

植物生态学报 ›› 2007, Vol. 31 ›› Issue (4): 619-624.DOI: 10.17521/cjpe.2007.0079
张丽丽1,2, 董鸣1, 李仁强1,2, 王艳红1,2, 崔清国1,2, 何维明1,*( )
)
收稿日期:2006-06-27
接受日期:2007-01-05
出版日期:2007-06-27
发布日期:2007-07-30
通讯作者:
何维明
作者简介:*E-mail: weiminghe@ibcas.ac.c基金资助:
ZHANG Li-Li1,2, DONG Ming1, LI Ren-Qiang1,2, WANG Yan-Hong1,2, CUI Qing-Guo1,2, HE Wei-Ming1,*( )
)
Received:2006-06-27
Accepted:2007-01-05
Online:2007-06-27
Published:2007-07-30
Contact:
HE Wei-Ming
摘要:
克隆植物相连片段(或分株)常常生长在不同的土壤养分斑块中。克隆整合使得生长在异质养分斑块中的克隆片段(或分株)产生局部和非局部反应,从而影响相连片段(或分株)的表型可塑性。为了揭示养分斑块对比度对活血丹(Glechoma longituba)克隆整合的影响,在一控制实验中,将活血丹克隆片断种植于4种不同对比度的环境中,即:无对比度(对照)、低对比度、中对比度和高对比度。活血丹在气体交换、水势、荧光、形态、生长与分配方面的克隆整合强度随养分斑块对比度的增强而表现出增强或减弱的变化趋势;养分斑块对比度越强,活血丹气体交换和荧光暗反应的整合强度越小,叶片水势整合强度越大。斑块对比度可改变部分性状的克隆整合方向;超过一定的对比阈值,整合强度随养分对比度的变化趋势会向着相反方向转变。克隆整合对生理特征的修饰幅度小于对生长特征的修饰幅度。这些结果指示:养分斑块对比度可通过修饰克隆整合格局(即强度和方向)而改变克隆植物的表型可塑性。
张丽丽, 董鸣, 李仁强, 王艳红, 崔清国, 何维明. 土壤养分斑块对比度改变活血丹克隆整合强度和方向. 植物生态学报, 2007, 31(4): 619-624. DOI: 10.17521/cjpe.2007.0079
ZHANG Li-Li, DONG Ming, LI Ren-Qiang, WANG Yan-Hong, CUI Qing-Guo, HE Wei-Ming. SOIL-NUTRIENT PATCH CONTRAST MODIFIES INTENSITY AND DIRECTION OF CLONAL INTEGRATION IN GLECHOMA LONGITUBA. Chinese Journal of Plant Ecology, 2007, 31(4): 619-624. DOI: 10.17521/cjpe.2007.0079
图1 实验设计示意图(生长槽中点的浓淡深浅表征养分浓度的高低)
Fig.1 Layout of the experiment (The four different patterns stand for four different nutrient patches) C: Control LC: Low contrast MC: Medium contrast HC: high contrast
| 养分总量 Total amount of nutrients (g·m-3) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 对照 Control (C) | 低对比强度 Low contrast (LC) | 中对比强度 Medium contrast (MC) | 高对比强度 High contrast (HC) | ||||||||
| O | T | O | T | O | T | O | T | ||||
| N | 1 125 | 1 125 | 1 125 | 1 000 | 1 125 | 875 | 1 125 | 750 | |||
| P | 1 125 | 1 125 | 1 125 | 1 000 | 1 125 | 875 | 1 125 | 750 | |||
| K | 1 125 | 1 125 | 1 125 | 1 000 | 1 125 | 875 | 1 125 | 750 | |||
表1 不同对比养分斑块中的养分状况
Table 1 Total amount of nutrients used in the four contrasting nutrient patches
