

Chin J Plant Ecol ›› 2018, Vol. 42 ›› Issue (11): 1113-1119.DOI: 10.17521/cjpe.2018.0145
• Research Articles • Previous Articles Next Articles
AN Rui1,MENG Feng1,YIN Peng-Xian2,DU Guang-Yuan1,*( )
)
Received:2018-06-19
Accepted:2018-10-19
Online:2018-11-20
Published:2019-03-13
Contact:
Guang-Yuan DU
Supported by:AN Rui, MENG Feng, YIN Peng-Xian, DU Guang-Yuan. Comparison of methods for detecting vulnerability of xylem embolism in Robinia pseudoacacia[J]. Chin J Plant Ecol, 2018, 42(11): 1113-1119.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.17521/cjpe.2018.0145
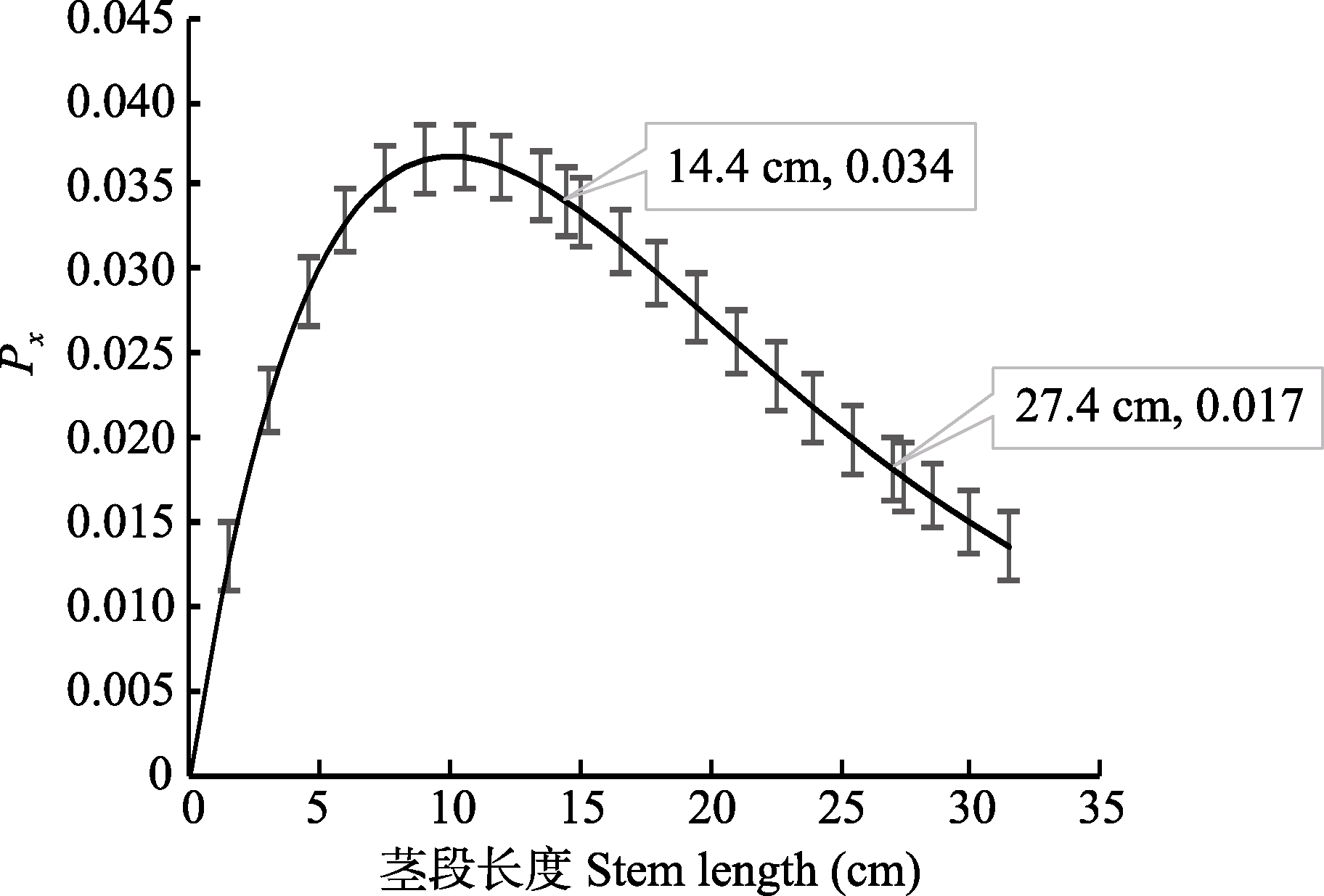
Fig. 1 The probability that the xylem conduit is within the length x interval (Px) and stem length (x) derived from the xylem conduit length distribution of Robinia pseudoacacia (mean ± SD).
| 栓塞脆弱性曲线的建立方法 Methods in establishing embolism vulnerability curves | P50 (MPa) | 样品数 Number of samples | 茎段长度 Stem length (cm) | 来源 Source |
|---|---|---|---|---|
| Sperry离心机法 Sperry centrifugation method | -0.93 ± 0.021a | 6 | 27.4 | |
| -0.92 ± 0.058a | 6 | 14.4 | ||
| 自然干燥法 Bench top dehydration method | -2.91b | 31 | ||
| Cochard Cavitron 离心机法 Cochard Cavitron centrifugation method | -0.38 ± 0.044c | 6 | 27.4 | |
| -0.46 ± 0.030d | 6 | 27.4 | al., 2017 | |
| -0.22 ± 0.026e | 6 | 27.4 |
Table 1 Tension at 50% hydraulic conductivity loss (P50) in branches of Robinia pseudoacacia calculated based on different methods
| 栓塞脆弱性曲线的建立方法 Methods in establishing embolism vulnerability curves | P50 (MPa) | 样品数 Number of samples | 茎段长度 Stem length (cm) | 来源 Source |
|---|---|---|---|---|
| Sperry离心机法 Sperry centrifugation method | -0.93 ± 0.021a | 6 | 27.4 | |
| -0.92 ± 0.058a | 6 | 14.4 | ||
| 自然干燥法 Bench top dehydration method | -2.91b | 31 | ||
| Cochard Cavitron 离心机法 Cochard Cavitron centrifugation method | -0.38 ± 0.044c | 6 | 27.4 | |
| -0.46 ± 0.030d | 6 | 27.4 | al., 2017 | |
| -0.22 ± 0.026e | 6 | 27.4 |
| [1] |
Adams HD, Zeppel MJB, Anderegg WRL, Hartmann H, Landhäusser SM, Tissue DT, Huxman TE, Hudson PJ, Franz TE, Allen CD, Anderegg LDL, Barron-Gafford GA, Beerling DJ, Breshears DD, Brodribb TJ, Bugmann H, Cobb RC, Collins AD, Dickman LT, Duan H, Ewers BE, Galiano L, Galvez DA, Garcia-Forner N, Gaylord ML, Germino MJ, Gessler A, Hacke UG, Hakamada R, Hector A, Jenkins MW, Kane JM, Kolb TE, Law DJ, Lewis JD, Limousin JM, Love DM, Macalady AK, Martínez-Vilalta J, Mencuccini M, Mitchell PJ, Muss JD, O’Brien MJ, O’Grady AP, Pangle RE, Pinkard EA, Piper FI, Plaut JA, Pockman WT, Quirk J, Reinhardt K, Ripullone F, Ryan MG, Sala A, Sevanto S, Sperry JS, Vargas R, Vennetier M, Way DA, Xu C, Yepez EA, McDowell NG ( 2017). A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nature Ecology & Evolution, 1, 1285-1291.
DOI URL PMID |
| [2] | Allen CD, Macalady AK, Chenchouni H, Bachelet D, Dowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg HH, Gonzalez P, Fensham R, Zhen Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Cobb N ( 2010). A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. Forest Ecology & Management, 259, 660-684. |
| [3] |
Choat B, Drayton WM, Brodersen C, Matthews MA, Shackel KA, Wada H, McElrone AJ ( 2010). Measurement of vulnerability to water stress-induced cavitation in grapevine: A comparison of four techniques applied to long-vesseled species. Plant, Cell & Environment, 33, 1502-1512.
DOI URL PMID |
| [4] |
Choat B, Lahr E, Melcher PJ, Zwieniecki MA, Holbrook NM ( 2005). The spatial pattern of air seeding thresholds in mature sugar maple trees. Plant, Cell & Environment, 28, 1082-1089.
DOI URL |
| [5] |
Cochard H ( 2002). A technique for measuring xylem hydraulic conductance under high negative pressures. Plant, Cell & Environment, 25, 815-819.
DOI URL |
| [6] |
Cochard H, Badel E, Herbette S, Delzon S, Choat B, Jansen S ( 2013). Methods for measuring plant vulnerability to cavitation: A critical review. Experimental Botany, 64, 4779-4791.
DOI URL PMID |
| [7] |
Cochard H, Damour G, Bodet C, Tharwat I, Poirier M, Améglio T ( 2005). Evaluation of a new centrifuge technique for rapid generation of xylem vulnerability curves. Physiologia Plantarum, 124, 410-418.
