

Chin J Plant Ecol ›› 2017, Vol. 41 ›› Issue (6): 683-692.DOI: 10.17521/cjpe.2016.0136
• Research Articles • Previous Articles Next Articles
Rui GUO1,2,*( ), Ji ZHOU3, Fan YANG4, Feng LI1
), Ji ZHOU3, Fan YANG4, Feng LI1
Received:2017-04-05
Accepted:2016-04-14
Online:2017-06-10
Published:2017-07-19
Contact:
Rui GUO
About author:KANG Jing-yao(1991-), E-mail: Rui GUO, Ji ZHOU, Fan YANG, Feng LI. Metabolic responses of wheat roots to alkaline stress[J]. Chin J Plant Ecol, 2017, 41(6): 683-692.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.17521/cjpe.2016.0136
| 处理 Treatment | 碱浓度 Alkaline concentration (mmol·L-1) | 电导率 EC (dS·m-1) | 渗透势 Osmotic potential (MPa) | pH值 pH value |
|---|---|---|---|---|
| 对照 Control | 0 | 2.16 | -0.05 | 6.95 |
| 碱胁迫 Alkaline stress | 50 | 4.98 | -0.28 | 9.69 |
| 100 | 9.21 | -0.51 | 9.92 |
Table 1 The electrical conductivity (EC), pH value and osmotic potential of salinity stress treatment solutions
| 处理 Treatment | 碱浓度 Alkaline concentration (mmol·L-1) | 电导率 EC (dS·m-1) | 渗透势 Osmotic potential (MPa) | pH值 pH value |
|---|---|---|---|---|
| 对照 Control | 0 | 2.16 | -0.05 | 6.95 |
| 碱胁迫 Alkaline stress | 50 | 4.98 | -0.28 | 9.69 |
| 100 | 9.21 | -0.51 | 9.92 |
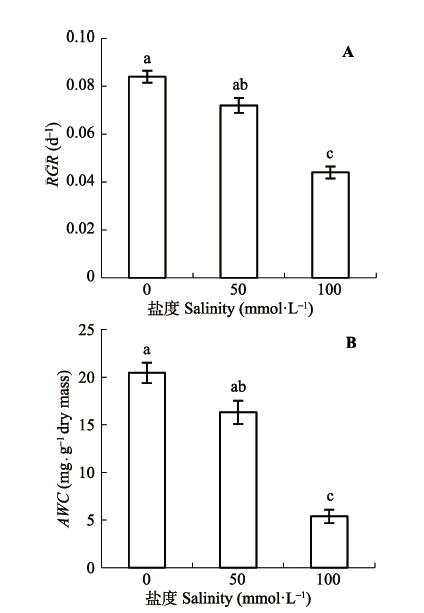
Fig. 1 Effects of alkaline concentration on root relative growth rate (RGR) and absolute water content (AWC) (mean ± SE, n = 5). Different lowercase letters indicate significant differences among the various treatments (p < 0.05).
| 处理 Treatment | 碱胁迫浓度 Alkaline concentration (mmol) | 矿质元素 Metal elements (mmol·g-1 dry mass) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Na | K | Ca | Mg | Cu | Fe | Zn | Mn | ||
| 对照 Control | 0 | 4.25 ± 1.01b | 69.90 ± 3.03a | 17.11 ± 1.01b | 7.80 ± 0.29a | 1.25 ± 0.04a | 1.56 ± 0.08a | 0.07 ± 0.01a | 0.06 ± 0.00a |
| 碱胁迫 Alkaline stress | 50 | 15.89 ± 2.06 ab | 45.20 ± 1.54b | 20.53 ± 1.00b | 6.01 ± 0.16b | 1.02 ± 0.02a | 1.26 ± 0.03a | 0.05 ± 0.00b | 0.05 ± 0.00a |
| 100 | 19.09 ± 2.11a | 16.83 ± 1.02c | 32.85 ± 1.30a | 4.96 ± 0.47c | 0.55 ± 0.03b | 0.84 ± 0.05b | 0.03 ± 0.00c | 0.06 ± 0.00a | |
