

植物生态学报 ›› 2015, Vol. 39 ›› Issue (1): 72-80.DOI: 10.17521/cjpe.2015.0008
收稿日期:2014-06-09
接受日期:2014-11-02
出版日期:2015-01-10
发布日期:2015-01-22
通讯作者:
高玉葆
作者简介:# 共同第一作者
基金资助:
JIA Tong, REN An-Zhi, WEI Mao-Ying, YIN Li-Jia, GAO Yu-Bao*( )
)
Received:2014-06-09
Accepted:2014-11-02
Online:2015-01-10
Published:2015-01-22
Contact:
Yu-Bao GAO
About author:# Co-first authors
摘要:
通过田间试验, 研究了垂直传播的Neotyphodium属内生真菌和水平传播的Epichloë属内生真菌对羽茅(Achnatherum sibiricum)生理生态的影响。结果表明: Neotyphodium属内生真菌感染对羽茅的株高和叶长有显著的正效应, 而Epichloë属内生真菌感染对羽茅株高和叶长的增益作用较少。感染Neotyphodium属内生真菌的羽茅最大净光合速率显著高于感染Epichloë属内生真菌的羽茅, Neotyphodium属内生真菌显著提升了羽茅的气孔限制值和水分利用效率, 而实验测定的第一年结果显示: Epichloë属内生真菌对羽茅的各个光合指标具有明显的负效应(胞间CO2浓度除外)。Neotyphodium属内生真菌感染使得宿主植物积累的可溶性糖含量显著高于感染Epichloë属内生真菌的羽茅和不染菌的羽茅植株。感染Epichloë属内生真菌的羽茅中氮含量显著高于感染Neotyphodium属内生真菌的羽茅。总之, 内生真菌的传播方式是影响羽茅生理生态学特性的一个因素, 并且垂直传播的内生真菌对羽茅生长和生理特性的益处高于水平传播的内生真菌。
贾彤, 任安芝, 魏茂英, 尹立佳, 高玉葆. 不同传播方式的内生真菌感染对羽茅的生理生态影响. 植物生态学报, 2015, 39(1): 72-80. DOI: 10.17521/cjpe.2015.0008
