

Chinese Journal of Plant Ecology >
Changes in hydraulic traits of nine vascular plants from different evolutionary lineages
Online published: 2018-04-16
Supported by
Supported by the National Natural Science Foundation of China.(31670411)
Aims As vascular plants evolve from ferns to gymnosperms and angiosperms, their physiological structures and functions are assumed more adaptable to arid environment. Whether the three plant groups from early to late evolved lineages have improved their water transport and use efficiency has been studied on the basis of the morphological structure of leaf veins and stomata.Moreover, the water transportation rate was directly measured in the angiosperms. Therefore, we measured structural and functional traits related to water relations in all three plant groups simultaneously, to test the hypothesis on the evolutionary process of plant hydraulics.
Methods We selected three species in each group grown in South China Botanical Garden, Guangzhou, China, including ferns (Dicranopteris pedata, Cyclosorus parasiticus and Blechnum orientale), gymnosperms (Podocarpus macrophyllus, Podocarpus nagi and Taxodium distichum) and angiosperms (Manglietia fordiana var. hainanensis, Sindora tonkinensis and Bauhinia purpurea).
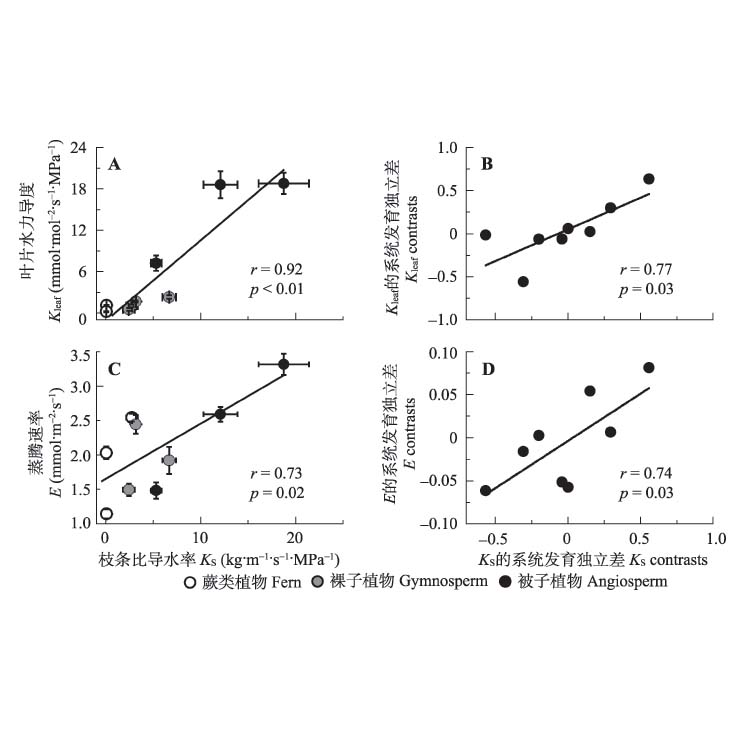
Important findings Sapwood and leaf specific hydraulic conductivities (KS and KL, respectively), and leaf conductance (Kleaf) significantly increased from ferns, gymnosperms to angiosperms. However, no significant trends were found in transpiration rate (E) and intrinsic water use efficiency. Meanwhile, neither the size and density of stomata nor wood density showed significant difference among three plant groups. The hydraulic functional traits (KS, KL and Kleaf) had significantly positive correlations with each other, but had no relationships with the two measured structural traits. Phylogenetic independent contrasts analyses showed that the coordination between KS and Kleaf, and between KS and E were independent of the phylogeny. Based on the nine vascular species, this study demonstrated that water transport related traits are improved as vascular plants evolved, and the co-evolution between water transport and transpiration traits were identified. For further study, it is necessary to consolidate our data with investigations of more detailed water-transport structures in more species from different evolutionary lineages.