| 养分总量 Total amount of nutrients (g·m-3) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 对照 Control (C) | 低对比强度 Low contrast (LC) | 中对比强度 Medium contrast (MC) | 高对比强度 High contrast (HC) | ||||||||
| O | T | O | T | O | T | O | T | ||||
| N | 1 125 | 1 125 | 1 125 | 1 000 | 1 125 | 875 | 1 125 | 750 | |||
| P | 1 125 | 1 125 | 1 125 | 1 000 | 1 125 | 875 | 1 125 | 750 | |||
| K | 1 125 | 1 125 | 1 125 | 1 000 | 1 125 | 875 | 1 125 | 750 | |||
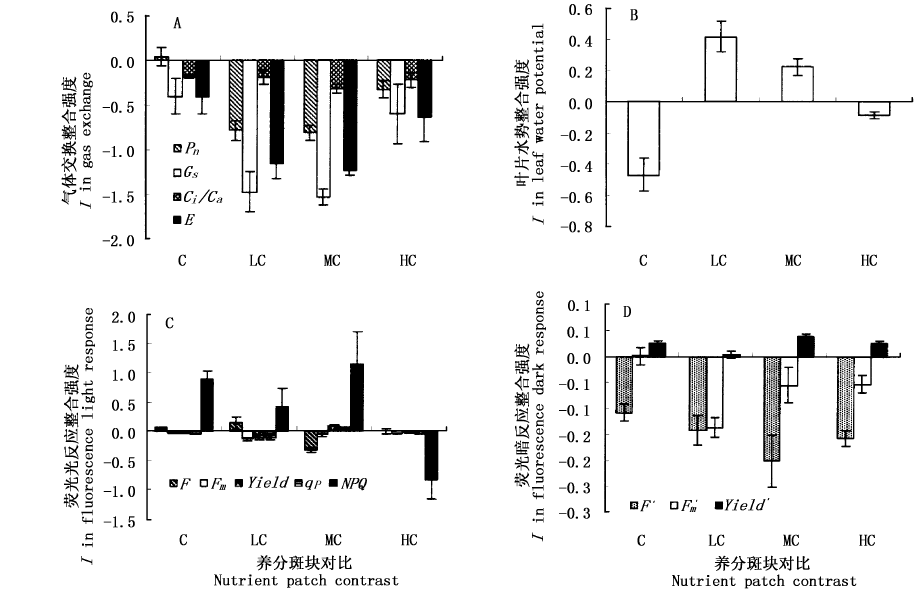
图2 不同养分斑块对比度下气体交换(A),水势(B)和荧光反应(C, D)的克隆整合格局
Fig.2 Clonal integration (I) in physiological function along a nutrient-patch contrast gradient Pn:叶片净光合速率 Net photosynthetic rate Gs:气孔导度 Stomatal conductance Ci/Ca:胞内外CO2浓度比 Ratio of substomatal CO2 to atmospheric CO2 E:蒸腾速率 Transpiration rate F:最小荧光Minimal fluorescence in the light conditions Fm:最大荧光 Maximal fluorescence in the light conditions Yield:最大光化学量子产量 Fluorescence efficiency of PSⅡ in the light conditions qP:光化学淬灭系数 Photochemical quenching in the light conditions NPQ:非光化学淬灭系数 Nonphotochemical quenching in the light conditions F':最小荧光 Minimal fluorescence in the dark conditions Fm':最大荧光 Maximal fluorescence in the dark conditions Yield':最大光化学量子产量 Fluorescence efficiency of PSⅡin the dark conditions C、LC、MC、HC:同图1 See Fig.1
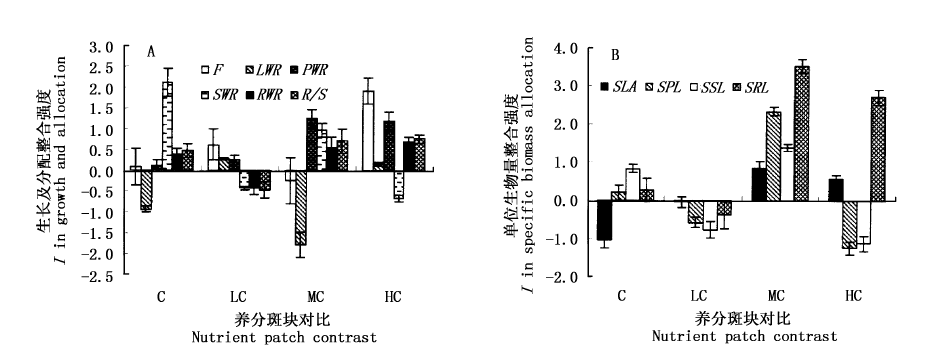
图3 养分斑块对比度对生长与分配(A)和比生物量分配(B)整合强度的影响
Fig.3 Clonal integration (I) in biomass growth and allocation (A) and specific biomass allocation (B) along a nutrient-patch contrast gradient F:克隆片段生物量 Fragment biomass LWR:叶片生物量比 Lamina weight ratio PWR:叶柄生物量比 Petiole weight ratio SWR:匍匐茎生物量比 Stolon weight ratio RWR:根生物量比 Root weight ratio R/S:根冠比 Root/shoot ratio SLA:比叶面积 Specific lamina area SPL:比叶柄长 Specific petiole length SSL:比茎长 Specific stolon length SRL:比根长 Specific root length C、LC、MC、HC:同图1 See Fig.1
| [1] |
Alpert P (1999). Effects of clonal integration on plant plasticity in Fragaria chiloensis. Plant Ecology, 141, 99-106.