Table 2 Effects of alkaline stress on the contents of metal elements in the roots of wheat seedlings (mean ± SE, n = 5)
| 处理 Treatment | 碱胁迫浓度 Alkaline concentration (mmol) | 矿质元素 Metal elements (mmol·g-1 dry mass) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Na | K | Ca | Mg | Cu | Fe | Zn | Mn | ||
| 对照 Control | 0 | 4.25 ± 1.01b | 69.90 ± 3.03a | 17.11 ± 1.01b | 7.80 ± 0.29a | 1.25 ± 0.04a | 1.56 ± 0.08a | 0.07 ± 0.01a | 0.06 ± 0.00a |
| 碱胁迫 Alkaline stress | 50 | 15.89 ± 2.06 ab | 45.20 ± 1.54b | 20.53 ± 1.00b | 6.01 ± 0.16b | 1.02 ± 0.02a | 1.26 ± 0.03a | 0.05 ± 0.00b | 0.05 ± 0.00a |
| 100 | 19.09 ± 2.11a | 16.83 ± 1.02c | 32.85 ± 1.30a | 4.96 ± 0.47c | 0.55 ± 0.03b | 0.84 ± 0.05b | 0.03 ± 0.00c | 0.06 ± 0.00a | |
| 处理 Treatment | 碱胁迫浓度 Alkaline concentration (mmol) | 阴离子 Anions (mmol·g-1 dry mass) | |||
|---|---|---|---|---|---|
| Cl- | NO3- | H2PO4- | SO42- | ||
| 对照 Control | 0 | 0.12 ± 0.01a | 0.41 ± 0.02a | 0.05 ± 0.00a | 0.03 ± 0.00a |
| 碱胁迫 Alkaline stress | 50 | 0.09 ± 0.00b | 0.34 ± 0.01b | 0.03 ± 0.01b | 0.02 ± 0.00a |
| 100 | 0.04 ± 0.01c | 0.07 ± 0.00c | 0.02 ± 0.00b | 0.03 ± 0.00a | |
Table 3 Effects of alkaline stress on the contents of Cl-, NO3-, H2PO4-, SO42- in the roots of wheat seedlings (mean ± SE, n = 5).
| 处理 Treatment | 碱胁迫浓度 Alkaline concentration (mmol) | 阴离子 Anions (mmol·g-1 dry mass) | |||
|---|---|---|---|---|---|
| Cl- | NO3- | H2PO4- | SO42- | ||
| 对照 Control | 0 | 0.12 ± 0.01a | 0.41 ± 0.02a | 0.05 ± 0.00a | 0.03 ± 0.00a |
| 碱胁迫 Alkaline stress | 50 | 0.09 ± 0.00b | 0.34 ± 0.01b | 0.03 ± 0.01b | 0.02 ± 0.00a |
| 100 | 0.04 ± 0.01c | 0.07 ± 0.00c | 0.02 ± 0.00b | 0.03 ± 0.00a | |
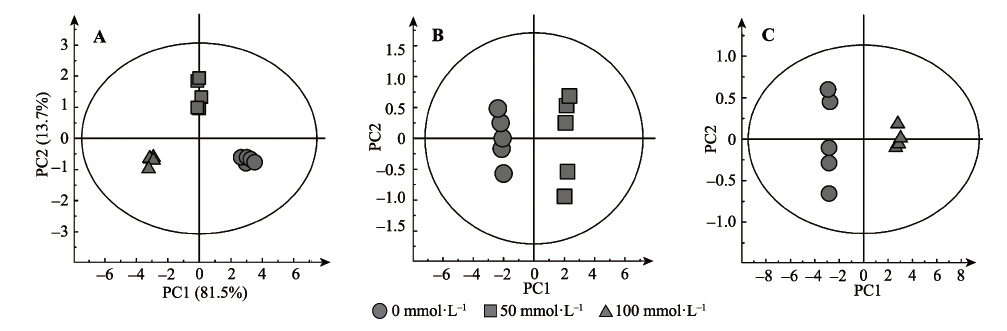
Fig. 2 SIMCA analyzed score plots showing the metabolomic trajectory of roots of wheat seedlings under different salinity concentration treatments. Principal component analysis (PCA) score plots (A). Orthogonal partial least squares discriminant analysis (OPLS-DA) scores: CK vs. AS-50 mmol·L-1 (B) and CK vs. AS-100 mmol·L-1 (C).