ZHAOLe-Wen, CHEN Zi-Yi, ZOU Ying, FU Zi-Zhao, WU Gui-Lin, LIU Xiao-Rong, LUO Qi, LIN Yi-Xue, LI Xiong-Ju, LIU Zhi-Tong, LIU Hui . Changes in hydraulic traits of nine vascular plants from different evolutionary lineages[J]. Chinese Journal of Plant Ecology, 2018 , 42(2) : 220 -228 . DOI: 10.17521/cjpe.2017.0258
| [1] | Azani N, Babineau M, Bailey CD, Banks H, Barbosa AR, Pinto RB, Boatwright JS, Borges LM, Brown GK, Bruneau A, Candido E, Cardoso D, Chung K-F, Clark RP, Concei??o AdS, Crisp M, Cubas P, Delgado-Salinas A, Dexter KG, Doyle JJ, Duminil J, Egan AN, De La Estrella M, Falc?o MJ, Filatov DA, Fortuna-Perez AP, Fortunato RH, Gagnon E, Gasson P, Rando JG, Azevedo Tozzi AMGd, Gunn B, Harris D, Haston E, Hawkins JA, Herendeen PS, Hughes CE, Iganci JRV, Javadi F, Kanu SA, Kazempour- Osaloo S, Kite GC, Klitgaard BB, Kochanovski FJ, Koenen EJM, Kovar L, Lavin M, Roux Ml, Lewis GP, de Lima HC, López Roberts MC, Mackinder B, Maia VH, Malécot V, Mansano VF, Marazzi B, Mattapha S, Miller JT, Mitsuyuki C, Moura T, Murphy DJ, Nageswara-Rao M, Nevado B, Neves D, Ojeda DI, Pennington RT, Prado DE, Prenner G, de Queiroz LP, Ramos G, Ranzato Filardi FL, Ribeiro PG, Rico-Arce MdL, Sanderson MJ, Santos- Silva J, S?o-Mateus WMB, Silva MJS, Simon MF, Sinou C, Snak C, de Souza éR, Sprent J, Steele KP, Steier JE, Steeves R, Stirton CH, Tagane S, Torke BM, Toyama H, Cruz DTd, Vatanparast M, Wieringa JJ, Wink M, Wojciechowski MF, Yahara T, Yi T, Zimmerman E ( 2017). A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny, The Legume Phylogeny Working Group (LPWG). Taxon, 66, 44-77. |
| [2] | Boyce CK, Brodribb TJ, Feild TS, Zwieniecki MA ( 2009). Angiosperm leaf vein evolution was physiologically and environmentally transformative. Proceedings of the Royal Society B, 276, 1771-1776. |
| [3] | Brodribb TJ, Bienaimé D, Marmottant P ( 2016). Revealing catastrophic failure of leaf networks under stress. Proceedings of the National Academy of Sciences of the United States of America, 113, 4865-4869. |
| [4] | Brodribb TJ, Feild TS ( 2010). Leaf hydraulic evolution led a surge in leaf photosynthetic capacity during early angiosperm diversification. Ecology Letters, 13, 175-183. |
| [5] | Brodribb TJ, Feild TS, Jordan GJ ( 2007). Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiology, 144, 1890-1898. |
| [6] | Brodribb TJ, Holbrook NM ( 2003 a). Changes in leaf hydraulic conductance during leaf shedding in seasonally dry tropical forest. New Phytologist, 158, 295-303. |
| [7] | Brodribb TJ, Holbrook NM ( 2003 b). Stomatal closure during leaf dehydration, correlation with other leaf physiological traits. Plant Physiology, 132, 2166-2173. |
| [8] | Brodribb TJ, Holbrook NM, Zwieniecki MA, Palma B ( 2005). Leaf hydraulic capacity in ferns, conifers and angiosperms, impacts on photosynthetic maxima. New Phytologist, 165, 839-846. |
| [9] | Buckley LB, Kingsolver JG ( 2012). Functional and phylogenetic approaches to forecasting species responses to climate change. Annual Review of Ecology, Evolution, and Systematics, 43, 205-264. |
| [10] | Darwin C ( 1859). On the Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life. John Murray, London. |
| [11] | Escalona JM, Flexas J, Medrano H ( 1999). Stomatal and non-stomatal limitations of photosynthesis under water stress in field-grown grapevines. Functional Plant Biology, 26, 421-433. |
| [12] | Fan ZX, Zhang SB, Hao GY, Ferry Slik JW, Cao KF ( 2012). Hydraulic conductivity traits predict growth rates and adult stature of 40 Asian tropical tree species better than wood density. Journal of Ecology, 100, 732-741. |
| [13] | Franks PJ, Beerling DJ ( 2009). Maximum leaf conductance driven by CO2 effects on stomatal size and density over geologic time. Proceedings of the National Academy of Sciences of the United States of America, 106, 10343-10347. |
| [14] | Franks PJ, Drake PL, Beerling DJ ( 2009). Plasticity in maximum stomatal conductance constrained by negative correlation between stomatal size and density, an analysis using Eucalyptus globulus. Plant, Cell & Environment, 32, 1737-1748. |
| [15] | Freckleton RP, Harvey PH, Pagel M ( 2002). Phylogenetic analysis and comparative data, a test and review of evidence. The American Naturalist, 160, 712-726. |
| [16] | Givnish TJ ( 2002). Adaptive significance of evergreen vs. deciduous leaves, solving the triple paradox. Silva Fennica, 36, 703-743. |
| [17] | Guyot G, Scoffoni C, Sack L ( 2012). Combined impacts of irradiance and dehydration on leaf hydraulic conductance, insights into vulnerability and stomatal control. Plant, Cell & Environment, 35, 857-871. |
| [18] | Hacke UG, Sperry JS, Pockman WT, Davis SD, McCulloh KA ( 2001). Trends in wood density and structure are linked to prevention of xylem implosion by negative pressure. Oecologia, 126, 457-461. |
| [19] | Ishida A, Nakano T, Yazaki K, Matsuki S, Koike N, Lauenstein D, Shimizu M, Yamashita N ( 2008). Coordination between leaf and stem traits related to leaf carbon gain and hydraulics across 32 drought-tolerant angiosperms. Oecologia, 156, 193-202. |
| [20] | Liu H, Xu QY, He PC, Santiago LS, Yang KM, Ye Q ( 2015 a). Strong phylogenetic signals and phylogenetic niche conservatism in ecophysiological traits across divergent lineages of Magnoliaceae. Scientific Reports, 5, 12246. DOI: 10.1038/srep12246. |
| [21] | Liu YY, Song J, Wang M, Li N, Niu CY, Hao GY ( 2015 b). Coordination of xylem hydraulics and stomatal regulation in keeping the integrity of xylem water transport in shoots of two compound-leaved tree species. Tree Physiology, 35, 1333-1342. |
| [22] | Nicotra AB, Davidson A ( 2010). Adaptive phenotypic plasticity and plant water use. Functional Plant Biology, 37, 117-127. |
| [23] | Nicotra AB, Leigh A, Boyce CK, Jones CS, Niklas KJ, Royer DL, Tsukaya H ( 2011). The evolution and functional significance of leaf shape in the angiosperms. Functional Plant Biology, 38, 535-552. |
| [24] | Oren R, Sperry JS, Ewers BE, Pataki DE, Philips N, Megonigal JP ( 2001). Sensitivity of mean canopy stomatal conductance to vapor pressure deficit in a flooded Taxodium distichum L. forest: Hydraulic and non-hydraulic effects. Oecologia, 126, 21-29. |
| [25] | Paradis E, Claude J, Strimmer K ( 2004). APE, analyses of phylogenetics and evolution in R language. Bioinformatics, 20, 289-290. |
| [26] | Quinn CJ, Price RA, Gadek PA ( 2002). Familial concepts and relationships in the conifer based on rbcL and matK sequence comparisons. Kew Bulletin, 57, 513-531. |
| [27] | Sack L, Cowan PD, Jaikumar N, Holbrook NM ( 2003). The ‘hydrology’ of leaves, co-ordination of structure and function in temperature woody species. Plant, Cell & Environment, 26, 1343-1356. |
| [28] | Santiago LS, Goldstein G, Meinzer FC, Fisher JB, Machado K, Woodruff D, Jones T ( 2004). Leaf photosynthetic traits scale with hydraulic conductivity and wood density in Panamanian forest canopy trees. Oecologia, 140, 543-550. |
| [29] | Scoffoni C, McKown AD, Rawls M, Sack L ( 2012). Dynamics of leaf hydraulic conductance with water status, quantification and analysis of species differences under steady state. Journal of Experimental Botany, 63, 643-658. |
| [30] | Song J, Li RH, Zhu SD, Ye Q ( 2013). Leaf functional traits of ferns from different habitats in monsoon evergreen broad- leaved forest in Dinghushan Mountain . Journal of Tropical and Subtropical Botany, 21, 489-495. |
| [30] | 宋娟, 李荣华, 朱师丹 鼎湖山季风常绿阔叶林不同生境蕨类植物的叶片功能性状研究. 热带亚热带植物学报, 21, 489-495.] |
| [31] | Sperry JS, Donnelly JR, Tyree MT ( 1988). A method for measuring hydraulic conductivity and embolism in xylem. Plant, Cell & Environment, 11, 35-40. |
| [32] | Testo W, Sundue M ( 2016). A 4000-species dataset provides new insight into the evolution of ferns. Molecular Phylogenetics and Evolution, 105, 200-211. |
| [33] | Trifiló P, Raimondo F, Savi T, Lo Gullo MA, Nardini A ( 2016). The contribution of vascular and extra-vascular water pathways to drought-induced decline of leaf hydraulic conductance. Journal of Experimental Botany, 67, erw268. DOI: 10.1093/jxb/erw268. |
| [34] | Tuzet A, Perrier A, Leuning R ( 2003). A coupled model of stomatal conductance, photosynthesis and transpiration. Plant, Cell & Environment, 26, 1097-1116. |
| [35] | Webb CO, Ackerly DD, Kembel SW ( 2008). Phylocom, software for the analysis of phylogenetic community structure trait evolution. Bioinformatics, 24, 2098-2100. |
| [36] | Wu DL ( 2006). loral of Guangdong . Guangdong Science & Technology Press, Guangzhou. |
| [36] | [ 吴德邻 ( 2006). 广东植物志 .广东科技出版社, 广州.] |
| [37] | Zhan XY, Yu GR, Sheng WP, Fang HJ ( 2012). Foliar water use efficiency and nitrogen use efficiency of dominant plant species in main forests along the North-South Transect of East China. Chinese Journal of Applied Ecology, 23, 587-594. |
| [37] | [ 展小云, 于贵瑞, 盛文萍, 方华军 ( 2012). 中国东部南北样带森林优势植物叶片的水分利用效率和氮素利用效率. 应用生态学报 , 23, 587-594.] |
| [38] | Zhang Y, Yang SJ, Sun M, Cao KF ( 2014). Stomatal traits are evolutionarily associated with vein density in basal angiosperms. Plant Science Journal, 32, 320-328. |
| [38] | [ 张亚, 杨石建, 孙梅, 曹坤芳 ( 2014). 基部被子植物气孔性状与叶脉密度的关联演化.植物科学学报 , 32, 320-328.] |
/
| 〈 |
|
〉 |