
|---|---|---|---|---|---|---|---|---|---|
| 舌瓣鼠尾草 S. liguliloba | 23.67 ± 0.74b | 5.31 ± 0.34b | 7.30 ± 0.67b | 19.49 ± 0.8b | 4.87 ± 0.61a | 4.41 ± 0.52b | 2.39 ± 0.14b | 5.19 ± 0.33a | 23.74 ± 0.43b |
| 毛地黄鼠尾草 S. digitaloides | 39.80 ± 2.60a | 12.52 ± 1.49a | 22.98 ± 2.62a | 28.99 ± 2.01a | 4.33 ± 0.68b | 6.80 ± 0.72a | 7.33 ± 0.46a | 5.10 ± 0.43a | 34.89 ± 2.30a |
Table 1 Comparison between Salvia liguliloba and S. digitaloides on flower features (mean ± SD) (mm)
| 花冠长 Corolla Length | 花冠宽 Corolla width | 花冠高 Corolla height | 冠筒长 Tube Length | 冠口高 Entrance height | 冠口宽 Entrance width | 花丝长 Filament length | 药隔长 Connective length | 雌蕊长 Pistil Length | |
|---|---|---|---|---|---|---|---|---|---|
| 舌瓣鼠尾草 S. liguliloba | 23.67 ± 0.74b | 5.31 ± 0.34b | 7.30 ± 0.67b | 19.49 ± 0.8b | 4.87 ± 0.61a | 4.41 ± 0.52b | 2.39 ± 0.14b | 5.19 ± 0.33a | 23.74 ± 0.43b |
| 毛地黄鼠尾草 S. digitaloides | 39.80 ± 2.60a | 12.52 ± 1.49a | 22.98 ± 2.62a | 28.99 ± 2.01a | 4.33 ± 0.68b | 6.80 ± 0.72a | 7.33 ± 0.46a | 5.10 ± 0.43a | 34.89 ± 2.30a |
| 访花昆虫 Visiting insect | 体长 Body length | 胸宽 Thorax width | 胸厚 Thorax thickness | 喙长 Tongue length |
|---|---|---|---|---|
| 三条熊蜂 Bombus trifasciatus (n = 10) | 21.8 ± 1.29 | 7.35 ± 0.41 | 5.10 ± 0.24 | 11.0 ± 1.32 |
| 隧蜂科昆虫 Halictidae (n = 1) | 6.88 | 1.91 | 1.66 | 0.73 |
Table 2 Morphometric data of visiting insects from Salvia liguliloba in the study (mean ± SD) (mm)
| 访花昆虫 Visiting insect | 体长 Body length | 胸宽 Thorax width | 胸厚 Thorax thickness | 喙长 Tongue length |
|---|---|---|---|---|
| 三条熊蜂 Bombus trifasciatus (n = 10) | 21.8 ± 1.29 | 7.35 ± 0.41 | 5.10 ± 0.24 | 11.0 ± 1.32 |
| 隧蜂科昆虫 Halictidae (n = 1) | 6.88 | 1.91 | 1.66 | 0.73 |
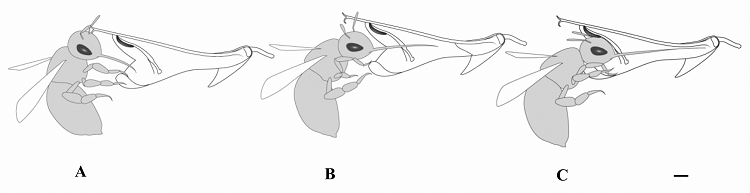
Fig. 4 The pollination process of Bombus trifasciatus, an effective pollinator of Salvia liguliloba. A, Approaching to the corolla. B, Accessing to flower tube and the pollinator’s tongue is extending. C, B. trifasciatus is sucking nectar and its forehead touching the fertile anther. Bar = 2 mm.