| 代谢路径和代谢物名称 Metabolic pathways and metabolites | 相对含量 Relative concentration | 倍性变量Fold changes | ||||
|---|---|---|---|---|---|---|
| CK | AS-50 mmol | AS-100 mmol | log2(50/CK) | log2(100/CK) | ||
| 三羧酸循环 Tricarboxylic acid cycle | 柠檬酸 Citric acid | 75.73 | 150.14 | 231.23 | 0.99* | 1.61** |
| 乌头酸 Aconitic acid | 1.34 | 1.70 | 5.13 | 0.34 | 1.93** | |
| α-酮戊二酸 α-ketoglutaric acid | 0.18 | 0.33 | 0.85 | 0.89 | 2.23** | |
| 琥珀酸 Succinic acid | 19.05 | 61.45 | 120.69 | 1.69** | 2.66** | |
| 延胡索酸 Fumaric acid | 1.34 | 1.56 | 9.63 | 0.23 | 2.85** | |
| 苹果酸 Malic acid | 10.47 | 23.10 | 30.35 | 1.14* | 1.54** | |
| 糖酵解过程 Glycolysis | 葡萄糖 Glucose | 26.37 | 13.67 | 7.91 | -0.95* | -1.74** |
| 葡萄糖-6-磷酸 Fructose-6-phosphate | 0.57 | 0.25 | 0.08 | -1.16* | -2.75** | |
| 果糖-6-磷酸 Glucose-6-phosphate | 0.22 | 0.13 | 0.04 | -0.80 | -2.58** | |
| 3-磷酸甘油酸 3-phosphoglyceric acid | 0.50 | 0.34 | 0.11 | -0.57 | -2.19** | |
| 丙酮酸 Pyruvate | 0.54 | 0.50 | 0.31 | -0.10 | -0.81 | |
| 磷酸烯醇式丙酮酸 Enolphosphopyruvate | 0.73 | 0.61 | 0.22 | -0.26 | -1.74** | |
| 莽草酸途径 Shikimic path way | 莽草酸 Shikimic acid | 1.84 | 1.22 | 3.84 | -0.60 | 1.06* |
| 奎尼酸 Quinic acid | 4.17 | 6.17 | 23.11 | 0.58 | 2.47** | |
| 苯丙氨酸 Phenylalanine | 0.65 | 0.17 | 0.11 | -1.91* | -2.60** | |
| 色氨酸 Tryptophan | 0.05 | 0.03 | 0.02 | -0.71 | -1.45* | |
| 酪氨酸 Tyrosine | 1.59 | 0.03 | 0.02 | -4.18** | -6.57** | |
| 肉桂酸 Cinnamic acid | 0.25 | 0.15 | 0.12 | -0.77 | -1.12* | |
| 细胞膜脂代谢 Metabolism of plasma membrane | 肌醇 Myo-inositol | 19.17 | 9.63 | 3.07 | -0.99* | -2.64** |
| 甘氨酸 Glycine | 0.74 | 0.34 | 0.12 | -1.56** | -2.69** | |
| 丝氨酸 Serine | 17.83 | 9.56 | 1.64 | -0.90* | -3.44** | |
| 乙醇胺 Ethanolamine | 20.23 | 11.31 | 3.26 | -0.84* | -2.63** | |
| 氨基酸 Amino acid | γ-氨基丁酸 γ-aminobutyric acid | 137.19 | 41.77 | 23.92 | -1.72** | -2.52** |