DOI URL |
| [2] |
Caraco T, Kelly CK (1991). On the adaptive value of physiological integration in clonal plants. Ecology, 72, 81-93.
DOI URL |
| [3] |
Dong M (1995). Morphological responses to local light conditions in clonal herbs from contrasting habitats, and their modification due to physiological integration. Oecologia, 101, 282-288.
DOI URL PMID |
| [4] | Dong M (董鸣) (1996). Clonal growth in plants in relation to resource heterogeneity: foraging behavior. Acta Botanica Sinica (植物学报), 88, 828-835. (in Chinese with English abstract) |
| [5] | Eriksson O, Jerling L 1990. Hierarchical selection and risk spreading in clonal plants. In: van Groenendael J, de Kroon H eds. Clonal Growth in Plants: Regulation and Function. SPB Academic Publishing, the Hague, the Netherlands, 79-94. |
| [6] |
Evans JP (1992). The effect of local resource availability and clonal integration on ramet functional morphology in Hydrocotyle bonariensis. Oecologia, 86, 268-275.
DOI URL PMID |
| [7] |
Farley RA, Fitter AH (1999). The response of seven co-occurring woodland herbaceous perennials to localized nutrient-rich patches. Journal of Ecology, 87, 849-859.
DOI URL |
| [8] | He WM (何维明), Dong M (董鸣) (2002). Ramets and genets in the tillering clonal herb Panicum miliaceum in hierarchical response to heterogeneous nutrient environments. Acta Ecologica Sinica (生态学报), 20, 169-175. (in Chinese with English abstract) |
| [9] |
Hodge A (2004). The plastic plant: root responses to heterogeneous supplies of nutrients. New Phytologist, 162, 9-24.
DOI URL |
| [10] | Hutchings MJ, de Kroon H (1994). Foraging in plants: the role of morphological plasticity in resource acquisition. Advances in Ecological Research, 25, 159-238. |
| [11] |
Hutchings MJ, Wijesinghe DK (1997). Patchy habitats, division of labour and growth dividends in clonal plants. Trends in Ecology and Evolution, 12, 390-394.
DOI URL PMID |
| [12] |
Jónsdóttir IS, Callaghan TV (1990). Intraclonal traslocation of ammonium and nitrate in Carex bigelowii using 15N and nitrate reductase assays. New Phytologist, 114, 419-426.
DOI URL |
| [13] | Jónsdóttir IS, Watson MA 1997. Extensive physiological integration: an adaptive trait in resource-poor environments? In: de Kroon H, van Groenendael J eds. The Ecology and Evolution of Clonal Plants. Backhuys Publishers, Leiden, the Netherlands, 109-136. |
| [14] | Pitelka LF, Ashmun JW 1985. Physiology and integration of ramets in clonal plants. In: Jackson JBC, Buss LW, Cook RE eds. Population Biology and Evolution of Clonal Organisms. Yale University Press, New Haven, London, 1-55. |
| [15] | Shan BQ (单保庆), Du GZ (杜国祯), Liu ZH (刘振恒) (2000). Clonal growth of Ligularia virgaurea: morphological responses to nutritional variation. Acta Phytoecologica Sinica (植物生态学报), 24, 46-51. (in Chinese with English abstract) |
| [16] |
Stuefer JF, During HJ, de Kroon H (1994). High benefits of clonal integration in two stoloniferous species, in response to heterogeneous light environments. Journal of Ecology, 82, 511-518.
DOI URL |
| [17] |
Stuefer JF, Hutchings MJ (1994). Environmental heterogeneity and clonal growth: a study of the capacity for reciprocal translocation in Glechoma hederacea L. Oecologia, 100, 302-308.
DOI URL PMID |
| [18] |
Stuefer JF (1996). Potential and limitations of current concepts regarding the response of clonal plants to environmental heterogeneity. Vegetatio, 127, 55-70.