JIA Tong,REN An-Zhi,WEI Mao-Ying,YIN Li-Jia,GAO Yu-Bao. Effects of endophyte transmission on ecophysiological characteristics of Achnatherum sibiricum. Chinese Journal of Plant Ecology, 2015, 39(1): 72-80. DOI: 10.17521/cjpe.2015.0008
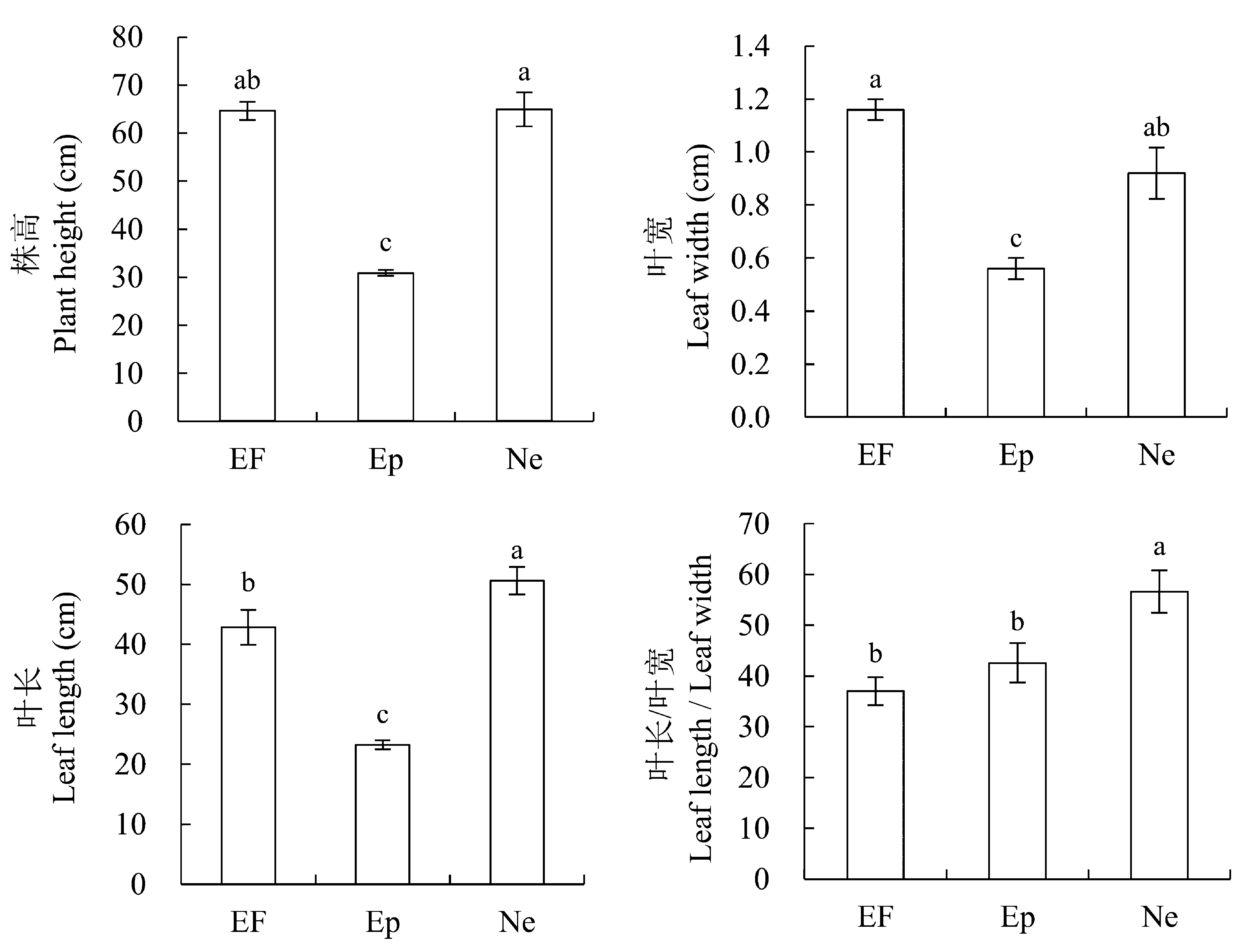
图1 2011年感染不同传播方式内生真菌Epichloë和Neotyphodium对羽茅生长状况的影响(平均值±标准误差)。不同小写字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。
Fig. 1 Effects of Epichloë and Neotyphodium infection on growth of Achnatherum sibiricum in 2011 (mean ± SE). Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium.
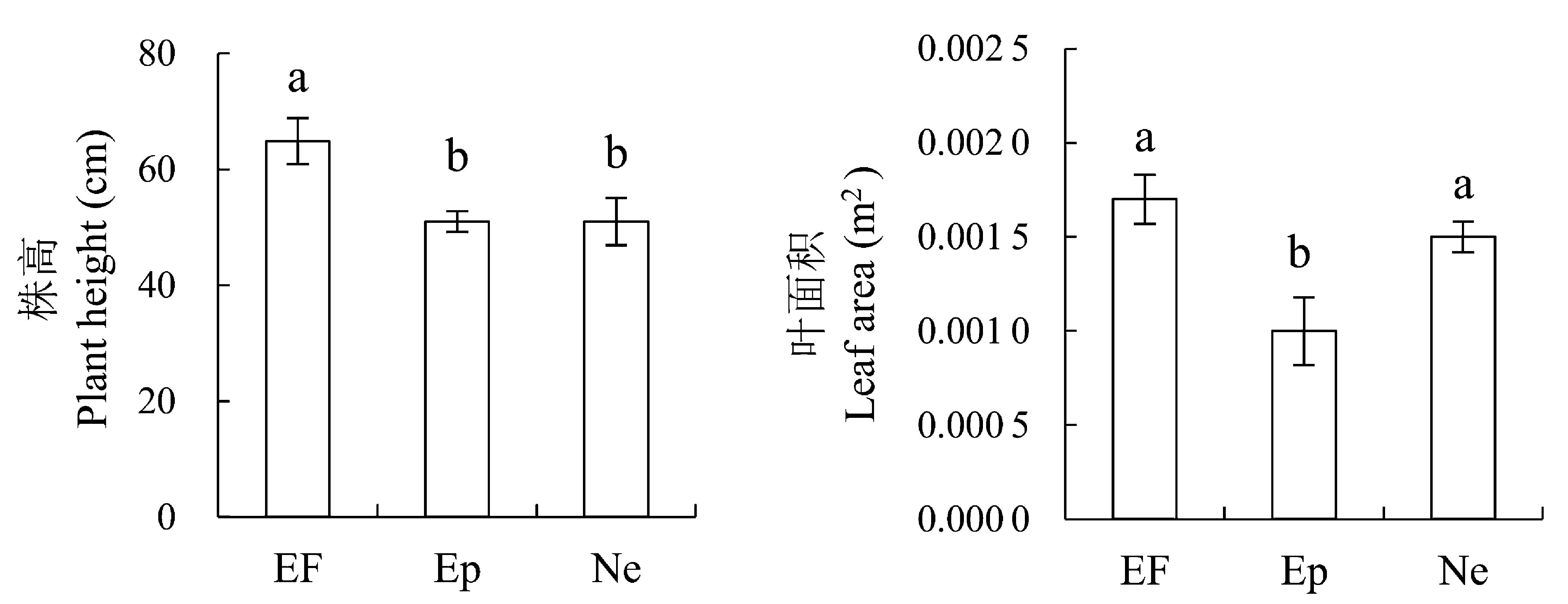
图2 2012年感染不同传播方式内生真菌Epichloë和Neotyphodium对羽茅生长状况的影响(平均值±标准偏差)。不同小写字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。
Fig. 2 Effects of Epichloë and Neotyphodium infection on growth of Achnatherum sibiricum in 2012 (mean ± SD). Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium.
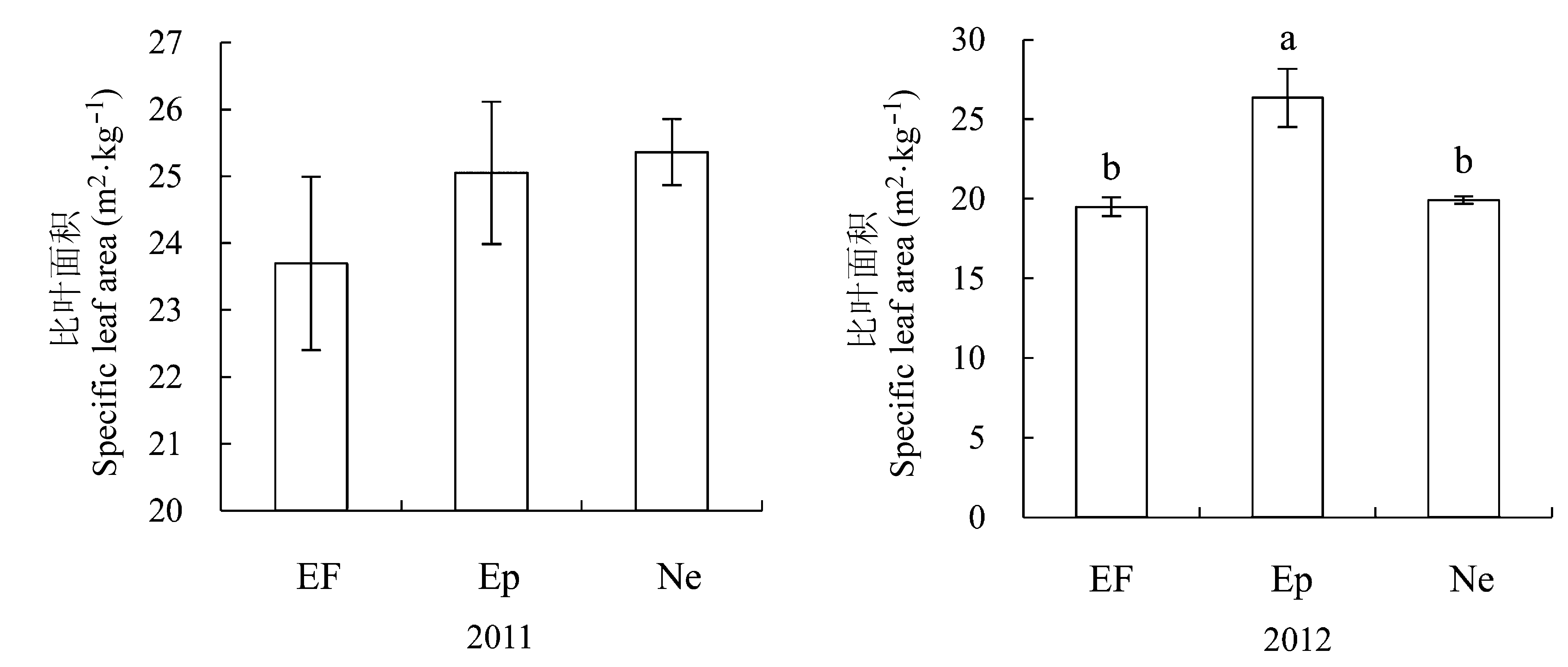
图3 感染不同传播方式内生真菌Epichloë和Neotyphodium对羽茅比叶面积的影响(平均值±标准偏差)。不同小写字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。
Fig. 3 Effects of Epichloë and Neotyphodium infection on specific leaf area in Achnatherum sibiricum (mean ± SD). Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium.
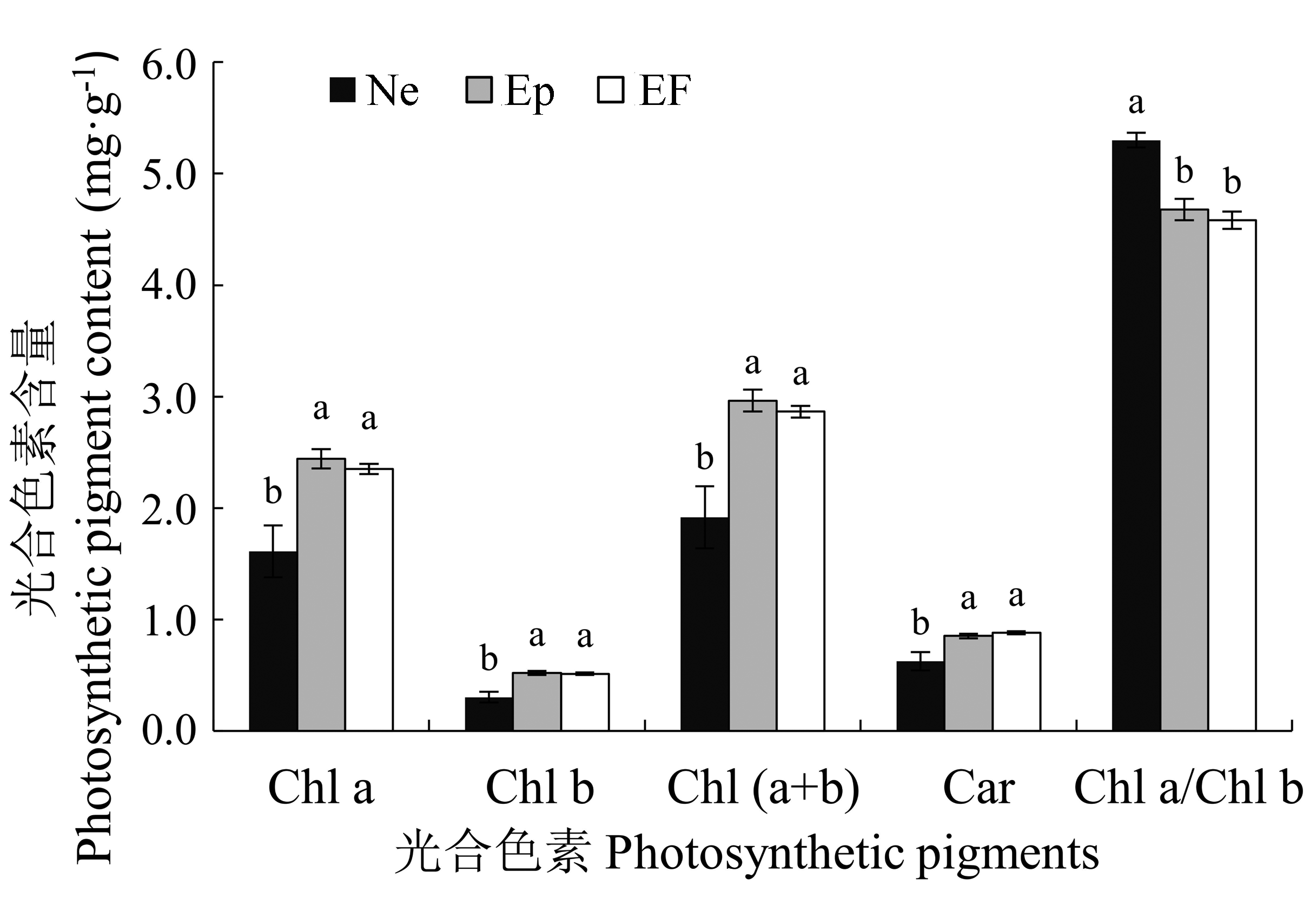
图4 2011年感染不同传播方式内生真菌Epichloë和Neotyphodium对羽茅光合色素的影响(平均值±标准误差)。不同小写字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。Car, 胡萝卜素; Chl a, 叶绿素a; Chl b, 叶绿素b。
Fig. 4 The influence of Epichloë and Neotyphodium infection on photosynthetic pigments in Achnatherum sibiricum in 2011 (mean ± SE). Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium. Car, carotene; Chl a, chlorophyll a; Chl b, chlorophyll b.
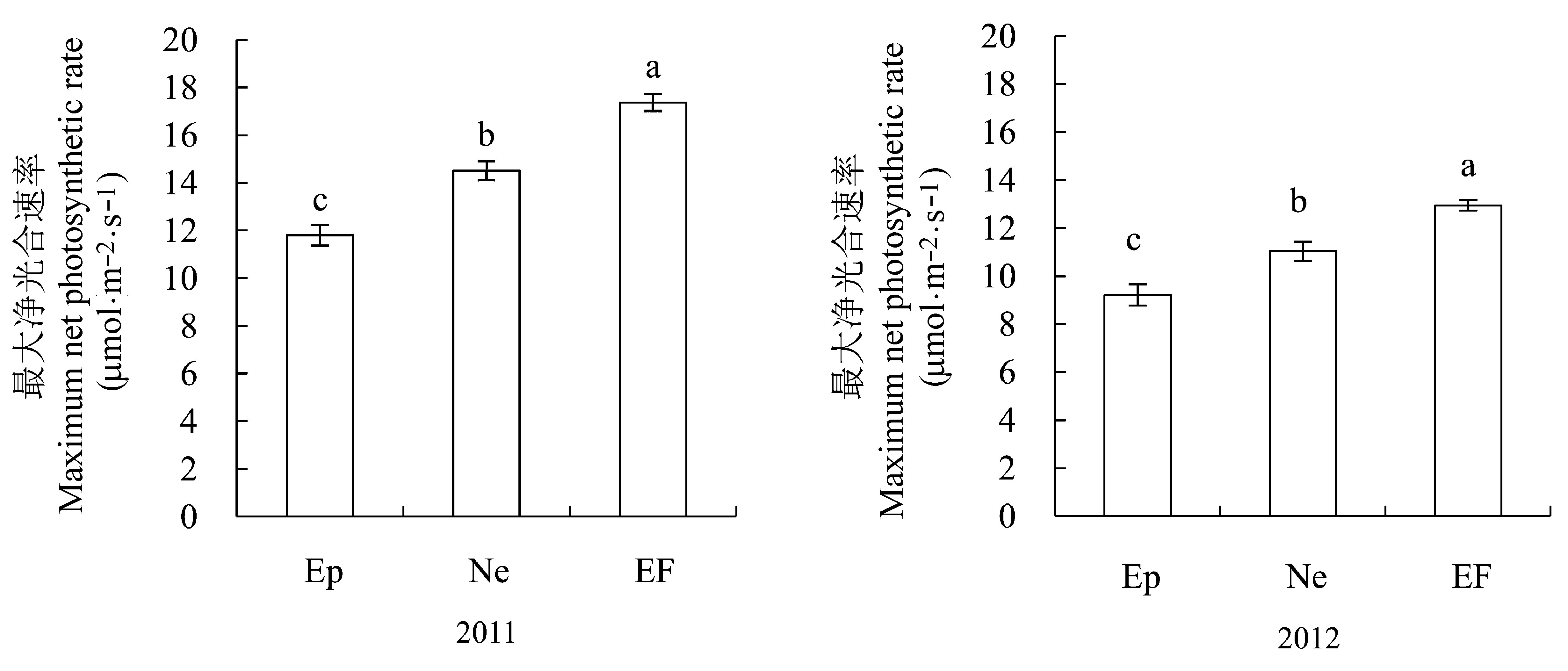
图5 感染不同传播方式内生真菌Epichloë和Neotyphodium对羽茅最大净光合速率的影响(平均值±标准误差)。不同字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。
Fig. 5 The influence of Epichloë and Neotyphodium infection on maximum net photosynthetic rate in Achnatherum sibiricum (mean ± SE). Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium.
| Year | 蒸腾速率 Transpiration rate (mmol·m-2·s-1) | 气孔导度 Stomatal conductance (mol·m-2·s-1) | 胞间CO2浓度 Intercellular CO2 concentration (μmol·mol-1) | 气孔限制值 Stomatal limitation | 水分利用效率 Water use efficiency (mmol·mmol-1) | 光能利用效率 Light use efficiency (μmol·μmol-1) | |
|---|---|---|---|---|---|---|---|
| 2011 | Ep | 5.83b | 0.33b | 320.6a | 0.19c | 2.035c | 0.009 8c |
| Ne | 4.76c | 0.30b | 294.8c | 0.26a | 3.086a | 0.012 1b | |
| EF | 6.47a | 0.42a | 306.1b | 0.23b | 2.712b | 0.014 5a | |
| 2012 | Ep | 2.07c | 0.09c | 210.47b | 0.48b | 4.52a | 0.008c |
| Ne | 2.44b | 0.09b | 199.80b | 0.51a | 4.53a | 0.009b | |
| EF | 3.45a | 0.15a | 249.13a | 0.38c | 3.74b | 0.011a |
表1 羽茅光合作用拟合值的比较
Table 1 The comparison of fitted values of photosynthetic characteristics in Achnatherum sibiricum
| Year | 蒸腾速率 Transpiration rate (mmol·m-2·s-1) | 气孔导度 Stomatal conductance (mol·m-2·s-1) | 胞间CO2浓度 Intercellular CO2 concentration (μmol·mol-1) | 气孔限制值 Stomatal limitation | 水分利用效率 Water use efficiency (mmol·mmol-1) | 光能利用效率 Light use efficiency (μmol·μmol-1) | |
|---|---|---|---|---|---|---|---|
| 2011 | Ep | 5.83b | 0.33b | 320.6a | 0.19c | 2.035c | 0.009 8c |
| Ne | 4.76c | 0.30b | 294.8c | 0.26a | 3.086a | 0.012 1b | |
| EF | 6.47a | 0.42a | 306.1b | 0.23b | 2.712b | 0.014 5a | |
| 2012 | Ep | 2.07c | 0.09c | 210.47b | 0.48b | 4.52a | 0.008c |
| Ne | 2.44b | 0.09b | 199.80b | 0.51a | 4.53a | 0.009b | |
| EF | 3.45a | 0.15a | 249.13a | 0.38c | 3.74b | 0.011a |
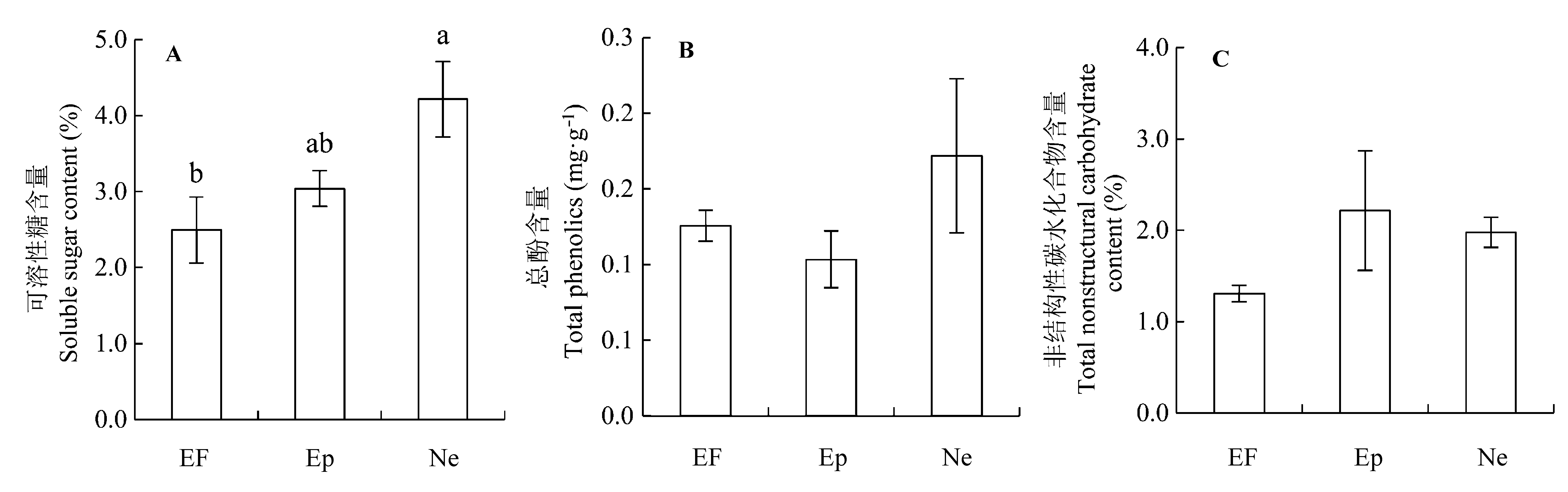
图6 感染不同传播方式内生真菌Epichloë和Neotyphodium对2011年羽茅可溶性糖和总酚含量的影响以及对2012年羽茅的非结构性碳含量的影响(平均值±标准偏差)。A, 可溶性糖含量。B, 总酚含量。C, 非结构性碳水化合物含量。不同小写字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。
Fig. 6 Effects of Epichloë and Neotyphodium infection on soluble sugar and total phenolic contents in 2011 and the total nonstructural carbohydrate in 2012 in Achnatherum sibiricum (mean ± SD). A, Soluble sugar content. B, Total phenolic content. C, Total nonstructural carbohydrate content. Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium.
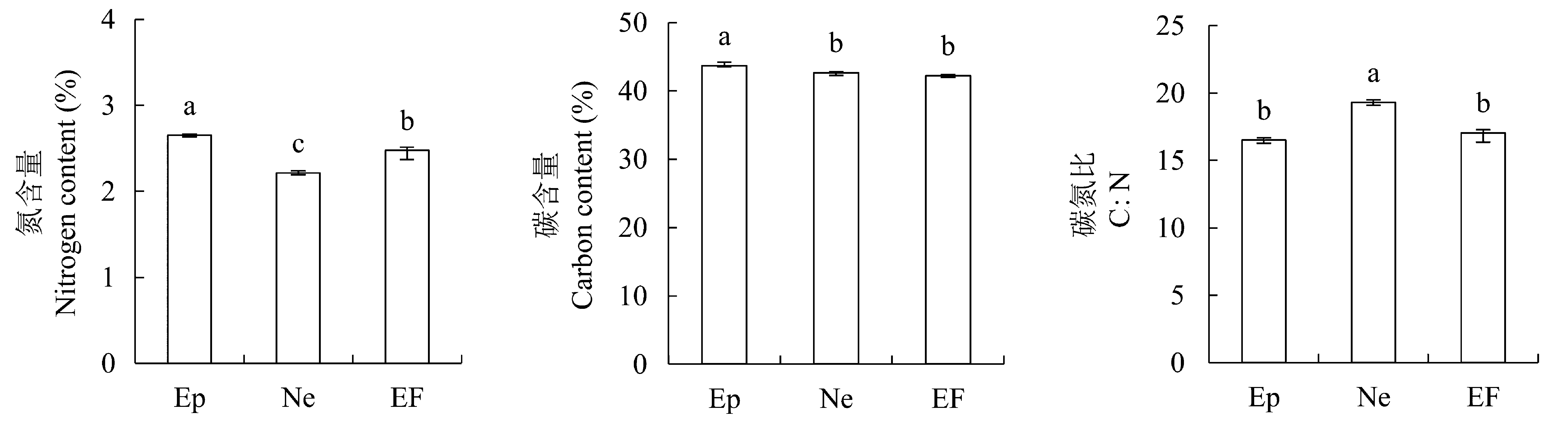
图7 2012年感染不同传播方式内生真菌Epichloë和Neotyphodium对羽茅的碳、氮含量以及碳氮比的影响(平均值±标准偏差)。不同小写字母表示p值在0.05的水平上差异显著。EF, 不染菌植株; Ep, 感染Epichloë的植株; Ne, 感染Neotyphodium的植株。
Fig. 7 Effects of Epichloë and Neotyphodium infection on nitrogen content, carbon content and C:N in Achnatherum sibiricum in 2012 (mean ± SD). Different lowercase letters indicate significant differences at the 0.05 level. EF, endophyte-free plant; Ep, plant infected by Epichloë; Ne, plant infected by Neotyphodium.