| 丙氨酸 Alanine | 106.58 | 56.88 | 10.64 | -0.91* | -3.32** | |
| 谷氨酸 Glutamate | 24.93 | 13.73 | 8.40 | -0.86* | -1.57** | |
| 天冬酰胺 Asparagine | 12.65 | 4.38 | 0.91 | -1.53** | -3.79** | |
| 天冬氨酸 Aspartic acid | 7.60 | 4.06 | 3.34 | -0.90* | -1.19* | |
| 脯氨酸 Proline | 10.04 | 15.47 | 11.57 | 0.62 | 0.20 | |
| 赖氨酸 Lysine | 17.32 | 0.33 | 0.11 | -5.72** | -7.26** | |
| 糖类及多元醇 Sugars and polyols | 果糖 Fructose | 814.31 | 574.62 | 173.68 | -0.78* | -2.23** |
| 蔗糖 Sucrose | 136.11 | 92.04 | 65.17 | -0.56 | -1.06* | |
| 塔罗糖 Talose | 114.17 | 75.07 | 23.13 | -0.60 | -2.30** | |
| 蔗果三糖 Kestose | 57.63 | 42.47 | 19.69 | -0.44 | -1.55** | |
| 核糖 Ribose | 8.24 | 4.25 | 1.38 | -0.96* | -2.58** | |
Table 4 Relative concentration and changes of major metabolites in roots of wheat seedlings after alkaline stress treatment
| 代谢路径和代谢物名称 Metabolic pathways and metabolites | 相对含量 Relative concentration | 倍性变量Fold changes | ||||
|---|---|---|---|---|---|---|
| CK | AS-50 mmol | AS-100 mmol | log2(50/CK) | log2(100/CK) | ||
| 三羧酸循环 Tricarboxylic acid cycle | 柠檬酸 Citric acid | 75.73 | 150.14 | 231.23 | 0.99* | 1.61** |
| 乌头酸 Aconitic acid | 1.34 | 1.70 | 5.13 | 0.34 | 1.93** | |
| α-酮戊二酸 α-ketoglutaric acid | 0.18 | 0.33 | 0.85 | 0.89 | 2.23** | |
| 琥珀酸 Succinic acid | 19.05 | 61.45 | 120.69 | 1.69** | 2.66** | |
| 延胡索酸 Fumaric acid | 1.34 | 1.56 | 9.63 | 0.23 | 2.85** | |
| 苹果酸 Malic acid | 10.47 | 23.10 | 30.35 | 1.14* | 1.54** | |
| 糖酵解过程 Glycolysis | 葡萄糖 Glucose | 26.37 | 13.67 | 7.91 | -0.95* | -1.74** |
| 葡萄糖-6-磷酸 Fructose-6-phosphate | 0.57 | 0.25 | 0.08 | -1.16* | -2.75** | |
| 果糖-6-磷酸 Glucose-6-phosphate | 0.22 | 0.13 | 0.04 | -0.80 | -2.58** | |
| 3-磷酸甘油酸 3-phosphoglyceric acid | 0.50 | 0.34 | 0.11 | -0.57 | -2.19** | |
| 丙酮酸 Pyruvate | 0.54 | 0.50 | 0.31 | -0.10 | -0.81 | |