| 访花昆虫 Visiting insects | 相对频次 Relative frequency | 单花访问时间 Visiting time per flower (s) | 活跃度 Activity rate | 访花频率 Visitation rate | 传粉方式 Pollination mode | |
|---|---|---|---|---|---|---|
| 舌瓣鼠尾草传粉昆虫 Pollinator of S. liguliloba | 三条熊蜂 Bombus trifasciatus | 0.959 ± 0.065 (n = 7) | 1.54 ± 0.60 (n = 1 035) | 18.2 ± 3.12 (n = 10) | 17.45 | F |
| 毛地黄鼠尾草传粉昆虫 Pollinators of S. digitaloides | 拟熊蜂属昆虫 Psithyrus sp. | 0.57 | 8.68 ± 0.90 | - | 3.94 | D |
| 小雅熊蜂 B. lepidus | 0.20 | 15.57 ± 1.20 | - | 0.77 | D/V | |
| B. infrequens | < 0.20 | - | - | - | D |
Table 3 Flower-visiting traits of pollinators from Salvia liguliloba and S. digitaloides (mean ± SD)
| 访花昆虫 Visiting insects | 相对频次 Relative frequency | 单花访问时间 Visiting time per flower (s) | 活跃度 Activity rate | 访花频率 Visitation rate | 传粉方式 Pollination mode | |
|---|---|---|---|---|---|---|
| 舌瓣鼠尾草传粉昆虫 Pollinator of S. liguliloba | 三条熊蜂 Bombus trifasciatus | 0.959 ± 0.065 (n = 7) | 1.54 ± 0.60 (n = 1 035) | 18.2 ± 3.12 (n = 10) | 17.45 | F |
| 毛地黄鼠尾草传粉昆虫 Pollinators of S. digitaloides | 拟熊蜂属昆虫 Psithyrus sp. | 0.57 | 8.68 ± 0.90 | - | 3.94 | D |
| 小雅熊蜂 B. lepidus | 0.20 | 15.57 ± 1.20 | - | 0.77 | D/V | |
| B. infrequens | < 0.20 | - | - | - | D |
| [1] | Claßen-Bockhoff R, Speck T, Tweraser E, Wester P, Thimm S, Reith M (2004). The staminal lever mechanism in Salvia L. (Lamiaceae): A key innovation for adaptive radiation?Organisms Diversity & Evolution, 4, 189-205. |
| [2] | Claßen-Bockhoff R, Wester P, Tweraser E (2003). The staminal lever mechanism in Salvia L. (Lamiaceae)—A review.Plant Biology, 5, 33-41. |
| [3] | Fang Q, Huang SQ (2014). Progress in pollination ecology at the community level.Chinese Science Bulletin, 59, 449-458. |
| (in Chinese with English abstract) [方强, 黄双全 (2014). 群落水平上传粉生态学的研究进展. 科学通报, 59, 449-458.] | |
| [4] | Feng JM, Wang XP, Xu CD, Yang YH, Fang JY (2006). Altitudinal patterns of plant species diversity and community structure on Yulong Mountains, Yunnan, China.Journal of Mountain Science, 24, 110-116. |
| (in Chinese with English abstract) [冯建孟, 王襄平, 徐成东, 杨元合, 方精云 (2006). 玉龙雪山植物物种多样性和群落结构沿海拔梯度的分布格局. 山地学报, 24, 110-116.] | |
| [5] | Gong YB, Huang SQ (2007). On methodology of foraging behavior of pollinating insects.Biodiversity Science, 15, 576-583. |
| (in Chinese with English abstract) [龚燕兵, 黄双全 (2007). 传粉昆虫行为的研究方法探讨. 生物多样性, 15, 576-583.] | |
| [6] | Huang SQ (2007). Studies on plant-pollinator interaction and its significances.Biodiversity Science, 15, 569-575. |
| (in Chinese with English abstract) [黄双全 (2007). 植物与传粉者相互作用的研究及其意义. 生物多样性, 15, 569-575.] | |
| [7] | Huang SQ, Guo YH (2000). New advances in pollination biology.Chinese Science Bulletin, 45, 225-237. |
| (in Chinese) [黄双全, 郭友好 (2000). 传粉生物学的研究进展. 科学通报, 45, 225-237.] | |
| [8] | Huang YB, Wei YK, Ge BJ, Wang Q (2014). Pollination Mechanisms of genus Salvia (Lamiaceae) in East Asia (China).Acta Ecologica Sinica, 34, 2282-2289. |
| (in Chinese with English abstract) [黄艳波, 魏宇昆, 葛斌杰, 王琦 (2014). 鼠尾草属东亚分支的传粉模式. 生态学报, 34, 2282-2289.] | |
| [9] | Li QQ, Li MH, Yuan QJ, Cui ZH, Huang LQ, Xiao PG (2013) Phylogenetic relationships of Salvia (Lamiaceae) in China: Evidence from DNA sequence datasets.Journal of Systematics and Evolution, 51, 184-195. |
| [10] | Talavera S, Bastida F, Ortiz PL, Arista M (2001). Pollinator attendance and reproductive success in Cistus libanotis L. (Cistaceae).International Journal of Plant Sciences, 162, 343-352. |
| [11] | Wang Q, Wei YK, Huang YB (2015). Research on distribution pattern of Subg. Salvia Benth. (Lamiaceae), an important group of medicinal plants in East Asia.Acta Ecologica Sinica, 35, 1470-1479. |
| (in Chinese with English abstract) [王琦, 魏宇昆, 黄艳波 (2015). 中国弧隔鼠尾草亚属(唇形科)的分布格局. 生态学报, 35, 1470-1479.] | |
| [12] | Wei YK, Wang Q, Huang YB (2015). Species diversity and distribution of Salvia (Lamiaceae).Biodiversity Science, 23, 3-10. |
| (in Chinese with English abstract) [魏宇昆, 王琦, 黄艳波 (2015). 唇形科鼠尾草属的物种多样性与分布. 生物多样性, 23, 3-10.] | |
| [13] | Xin HJ, He YQ, Li ZX, Wang SJ, Du JK, Wang CF, Pu T, Zhang W (2012). Inter-annual variation of temperature and precipitation gradient at the Eastern slope of Yulong Snow Mountain. Earth Science-Journal of China University of Geosciences, 37(Suppl.), 188-194. |
| (in Chinese with English abstract) [辛惠娟, 何元庆, 李宗省, 王世金, 杜建括, 王春凤, 蒲焘, 张蔚 (2012). 玉龙雪山东坡气温和降水梯度年内变化特征. 地球科学——中国地质大学学报, 37(Suppl.), 188-194.] | |
| [14] | Xin HJ, He YQ, Zhang T, Niu HW, Du JK (2013). The features of climate variation and glacier response in Mt. Yulong, southeastern Tibetan Plateau.Advances in Earth Science, 28, 1257-1268. |
| (in Chinese with English abstract) [辛惠娟, 何元庆, 张涛, 牛贺文, 杜建括 (2013). 青藏高原东南缘丽江玉龙雪山气候变化特征及其对冰川变化的影响. 地球科学进展, 28, 1257-1268.] | |
| [15] | Yang FC (1992). Comprehensive Investigation Report on Natural Resource of Tianmu Mountain Nature Reserve. Zhejiang Science and Technology Press, Hangzhou. |
| (in Chinese) [杨逢春 (1992). 天目山自然保护区自然资源综合考察报告. 浙江科学技术出版社, 杭州.] | |
| [16] | Zhang B, Claßen-Bockhoff R, Zhang ZQ, Sun S, Luo YJ, Li QJ (2011). Functional implications of the staminal lever mechanism in Salvia cyclostegia (Lamiaceae).Annals of Botany, 107, 621-628. |
| [17] | Zhang B, Li QJ (2014). Phenotypic selection on the staminal lever mechanism in Salvia digitaloides (Labiaceae).Evolutionary Ecology, 28, 373-386. |
| [18] | Zhang B, Sun S, Fang QE, Bai XM (2012). Evolutionary response of staminal lever mechanism of different species in Salvia to spatial variation in pollinators.Chinese Journal of Plant Ecology, 36, 681-689. |
| (in Chinese with English abstract) [张勃, 孙杉, 方强恩, 白小明 (2012). 鼠尾草属不同物种的雄蕊杠杆机制对传粉者空间变异的进化响应. 植物生态学报, 36, 681-689.] | |
| [19] | Zhang B, Sun S, Zhang ZQ, Li QJ (2010). A review of the evolutionary and ecological significance of lever-like stamens.Chinese Journal of Plant Ecology, 34, 89-99. |
| (in Chinese with English abstract) [张勃, 孙杉, 张志强, 李庆军 (2010). 杠杆状雄蕊及其进化生态学意义. 植物生态学报, 34, 89-99.] |
| [1] | TAN Ke, DONG Shu-Peng, LU Tao, ZHANG Ya-Jing, XU Shi-Tao, REN Ming-Xun. Diversity and evolution of samara in angiosperm [J]. Chin J Plan Ecolo, 2018, 42(8): 806-817. |
| [2] | Jun-Wei YE, Yang ZHANG, Xiao-Juan WANG. Phylogeographic breaks and the mechanisms of their formation in the Sino-Japanese floristic region [J]. Chin J Plan Ecolo, 2017, 41(9): 1003-1019. |
| [3] | YU Xiang-Qin, FENG Yu-Long, LI Qiao-Ming. Review of research advances and prospects of invasive Chromolaena odorata [J]. Chin J Plant Ecol, 2010, 34(5): 591-600. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2022 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn