

Chinese Journal of Plant Ecology >
Effects of drought stress on photosynthesis, growth and root structure of transgenic PtPIP2;8 poplar 84K (Populus alba × P. glandulosa)
Received date: 2020-03-06
Accepted date: 2020-04-23
Online published: 2020-06-12
Supported by
National Natural Science Foundation of China(31270648)
Aims In this study, changes in growth, photosynthesis and root structure in response to drought were tested in transgenic poplar 84K (Populus alba × P. glandulosa) seedlings with different expression levels of aquaporin gene (PtPIP2;8). The function of aquaporin gene PtPIP2;8 and its response to drought stress were analyzed.
Methods We selected PtPIP2;8 silencing line of poplar 84K, PtPIP2;8 overexpressing line of poplar 84K and wildtype (WT) as the experimental materials. The Real-time fluorescence quantitative PCR technique was used to detect the PtPIP2;8 expression in roots, stems and leaves. Root hydraulic conductance was measured by high pressure liquid flow meter. The photosynthetic light-response curve, and gas exchange parameters were measured by a LI-6400 photosynthetic system. Growth indexes were determined, and the root length, root surface area, root volume and total root tips were scanned and then analyzed with the root analysis software.
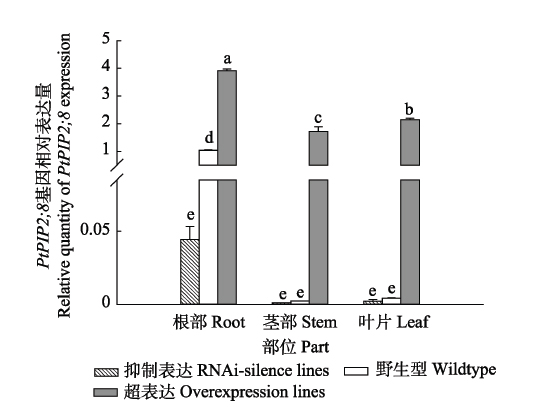
Important findings The results showed that: (1) The gene PtPIP2;8 was mainly expressed in the root system in WT, while its significant expression occurs not only in roots, but also in stems and leaves in PtPIP2;8 overexpressing poplar lines. The PtPIP2;8 RNAi-silence poplar lines only showed weak expression of PtPIP2;8 in the root, and the expression level were 1/20 and 1/80 of WT and overexpression line, respectively. (2) The root structure analysis showed that overexpression lines had significantly lower total root length, total root surface area, total root volume and total root tips than RNAi-silence line and WT, but higher root hydraulic conductance compared with RNAi-silence line and WT. These results showed that the aquaporin gene PtPIP2;8 participated in plant water transport and improved water transport efficiency. (3) Under normal water conditions, RNAi-silence lines showed lower plant height and leaf area but higher root-shoot ratio compared with overexpression line and WT. After drought stress, RNAi-silence lines only slightly decreased the net photosynthetic rate (Pn) and stomatal conductance (Gs), and maintained a relatively high Pn. Diurnal changes of Pn and Gs in RNAi-silence lines showed a single-peak pattern, in which the decrease of photosynthesis was caused by stomatal limitation. Diurnal changes of Pn in both overexpression lines and WT had a two-peak pattern, indicating the non-stomatal limitation of photosynthesis. Drought stress slightly decreased Pn of RNAi-silence lines, while largely decreased Pn of overexpression line and WT decreased, especially at 13:00 and 15:00, indicating that overexpression line and WT were more sensitive to drought stress compared with the RNAi-silence lines. (4) Under drought stress, RNAi-silence line showed the least decline in relative growth rate and total biomass, and the highest root-shoot ratio among the three poplar lines. The total root surface area, total root volume and total root tips of RNAi-silence line were significantly higher than those of WT. The results suggest that aquaporin gene PtPIP2;8 directly participates in the water transport and helps to improve the water transport efficiency, thus the transformation of aquaporin PtPIP2;8 gene may affect root development and growth of plants. Overexpression lines weaken their drought resistance with decreased root development and increased leaf area, while RNAi-silence line increases its drought resistance with reduced leaf area, increased root growth and root-shoot ratio. The results of this study indicate that aquaporin improves the efficiency of water transport across membranes, while the non-aquaporin water-conducting mechanism has greater tolerance to drought.
LIU Li-Yan, FENG Jin-Xia, LIU Wen-Xin, WAN Xian-Chong . Effects of drought stress on photosynthesis, growth and root structure of transgenic PtPIP2;8 poplar 84K (Populus alba × P. glandulosa)[J]. Chinese Journal of Plant Ecology, 2020 , 44(6) : 677 -686 . DOI: 10.17521/cjpe.2020.0058
| [1] | Aharon R, Shahak Y, Wininger S, Bendov R, Kapulnik Y, Galili G (2003). Overexpression of a plasma membrane aquaporin in transgenic tobacco improves plant vigor under favorable growth conditions but not under drought or salt stress. The Plant Cell, 15, 1-9. |
| [2] | Alexandersson E, Fraysse L, Sj?vall-Larsen S, Gustavsson S, Fellert M, Karlsson M, Johanson U, Kjellbom P (2005). Whole gene family expression and drought stress regulation of aquaporins. Plant Molecular Biology, 59, 469-484. |
| [3] | Bao ZLT, Gao L, Wang SM (2017). Physiological functions of plant aquaporin. Plant Physiology Journal, 53, 1171-1178. |
| [3] | [ 包珠拉太, 高丽, 王锁民 (2017). 植物水通道蛋白及其生理功能. 植物生理学报, 53, 1171-1178.] |
| [4] | Bots M, Feron R, Uehlein N, Weterings K, Kaldenhoff R, Mariani T (2005a). PIP1 and PIP2 aquaporins are differentially expressed during tobacco anther and stigma development. Journal of Experimental Botany, 56, 113-121. |
| [5] | Bots M, Vergeldt F, Wolters-Arts M, Weterings K, van As H, Mariani C (2005b). Aquaporins of the PIP2 class are required for efficient anther dehiscence in tobacco. Plant Physiology, 137, 1049-1056. |
| [6] | Boughalleb F, Hajlaoui H (2011). Physiological and anatomical changes induced by drought in two olive cultivars (cv Zalmati and Chemlali). Acta Physiologiae Plantarum, 33, 53-65. |
| [7] | Cai Q, Ding GJ, Wen XP (2016). Cloning of thePmPIP1 gene from Pinus massoniana and its expression with drought stress. Journal of Zhejiang A&F University, 33, 191-200. |
| [7] | [ 蔡琼, 丁贵杰, 文晓鹏 (2016). 马尾松水通道蛋白PmPIP1基因克隆及在干旱胁迫下的表达分析. 浙江农林大学学报, 33, 191-200.] |
| [8] | Chaumont F, Tyerman SD (2014). Aquaporins: highly regulated channels controlling plant water relations. Plant Physiology, 164, 1600-1618. |
| [9] | Fotiadis D, Jen? P, Mini T, Wirtz S, Müller SA, Fraysse L, Kjellbom P, Engel A (2001). Structural characterization of two aquaporins isolated from native spinach leaf plasma membranes. Journal of Biological Chemistry, 276, 1707-1714. |
| [10] | Huang J, Chen C, Zhang WX, Ding CJ, Su XH, Huang QJ (2017). Effects of drought stress on anatomical structure and photosynthetic characteristics of transgenic JERF36Populus alba × P. berolinensis seedling leaves. Scientia Silvae Sinicae, 53(5), 8-15. |
| [10] | [ 黄绢, 陈存, 张伟溪, 丁昌俊, 苏晓华, 黄秦军 (2017). 干旱胁迫对转JERF36银中杨苗木叶片解剖结构及光合特性的影响. 林业科学, 53(5), 8-15.] |
| [11] | Javot H, Maurel C (2002). The role of aquaporins in root water uptake. Annals of Botany, 90, 301-313. |
| [12] | Jiang LJ, Chen CH, Yan X, Yang SM (2018). Research progress on responsive mechanism of aquaporins to drought stress in plants. Guihaia, 38, 672-680. |
| [12] | [ 江林娟, 陈春华, 颜旭, 杨世民 (2018). 植物水通道蛋白的干旱应答机制研究进展. 广西植物, 38, 672-680.] |
| [13] | Johansson I, Karlsson M, Johanson U, Larsson C, Kjellbom P (2000). The role of aquaporins in cellular and whole plant water balance. Biochimica et Biophysica Acta, 1465, 324-342. |
| [14] | Kaldenhoff R, Grote K, Zhu JJ, Zimmermann U (1998). Significance of plasmalemma aquaporins for water-transport in Arabidopsis thaliana. The Plant Journal, 14, 121-128. |
| [15] | Kammerloher W, Fischer U, Piechottka GP, Sch?ffner AR (1994). Water channels in the plant plasma membrane cloned by immunoselection from a mammalian expression. The Plant Journal, 6, 187-199. |
| [16] | Katsuhara M, Koshio K, Shibasaka M, Hayashi Y, Hayakawa T, Kasamo K (2003). Over-expression of a barley aquaporin increased the shoot/root ratio and raised salt sensitivity in transgenic rice plants. Plant and Cell Physiology, 44, 1378-1383. |
| [17] | Knapp AK, Smith MD (2001). Variation among biomes in temporal dynamics of aboveground primary production. Science, 291, 481-484. |
| [18] | Knipfer T, Besse M, Verdeil JL, Fricke W (2011). Aquaporin- facilitated water uptake in barley (Hordeum vulgare L.) roots. Journal of Experimental Botany, 62, 4115-4126. |
| [19] | Leng HN (2012). Cloning and Expression of PIPs Gene in Populus and Aquaporins Role in Embolism Recovery. PhD dissertation, Chinese Academy of Forestry, Beijing. 23-95. |
| [19] | [ 冷华妮 (2012). 植物栓塞修复机制与质膜内在水通道蛋白基因的克隆、表达和转基因研究. 博士学位论文, 中国林业科学研究院, 北京. 23-95.] |
| [20] | Lopez F, Bousser A, Sisso?ff I, Gaspar M, Lachaise B, Hoarau J, Mahé A (2003). Diurnal regulation of water transport and aquaporin gene expression in maize roots: contribution of PIP2 proteins. Plant and Cell Physiology, 44, 1384-1395. |
| [21] | Lovisolo C, Secchi F, Nardini A, Salleo S, Buffa R, Schubert A (2007). Expression of PIP1 and PIP2 aquaporins is enhanced in olive dwarf genotypes and is related to root and leaf hydraulic conductance. Physiologia Plantarum, 130, 543-551. |
| [22] | Martre P, Morillon R, Barrieu F, North GB, Nobel PS, Chrispeels MJ (2002). Plasma membrane aquaporins play a significant role during recovery from water deficit. Plant Physiology, 130, 2101-2110. |
| [23] | Martre P, North GB, Nobel PS (2001). Hydraulic conductance and mercury-sensitive water transport for roots of Opuntia acanthocarpa in relation to soil drying and rewetting. Plant Physiology, 126, 352-362. |
| [24] | Matsunami M, Toyofuku K, Ishikawa-Sakurai J, Ogawa A, Matsunami T, Kokubun M (2016). Root development and the expression of aquaporin genes in rice seedlings under osmotic stress. Plant Production Science, 19, 315-322. |
| [25] | O’Brien M, Bertrand C, Matton DP (2002). Characterization of a fertilization-induced and developmentally regulated plasma-membrane aquaporin expressed in reproductive tissues, in the wild potato Solanum chacoense Bitt. Planta, 215, 485-493. |
| [26] | Prieto I, Armas C, Pugnaire FI (2012). Water release through plant roots: new insights into its consequences at the plant and ecosystem level. New Phytologist, 193, 830-841. |
| [27] | Schuurmans JAMJ, van Dongen JT, Rutjens BPW, Boonman A, Pieterse CMJ, Borstlap AC (2003). Members of the aquaporin family in the developing pea seed coat include representatives of the PIP, TIP, and NIP subfamilies. Plant Molecular Biology, 53, 655-667. |
| [28] | Siefritz F, Tyree MT, Lovisolo C, Schubert A, Kaldenhoff R (2002). PIP1 plasma membrane aquaporins in tobacco: from cellular effects to function in plants. The Plant Cell, 14, 869-876. |
| [29] | Steudle E, Peterson CA (1998). How does water get through roots? Journal of Experimental Botany, 49, 775-788. |
| [30] | Suga S, Imagawa S, Maeshima M (2001). Specificity of the accumulation of mRNAs and proteins of the plasma membrane and tonoplast aquaporins in radish organs. Planta, 212, 294-304. |
| [31] | Weig A, Deswarte C, Chrispeels MJ (1997). The major intrinsic protein family of Arabidopsis has 23 members that form three distinct groups with functional aquaporins in each group. Plant Physiology, 114, 1347-1357. |
| [32] | Yao QQ, Xie GS (2005). The photosynthetic stomatal and nonstomatal limitation under drought stress. Chinese Journal of Tropical Agriculture, 25(4), 80-85. |
| [32] | [ 姚庆群, 谢贵水 (2005). 干旱胁迫下光合作用的气孔与非气孔限制. 热带农业科学, 25(4), 80-85.] |
| [33] | Zelazny E, Borst JW, Muylaert M, Batoko H, Hemminga MA, Chaumont F (2007). FRET imaging in living maize cells reveals that plasma membrane aquaporins interact to regulate their subcellular localization. Proceedings of the National Academy of Sciences of the United States of America, 104, 12359-12364. |
| [34] | Zhao T, Cheng L, Wang C, You JQ, Zhu YF, Wang YX (2018). Effect of different apple scion-rootstock combinations on growth and photosynthesis characteristics. Acta Botanica Boreali-Occidentalia Sinica, 38, 1707-1716. |
| [34] | [ 赵通, 程丽, 王城, 游继权, 朱燕芳, 王延秀 (2018). 不同苹果砧穗组合的生长及光合特性. 西北植物学报, 38, 1707-1716.] |
| [35] | Zhou XY (2013). Research on 84K Populus Regeneration and Genetic Transformation System. Master degree dissertation, Northeast Forestry University, Harbin. 1-8. |
| [35] | [ 周熙莹 (2013). 84K杨再生和遗传转化体系的研究. 硕士学位论文, 东北林业大学, 哈尔滨. 1-8.] |
/
| 〈 |
|
〉 |