| 磷酸烯醇式丙酮酸 Enolphosphopyruvate | 0.73 | 0.61 | 0.22 | -0.26 | -1.74** | |
| 莽草酸途径 Shikimic path way | 莽草酸 Shikimic acid | 1.84 | 1.22 | 3.84 | -0.60 | 1.06* |
| 奎尼酸 Quinic acid | 4.17 | 6.17 | 23.11 | 0.58 | 2.47** | |
| 苯丙氨酸 Phenylalanine | 0.65 | 0.17 | 0.11 | -1.91* | -2.60** | |
| 色氨酸 Tryptophan | 0.05 | 0.03 | 0.02 | -0.71 | -1.45* | |
| 酪氨酸 Tyrosine | 1.59 | 0.03 | 0.02 | -4.18** | -6.57** | |
| 肉桂酸 Cinnamic acid | 0.25 | 0.15 | 0.12 | -0.77 | -1.12* | |
| 细胞膜脂代谢 Metabolism of plasma membrane | 肌醇 Myo-inositol | 19.17 | 9.63 | 3.07 | -0.99* | -2.64** |
| 甘氨酸 Glycine | 0.74 | 0.34 | 0.12 | -1.56** | -2.69** | |
| 丝氨酸 Serine | 17.83 | 9.56 | 1.64 | -0.90* | -3.44** | |
| 乙醇胺 Ethanolamine | 20.23 | 11.31 | 3.26 | -0.84* | -2.63** | |
| 氨基酸 Amino acid | γ-氨基丁酸 γ-aminobutyric acid | 137.19 | 41.77 | 23.92 | -1.72** | -2.52** |
| 丙氨酸 Alanine | 106.58 | 56.88 | 10.64 | -0.91* | -3.32** | |
| 谷氨酸 Glutamate | 24.93 | 13.73 | 8.40 | -0.86* | -1.57** | |
| 天冬酰胺 Asparagine | 12.65 | 4.38 | 0.91 | -1.53** | -3.79** | |
| 天冬氨酸 Aspartic acid | 7.60 | 4.06 | 3.34 | -0.90* | -1.19* | |
| 脯氨酸 Proline | 10.04 | 15.47 | 11.57 | 0.62 | 0.20 | |
| 赖氨酸 Lysine | 17.32 | 0.33 | 0.11 | -5.72** | -7.26** | |
| 糖类及多元醇 Sugars and polyols | 果糖 Fructose | 814.31 | 574.62 | 173.68 | -0.78* | -2.23** |
| 蔗糖 Sucrose | 136.11 | 92.04 | 65.17 | -0.56 | -1.06* | |
| 塔罗糖 Talose | 114.17 | 75.07 | 23.13 | -0.60 | -2.30** | |
| 蔗果三糖 Kestose | 57.63 | 42.47 | 19.69 | -0.44 | -1.55** | |
| 核糖 Ribose | 8.24 | 4.25 | 1.38 | -0.96* | -2.58** | |
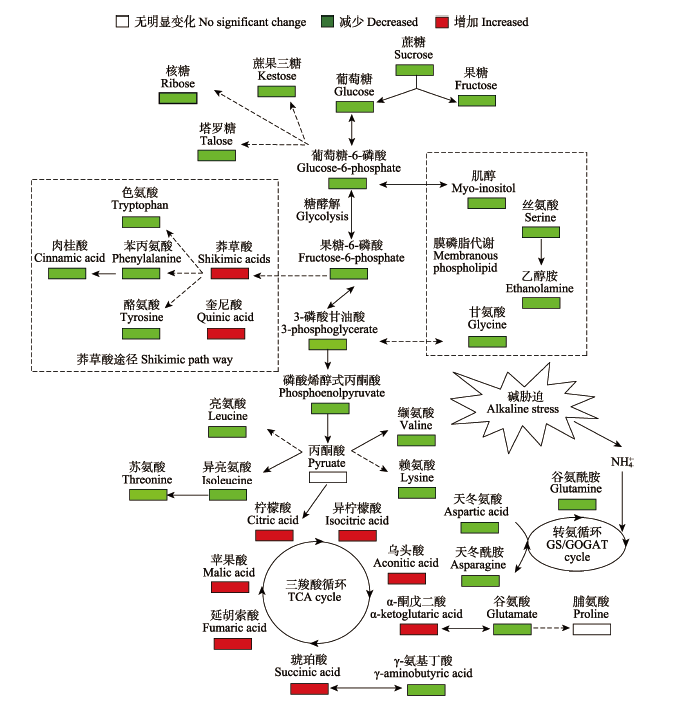
Fig. 3 Proposed metabolic network changes for wheat roots upon alkaline stress obtained from OPLS-DA analysis. The metabolite with white boxes denotes no significant change while red boxes denotes significant increases and green ones denotes significant decreases (p < 0.05).