DOI URL |
| [8] |
Cochard H, Herbette S, Barigah T, Badel E, Ennajeh M, Vilagrosa A ( 2010). Does sample length influence the shape of xylem embolism vulnerability curves? A test with the Cavitron spinning technique. Plant, Cell & Environment, 33, 1543-1552.
DOI URL PMID |
| [9] |
Cohen S, Benink J, Tyree M ( 2003). Air method measurements of apple vessel length distributions with improved apparatus and theory. Journal of Experimental Botany, 54, 1889-1897.
DOI URL PMID |
| [10] | Dai YX, Wang L, Wan XC ( 2015). Progress on researches of drought-induced tree mortality mechanisms. Chinese Journal of Ecology, 34, 3228-3236. |
| [ 代永欣, 王林, 万贤崇 ( 2015). 干旱导致树木死亡机制研究进展. 生态学杂志, 34, 3228-3236.] | |
| [11] |
Dang W, Jiang ZM, Li R, Zhang SX, Cai J ( 2017). Relationship between hydraulic traits and refilling of embolism in the xylem of one-year-old twigs of six tree species. Scientia Silvae Sinicae, 53(3), 49-59.
DOI URL |
|
[ 党维, 姜在民, 李荣, 张硕新, 蔡靖 ( 2017). 6个树种1年生枝木质部的水力特征及与栓塞修复能力的关系. 林业科学, 53(3), 49-59.]
DOI URL |
|
| [12] |
Dixon HH, Joly J ( 1895). On the ascent of sap. Philosophical Transactions of the Royal Society of London B, 186, 563-576.
DOI URL |
| [13] |
Domec JC, Gartner BL ( 2001). Cavitation and water storage capacity in bole xylem segments of mature and young Douglas-fir trees. Trees, 15, 204-214.
DOI URL |
| [14] |
Dong L, Li JY ( 2013). Relationship among drought, hydraulic metabolic, carbon starvation and vegetation mortality. Acta Ecologica Sinica, 33, 5477-5483.
DOI URL |
|
[ 董蕾, 李吉跃 ( 2013). 植物干旱胁迫下水分代谢、碳饥饿与死亡机理. 生态学报, 33, 5477-5483.]
DOI URL |
|
| [15] |
Hacke UG, Venturas MD, MacKinnon ED, Jacobsen AL, Sperry JS, Pratt RB ( 2015). The standard centrifuge method accurately measures vulnerability curves of long- vesselled olive stems. New Phytologist, 205, 116-127.
DOI URL PMID |
| [16] |
Jacobsen AL, Pratt RB ( 2012). No evidence for an open vessel effect in centrifuge-based vulnerability curves of a long-vesselled liana (Vitisvinifera). New Phytologist, 194, 982-990.
DOI URL PMID |
| [17] |
Jacobsen AL, Pratt RB, Davis SD, Tobin MF ( 2014). Geographic and seasonal variation in chaparral vulnerability to cavitation. Madrono, 61, 317-327.
DOI URL |
| [18] |
Li R, Dang W, Cai J, Zhang SX, Jiang ZM ( 2016). Relationships between xylem structure and embolism vulnerability in six drought tolerance trees. Chinese Journal of Plant Ecology, 40, 255-263.
DOI URL |
|
[ 李荣, 党维, 蔡靖, 张硕新, 姜在民 ( 2016). 6个耐旱树种木质部结构与栓塞脆弱性的关系. 植物生态学报, 40, 255-263.]
DOI URL |
|
| [19] |
Li R, Jiang ZM, Zhang SX, Cai J ( 2015). A review of new esearch progress on the vulnerability of xylem embolism of woody plants. Chinese Journal of Plant Ecology, 39, 838-848.
DOI URL |
|
[ 李荣, 姜在民, 张硕新, 蔡靖 ( 2015), 本木植物木质部栓塞脆弱性研究新进展. 植物生态学报, 39, 838-848.]
DOI URL |
|
| [20] |
Maherali H, Pockman WT, Jackson RB ( 2004). Adaptive variation in the vulnerability of woody plants to xylem cavitation. Ecology, 85, 2184-2199.
DOI URL |
| [21] |
Melcher PJ, Zwieniecki MA, Holbrook NM ( 2003). Vulnerability of xylem vessels to cavitation in sugar maple. Scaling from individual vessels to whole branches. Plant Physiology, 131, 1775-1780.
DOI URL |
| [22] |
Rockwell FE, Wheeler JK, Holbrook NM ( 2014). Cavitation and its discontents: Opportunities for resolving current controversies. Plant Physiology, 164, 1649-1660.
DOI URL PMID |
| [23] | Sperry JS, Christman MA, Torrez-Ruiz JM, Taneda H, Smith DD ( 2012). Vulnerability curves by centrifugation: Is there an open vessel artifact, and are “r” shaped curves necessarily invalid? Plant, Cell & Environment, 35, 601-610. |
| [24] |
Sperry JS, Donnelly JR, Tyree MT ( 1988). A method for measuring hydraulic conductivity and embolism in xylem. Plant, Cell & Environment, 11, 35-40.
DOI URL |
| [25] |
Sperry JS, Tyree MT ( 1988). Mechanism of water stress-induced xylem embolism. Plant Physiology, 88, 581-587.
DOI URL PMID |
| [26] | Tyree MT, Alexander J, Machado JL ( 1992). Loss of hydraulic conductivity due to water stress in intact juveniles of Quercus rubra and Populus deltoides. Tree Physiology, 10, 411-415. |
| [27] |
Torres-Ruiz JM, Jansen S, Choat B, McElrone AJ, Cochard H, Brodribb TJ, Badel E, Burlett R, Bouche PS, Brodersen CR, Li S, Morris H, Delzon S ( 2015). Direct micro-CT observation confirms the induction of embolism upon xylem cutting under tension. Plant Physiology, 167, 40-43.
DOI URL PMID |
| [28] |
Trifilò P, Nardini A, Gullo MAL, Barbera PM, Tadeja S ( 2015). Diurnal changes in embolism rate in nine dry forest trees: Relationships with species-specific xylem vulnerability, hydraulic strategy and wood traits. Tree Physiology, 57, 192-197.
DOI URL PMID |
| [29] |
Van den Honert TH ( 1948). Water transport in plants as a catenary process. Discussions of the Faraday Society, 3, 146-153.
DOI URL |
| [30] |
Venturas MD, Sperry JS, Hacke UG ( 2017). Plant xylem hydraulics: What we understand, current research, and future challenges. Journal of Integrative Plant Biology, 59, 356-389.
DOI URL |
| [31] | Wang RQ, Zhang LL, Zhang SX, Cai J, Tyree MT ( 2014). Water relations of Robinia pseudoacacia L.: Do vessels cavitate and refill diurnally or are R-shaped curves invalid in Robinia? Plant, Cell & Environment, 37, 2667-2678. |
| [32] | Zimmermann MH ( 1983). Xylem Structure and the Ascent of Sap. Springer, Berlin. |
| [1] | WANG Jia-Yi, WANG Xiang-Ping, XU Cheng-Yang, XIA Xin-Li, XIE Zong-Qiang, FENG Fei, FAN Da-Yong. Response of hydraulic architecture in Fraxinus velutina street trees to the percentage of impervious pavement in Beijing [J]. Chin J Plant Ecol, 2023, 47(7): 998-1009. |
| [2] | LU Shi-Tong, CHEN Sen, LI Yan, WANG Zhong-Yuan, PAN Tian-Tian, YE Lin-Feng, XIE Jiang-Bo. Relationships among xylem transport, anatomical structure and mechanical strength in stems and roots of three Podocarpaceae species [J]. Chin J Plant Ecol, 2021, 45(6): 659-669. |
| [3] | CHENG Xiang-Fen, MA Jin, ZHAO Han , JIANG Zai-Min, CAI Jing . Vessel length as a key hydraulic structure in woody plants: A review [J]. Chin J Plan Ecolo, 2018, 42(6): 609-618. |
| [4] | Rong LI, Wei DANG, Jing CAI, Shuo-Xin ZHANG, Zai-Min JIANG. Relationships between xylem structure and embolism vulnerability in six species of drought tolerance trees [J]. Chin J Plant Ecol, 2016, 40(3): 255-263. |
| [5] | LI Rong,JIANG Zai-Min,ZHANG Shuo-Xin,CAI Jing. A review of new research progress on the vulnerability of xylem embolism of woody plants [J]. Chin J Plan Ecolo, 2015, 39(8): 838-848. |
| [6] | XU Xin-Wu, FAN Da-Yong, XIE Zong-Qiang, ZHANG Shou-Ren, ZHANG Xiang-Ying. EFFECTS OF DIFFERENT FLUSH SOLUTIONS ON VALUES OF HYDRAULIC CONDUCTIVITIES AND CAVITATION RESISTANCE ABILITIES OF TRESS OF POPULUS TOMENTOSA AND PINUS TABULAEFORMIS [J]. Chin J Plant Ecol, 2009, 33(1): 150-160. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2026 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
![]()