DOI URL |
| [19] | Svesson BM, Callaghan TV (1988). Small-scale vegetation pattern related to the growth of Lycopodium annotinum and variations in its micro-environment. Vegetatio, 76, 167-177. |
| [20] |
Turkington R, Hamilton RS, Gliddon C (1991). Within population variation in localized and integrated responses of Trifolicum repens to biologically patchy environments. Oecologia, 86, 183-192.
DOI URL PMID |
| [21] |
Wijesinghe DK, Handel SN (1994). Advantages of clonal growth in heterogeneous habitats: an experiment with Potentilla simplex. Journal of Ecology, 82, 495-502.
DOI URL |
| [22] | Zhang CY (张称意), Yang C (杨持), Dong M (董鸣) (2001). The clonal integraion of photosynthate in the rhizomatous half-shrub Hedysarum leave. Acta Ecologica Sinica (生态学报), 21, 1986-1993. (in Chinese with English abstract) |
| [1] | 杜英东, 袁相洋, 冯兆忠. 不同形态氮对杨树光合特性及生长的影响[J]. 植物生态学报, 2023, 47(3): 348-360. |
| [2] | 杨丽婷, 谢燕燕, 左珂怡, 徐森, 谷瑞, 陈双林, 郭子武. 分株比例对异质光环境下美丽箬竹克隆系统光合生理的影响[J]. 植物生态学报, 2022, 46(1): 88-101. |
| [3] | 胡俊靖, 陈双林, 郭子武, 陈卫军, 杨清平, 李迎春. 美丽箬竹水分生理整合的分株比例效应——基于叶片抗氧化系统与光合色素[J]. 植物生态学报, 2015, 39(7): 762-772. |
| [4] | 张翠萍,孟平,张劲松,万贤崇. 固氮植物绿豆对核桃幼苗生长、叶片气孔气体交换及水力特征的作用[J]. 植物生态学报, 2014, 38(5): 499-506. |
| [5] | 夏江宝, 张淑勇, 赵自国, 赵艳云, 高源, 谷广义, 孙景宽. 贝壳堤岛旱柳光合效率的土壤水分临界效应及其阈值分级[J]. 植物生态学报, 2013, 37(9): 851-860. |
| [6] | 张梓瑜,龚吉蕊,刘敏,黄永梅,晏欣,祁瑜,王忆慧. 温带草原不同土地利用方式下优势种植物和生态系统的气体交换[J]. 植物生态学报, 2013, 37(8): 718-727. |
| [7] | 陈坚,李妮亚,刘强,钟才荣,黄敏,曾佳. NaCl处理下两种引进红树的光合及抗氧化防御能力[J]. 植物生态学报, 2013, 37(5): 443-453. |
| [8] | 王林, 冯锦霞, 万贤崇. 土层厚度对刺槐旱季水分状况和生长的影响[J]. 植物生态学报, 2013, 37(3): 248-255. |
| [9] | 胡凤琴, 牟溥. 植物吸收根的增殖和生长与养分变异的关系——臭椿、翠菊、加拿大一枝黄花分根实验的启示[J]. 植物生态学报, 2013, 37(2): 93-103. |
| [10] | 李维, 张亚黎, 胡渊渊, 杨美森, 吴洁, 张旺锋. 田间条件下棉花幼叶光合特性及光保护机制[J]. 植物生态学报, 2012, 36(7): 662-670. |
| [11] | 葛俊, 邢福. 克隆植物对种间竞争的适应策略[J]. 植物生态学报, 2012, 36(6): 587-596. |
| [12] | 唐艳, 王传宽. 东北主要树种光合作用可行的离体测定方法[J]. 植物生态学报, 2011, 35(4): 452-462. |
| [13] | 金清, 江洪, 余树全, 殷秀敏. 酸雨胁迫对苦槠幼苗气体交换与叶绿素荧光的影响[J]. 植物生态学报, 2010, 34(9): 1117-1124. |
| [14] | 何茜, 李吉跃, 沈应柏, 陈晓阳, 尚富华, 胡磊, 张志毅. 毛白杨杂种无性系叶片δ13C差异与气体交换参数[J]. 植物生态学报, 2010, 34(2): 144-150. |
| [15] | 顾舒平, 尹黎燕, 李洁琳, 李伟. 不同碱度条件下中华水韭昼夜CO2气体交换的特征[J]. 植物生态学报, 2009, 33(6): 1184-1190. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2022 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19