| [1] | Deng RL, Xu HR, Cao YF, Xiao K (2007). The molecular basis of ammonium transporters in plants.Plant Nutrition and Fertilizer Science, 13, 512-519. (in Chinese with English abstract)[邓若磊, 徐海荣, 曹云飞, 肖凯 (2007). 植物吸收铵态氮的分子生物学基础. 植物营养与肥料学报, 13, 512-519.] |
| [2] | FAO ( |
| [3] | Guo R, Li F, Zhou J, Li HR, Xia X, Liu Q (2016). Eco-physiological responses of linseed (Linum usitatissimum) to salt and alkali stresses. Chinese Journal of Plant Ecology, 40, 69-79. (in Chinese with English abstract)[郭瑞, 李峰, 周际, 李昊儒, 夏旭, 刘琪 (2016). 亚麻响应盐、碱胁迫的生理特征. 植物生态学报, 40, 69-79.] |
| [4] | Guo W, Yu LH (2012). Effects of salinity-alkalinity stress on root activity and phenylalanine ammonia-lyase activity of wheat seedlings.Crops, (1), 31-34. (in Chinese with English abstract)[郭伟, 于立河 (2012). 盐碱胁迫对小麦幼苗根系活力和苯丙氨酸解氨酶活性的影响. 作物杂志, (1), 31-34.] |
| [5] | Hu HY, He J, Zhao JJ, Ru ZG (2013). Dynamic change of cell defense enzyme activity in wheat seedling under different alkaline pH conditions.Journal of Henan Institute of Science and Technology (Natural Science Edition), 41(5), 1-5. (in Chinese with English abstract)[胡海燕, 贺杰, 赵俊杰, 茹振刚 (2013). 碱性pH条件下小麦幼苗保护酶活性的变化动态. 河南科技学院学报(自然科学版), 41(5), 1-5.] |
| [6] | Jianaer AH, Yang CW, Shi DC, Wang DL (2007). Physiological response of an alkali resistant halophyte Kochia sieversiana to salt and alkali stresses. Acta Botanica Boreali-Occidentalia Sinica, 27, 79-84. (in Chinese with English abstract)[贾娜尔·阿汗, 杨春武, 石德成, 王德利 (2007). 盐生植物碱地肤对盐碱胁迫的生理响应特点. 西北植物学报, 27, 79-84. ] |
| [7] | Jiang SX, Liu DX, Pang HX, Lü JY (2014). Effects of PEG stress and recovery on activities of key enzymes involved in proline metabolism in wheat cultivars with difference in drought tolerance.Acta Botanica Boreali-Occidentalia Sinica, 34, 1581-1587. (in Chinese with English abstract)[姜淑欣, 刘党校, 庞红喜, 吕金印 (2014). PEG胁迫及复水对不同抗旱性小麦幼苗脯氨酸代谢关键酶活性的影响. 西北植物学报, 34, 1581-1587.] |
| [8] | Kerepesi I, Galiba G (2000). Osmotic and salt stress induced alteration in soluble carbohydrate content in wheat seedlings.Crop Science, 40, 482-487. |
| [9] | Kingsbury RW, Epstein E, Peary RW (1984). Physiological responses to salinity in selected lines of wheat. Plant Physiology, 74, 417-423. |
| [10] | Li B, Wang ZC, Sun ZG, Chen Y, Yang F (2005). Resources and sustainable resource exploitation of salinized land in China.Agricultural Research in the Arid Areas, 23(2), 152-158. (in Chinese with English abstract)[李彬, 王志春, 孙志高, 陈渊, 杨福 (2005). 中国盐碱地资源与可持续利用研究. 干旱地区农业研究, 23(2), 152-158.] |
| [11] | Lisec J, Schauer N, Kopka J, Willmitzer L, Fernie AR (2006). Gas chromatography mass spectrometry-based metabolite profiling in plants.Nature Protocol, 1, 387-396. |
| [12] | Luo YY, Liu SK (2009). Research progress of ammonium transporter in plants.Genomics and Applied Biology, 28, 373-379. (in Chinese with English abstract)[骆媛嫒, 柳参奎 (2009). 植物中铵转运蛋白的研究进展. 基因组学与应用生物学, 28, 373-379.] |
| [13] | Munns R, Tester M (2008). Mechanisms of salinity tolerance.Annual Review of Plant Biology, 59, 651-681. |
| [14] | Parida AK, Das AB (2005). Salt tolerance and salinity effects on plants: A review.Ecotoxicology and Environmental Safety, 60, 324-349. |
| [15] | Parvaneh R, Shahrokh T, Hosseini SM (2012). Studying of salinity stress effect on germination, proline, sugar, protein, lipid and chlorophyll content in purslane (Portulace oloracea L.) leaves. Stress Physiology & Biochemistry, 8, 182-193. |