| 1 | Bradshaw AD, Snaydon RW (1959). Population differentiation within plant species in response to soil factors. Nature, 183, 129-130. |
| 2 | Bucheli E, Leuchtmann A (1996). Evidence for genetic |
| 3 | differentiation between choke-inducing and asymptomtic strains of the Epichloë grass endophyte from Brachy- podium sylvaticum.Evolution, 50, 1879-1887. |
| 4 | Bultman TL, Leuchtmann A (2003). A test of host specialization by insect vectors as a mechanism for reproductive isolation among entomophilous fungal species. Oikos, 103, 681-687. |
| 5 | Cheplick GP, Cho R (2003). Interactive effects of fungal endophyte infection and host genotype on growth and storage in Lolium perenne. New Phytologist, 158, 183-191. |
| 6 | Cheplick GP, Chui T (2001). Effects of competitive stress on vegetative growth, storage, and regrowth after defoliation in Phleum pratense. Oikos, 95, 291-299. |
| 7 | Cheplick GP, Faeth S (2009). Ecology and Evolution of the Grass-endophyte Symbiosis. Oxford University Press, Oxford, UK. |
| 8 | Clay K (1990). Fungal endophytes of grasses. Annual Review of Ecology and Systematics, 21, 275-297. |
| 9 | da Silveira AJ, Feitosa TFF, Stull JW (1978). A rapid technique for total nonstructural carbohydrate determination of plant tissue. Journal of Agricutural and Food Chemistry, 26, 770-772. |
| 10 | de Battista JP, Bacon CW, Severson R, Plattner RD, Bouton JH (1990). Indole acetic acid production by the fungal endophyte of tall fescue. Agronomy Journal, 82, 878-880. |
| 11 | Donaghy DJ, Fulkerson WJ (1997). The importance of water-soluble carbohydrate reserves on regrowth and root growth of Lolium perenne (L.). Grass and Forage Science, 52, 401-407. |
| 12 | Donaghy DJ, Fulkerson WJ (1998). Priority for allocation of water-soluble carbohydrate reserves during regrowth of Lolium perenne. Grass and Forage Science, 53, 211-218. |
| 13 | Elmi AA, West CP (1995). Endophyte infection effects on stomatal conductance, osmotic adjustment and drought recovery of tall fescue. New Phytologist, 131, 61-67. |
| 14 | Groppe K, Steinger T, Sanders I, Schmid B, Wiemken A, Boller T (1999). Interaction between the endophytic fungus Epichloe bromicola and the grass Bromus erectus: Effects of endophyte infection, fungal concentration and environment on grass growth and flowering. Molecular Ecology, 8, 1827-1835. |
| 15 | Hunt MG, Rasmussen S, Newton PCD, Parsons AJ, Newman JA (2005). Near-term impacts of elevated CO2, nitrogen and fungal endophyte-infection on Lolium perenne L. growth, chemical composition and alkaloid production. Plant, Cell & Environment, 28, 1345-1354. |
| 16 | Ji YL, Wang ZW, Yu HS, Wang SM (2003). Neotyphodium uncinatum, an endophytic fungus obtained from Festuca arundinacea Schreb. Journal of Nanjing Agricultural University, 26(2), 47-50. |
| (in Chinese with English abstract) [纪燕玲, 王志伟, 于汉寿, 王世梅 (2003). 分离自苇状羊茅(Festucaa rundinacea Schreb.)的内生真菌Neotyphodium uncinatum. 南京农业大学学报, 26(2), 47-50.] | |
| 17 | Kover PX, Clay K (1998). Trade-off between virulence and vertical transmission and the maintenance of a virulent plant pathogen. The American Naturalist, 152, 165-175. |
| 18 | Leuchtmann A, Schardl CL, Siegel MR (1994). Sexual compatibility and taxonomy of a new species of Epichloe symbiotic with fine Fescue grasses. Mycologia, 86, 802-812. |
| 19 | Leuchtmann A, Schmidt D, Bush LP (2000). Different levels of protective alkaloids in grasses with stroma-forming and seed-transmitted Epichloë/Neotyphodium endophytes. Jou- rnal of Chemical Ecology, 26, 1025-1036. |
| 20 | Li X, Han R, Ren AZ, Gao YB (2010). Using high-temperature treatment to construct endophyte-free Achnatherum sibiricum. Microbiology China, 37, 1395-1400. |
| [李夏, 韩荣, 任安芝, 高玉葆 (2010). 高温处理构建不感染内生真菌羽茅种群的方法探讨. 微生物学通报, 37, 1395-1400.] | |
| 21 | Malinowski DP, Alloush GA, Belesky DP (1998). Evidence for chemical changes on the root surface of tall fescue in response to infection with the fungal endophyte Neotyphodium coenophialum. Plant and Soil, 205, 1-12. |
| 22 | Malinowski DP, Belesky DP (2000). Adaptations of endophyte-infected cool-season grasses to environmental stresses: Mechanisms of drought and mineral stress tolerance. Crop Science, 40, 923-940. |
| 23 | Marks S, Clay K, Cheplick GP (1991). Effects of fungal endophytes on interspecific and intraspecific competition in the grasses Festuca arundinacea and Lolium perenne. Journal of Applied Ecology, 28, 194-204. |
| 24 | Moon CD, Craven KD, Leuchtmann A, Clement SL, Schardl CL (2004). Prevalence of interspecific hybrids amongst asexual fungal endophytes of grasses. Molecular Ecology, 13, 1455-1467. |
| 25 | Morse LJ, Day TA, Faeth SH (2002). Effect of Neotyphodium endophyte infection on growth and leaf gas exchange of Arizona fescue under contrasting water availability regimes. Environmental and Experimental Botany, 48, 257-268. |
| 26 | Rasmussen S, Parsons AJ, Bassett S, Christensen MJ, Hume DE, Johnson LJ, Johnson RD, Simpson WR, Stacke C, Voisey CR, Xue H, Newman JA (2007). High nitrogen supply and carbohydrate content reduce fungal endophyte and alkaloid concentration in Lolium perenne. New Phytologist, 173, 787-797. |
| 27 | Sánchez FJ, Manzanares M, de Andres EF, Tenorio JL, Ayerbe L (1998). Turgor maintenance, osmotic adjustment and soluble sugar and proline accumulation in 49 pea cultivars in response to water stress. Field Crops Research, 59, 225-235. |
| 28 | Schardl CL, Leuchtmann A, Spiering MJ (2004). Symbioses of grasses with seedborne fungal endophytes. Annual Review of Plant Biology, 55, 315-340. |
| 29 | Shen J, Tao WW, Chen C, Chen YG, Wang ZW (2009). Review on the grass endophyte research in China 9―Seed transmission characteristics and distribution in host plants of Epichloё yangzii. Pratacultural Science, 26, 146-151. |
| (in Chinese with English abstract) [申靖, 陶文文, 陈昌, 陈永敢, 王志伟 (2009). 禾本科植物内生真菌研究9——Epichloё yangzii的种传特性及其在宿主体内的分布. 草业科学, 26, 146-151.] | |
| 30 | Tintjer T, Leuchtmann A, Clay K (2008). Variation in horizontal and vertical transmission of the endophyte Epichloë elymi infecting the grass Elymus hystrix. New Phytologist, 179, 236-246. |
| 31 | Wei YK, Gao YB, Zhang X, Su D, Wang YH, Xu H, Lin F, Ren AZ, Chen L, Nie LY (2007). Distribution and diversity of Epichloë/Neotyphodium fungal endophytes from different populations of Achnatherum sibiricum (Poaceae) in the Inner Mongolia Steppe, China. Fungal Diversity, 24, 329-345. |
| 32 | White JF (1988). Endophyte-host associations in forage grasses. XI. A proposal concerning origin and evolution. Mycologia, 80, 442-446. |
| 33 | Zhang YP, Nan ZB (2007). Distribution of Epichloë endophytes in Chinese populations of Elymus dahuricus and variation in peramine levels. Symbiosis, 43, 13-19. |
| [1] | 石新建, 张靖歆, 秦天姿, 刘金铭, 高玉葆, 任安芝. 内生真菌感染对宿主羽茅及邻生植物抗病性的影响[J]. 植物生态学报, 2021, 45(8): 860-869. |
| [2] | 秦天姿, 任安芝, 樊晓雯, 高玉葆. 内生真菌种类和母本基因型对内生真菌-禾草共生体叶形状和叶面积的影响[J]. 植物生态学报, 2020, 44(6): 654-660. |
| [3] | 吴曼, 李娟娟, 刘金铭, 任安芝, 高玉葆. 刈割干扰和养分添加条件下Epichloë内生真菌感染对羽茅所在群落多样性和生产力的影响[J]. 植物生态学报, 2019, 43(2): 85-93. |
| [4] | 牛毅, 高远, 李隔萍, 任安芝, 高玉葆. 内生真菌对羽茅抗病性的影响[J]. 植物生态学报, 2016, 40(9): 925-932. |
| [5] | 魏宇昆, 高玉葆, 李川, 许华, 任安芝. 内蒙古中东部草原羽茅内生真菌的遗传多样性[J]. 植物生态学报, 2006, 30(4): 640-649. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2026 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
![]()