| [16] | Peng ZH, Peng KQ, Hu JJ, Xiao LT (2002). Research progress on accumulation of proline under osmotic stress in plants.Chinese Agriculture Science Bulletin, 18(4), 80-83. (in Chinese with English abstract)[彭志红, 彭克勤, 胡家金, 萧浪涛 (2002). 渗透胁迫下植物脯氨酸积累的研究进展. 中国农学通报, 18(4), 80-83.] |
| [17] | Sa RL, Liu JH, Liu L, Bai JH, Wang ZH (2014). Cation- responsive mechanisms of oats to alkali stress.Acta Agronomica Sinica, 40, 362-368. (in Chinese with English abstract)[萨如拉, 刘景辉, 刘伟, 白健慧, 王占海 (2014). 燕麦对碱胁迫的阳离子响应机制. 作物学报, 40, 362-368.] |
| [18] | Shi DC, Wang DL (2005). Effects of various salt-alkaline mixed stresses onAneurolepidium chinense (Trin.) Kitag. Plant and Soil, 271, 15-26. |
| [19] | Shi LX, Ma S, Fang Y, Xu J (2015). Crucial variations in growth and ion homeostasis of glycine gracilis seedlings under two types of salt stresses.Journal of Soil Science and Plant Nutrition, 15, 1-17. |
| [20] | Wang H, Wu ZH, Han JY, Zheng W, Yang CW (2012). Comparison of ion balance and nitrogen metabolism in old and young leaves of alkali-stressed rice plants.PLOS ONE, 7, e37817. doi: 10.1371/journal.pone.0037817. |
| [21] | Wang JY, Zhu SG, Xu CF (2002). Biochemistry. 3rd edn. Higher Education Press, Beijing. (in Chinese)[王镜岩, 朱圣庚, 徐长法 (2002). 生物化学. 第三版. 高等教育出版社, 北京.] |
| [22] | Wang XD, Wang C, Ma ZH, Hou RF, Gao Q, Chen Q (2011). Effect of short-term salt stress on the absorption of K+ and accumulation of Na+, K+ in seedlings of different wheat varieties.Acta Ecologica Sinica, 31, 2822-2830. (in Chinese with English abstract)[王晓冬, 王成, 马智宏, 侯瑞锋, 高权, 陈泉 (2011). 短期NaCl胁迫对不同小麦品种幼苗K+吸收和Na+、K+积累的影响. 生态学报, 31, 2822-2830.] |
| [23] | Wang XP, Geng SJ, Ri YJ, Cao DH, Liu J, Shi DC, Yang CW (2011). Physiological responses and adaptive strategies of tomato plants to salt and alkali stresses.Scientia Horticulturae, 130, 248-255. |
| [24] | Wu DZ, Shen QF, Cai SG, Chen ZH, Dai F, Zhang GP (2013). Ionomic responses and correlations between elements and metabolites under salt stress in wild and cultivated barley.Plant & Cell Physiology, 54, 1976-1988. |
| [25] | Yang CW, Li CY, Zhang ML, Liu J, Ju M, Shi DC (2008). pH and ion balance in wheat-wheatgrass under salt-or alkali stress.Chinese Journal of Applied Ecology, 19, 1000-1005. (in Chinese with English abstract)[杨春武, 李长有, 张美丽, 刘杰, 鞠淼, 石德成 (2008). 盐、碱胁迫下小冰麦体内的pH及离子平衡. 应用生态学报, 19, 1000-1005.] |
| [26] | Yang C, Shi D, Wang D (2008). Comparative effects of salt stress and alkali stress on growth, osmotic adjustment and ionic balance of an alkali resistant halophyteSuaeda glauca(Bge.). Plant Growth Regulation, 56, 179-190. |
| [27] | Yang T, Xie ZX, Yu Q, Liu XJ (2014). Effects of partial root salt stress on seedling growth and photosynthetic characteristics of winter wheat.Chinese Journal of Eco- Agrculture, 22, 1074-1078. (in Chinese with English abstract)[杨婷, 谢志霞, 喻琼, 刘小京 (2014). 局部根系盐胁迫对冬小麦生长和光合特征的影响. 中国生态农业学报, 22, 1074-1078.] |
| [28] | Yu RP (1999). Saline soil resources in China and their exploitation.Chinese Journal of Soil Science, 30(4), l58-159. (in Chinese with English abstract)[俞仁培 (1999). 我国盐渍土资源及其开发利用. 土壤通报, 30(4), l58-159.] |
| [29] | Zhu JK (2003). Regulation of ion homeostasis under salt stress.Current Opinion in Plant Biology, 6, 441-445. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2022 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn