

植物生态学报 ›› 2012, Vol. 36 ›› Issue (11): 1172-1183.DOI: 10.3724/SP.J.1258.2012.01172
收稿日期:2012-04-25
修回日期:2012-09-18
出版日期:2012-04-25
发布日期:2012-11-09
通讯作者:
牟溥
作者简介: (E-mail: ppmou@bnu.edu.cn)Received:2012-04-25
Revised:2012-09-18
Online:2012-04-25
Published:2012-11-09
Contact:
MOU Pu
摘要:
为验证以下3个假设: 1) NO3 -和NH4 +及其不同供给方式显著影响根系生长; 2) NO3 -和NH4 +以及不同供给方式对根内激素含量影响显著; 3)根构型(1级根长、单位2级根上1级侧根密度(分枝强度)和1级根在2级根上的根间距)与根内激素(生长素(IAA)、脱落酸(ABA)和细胞分裂素(玉米素核苷+玉米素) (CK (ZR + Z))含量显著相关, 采用营养液培养方法, 使实验植物翠菊(Callistephus chinensis)在两种氮肥(NO3 -和NH4 +)、不同施氮浓度(NO3 -: 0.2、1.0和18.0 mmol·L -1; NH4 +: 0.2、4.0和20.0 mmol·L -1), 以及脉冲和稳定两种施用方式处理下生长。在处理35天后收获植物, 测定根系生物量、根系构型指标(根系1级根长、单位2级根上1级侧根数和1级根在2级根上的根间距)和根系中激素含量(IAA、ABA和CK (ZR + Z))。结果显示: 1)实验处理对根生物量和根系中IAA、ABA和CK (ZR + Z)含量均有不同程度的显著影响: 施用NH4 +使根生物量和根内IAA含量显著低于施用NO3 -; 高浓度NO3 -和NH4 +处理亦使根生物量和IAA降低; 相对于稳定处理, 脉冲施氮显著降低根生物量和根内IAA含量; NO3 -使根内CK (ZR + Z)含量显著高于施用NH4 +, 且与施氮浓度及施氮方式无关; NO3 -处理下, 高浓度使根内ABA含量提高, 且脉冲处理使ABA含量升高。NH4 +处理下, 高浓度使根内ABA含量降低, 而施氮方式对其没有显著影响。2)根构型因素与根内激素关系各异: 各激素与1级根间距无显著关系; IAA和CK (ZR + Z)与1级根长和侧根密度有显著回归关系。3)根构型因素与根生物量的关系是根生物量与1级根长和侧根密度有显著正回归关系, 与1级根间距无显著回归关系。实验结果表明翠菊根生长的 “反常”可能是由于其对脉冲高浓度NH4 +耐受阈值低所致。该研究通过实验建立了氮养分种类/供应方式通过改变激素、影响根构型而影响根生长的联系, 进一步探究了植物根养分捕获塑性机制。
董佳, 牟溥. 翠菊根系养分捕获形态塑性及其生理机制. 植物生态学报, 2012, 36(11): 1172-1183. DOI: 10.3724/SP.J.1258.2012.01172
DONG Jia, MOU Pu. Root nutrient foraging of morphological plasticity and physiological mechanism in Calliste- phus chinensis. Chinese Journal of Plant Ecology, 2012, 36(11): 1172-1183. DOI: 10.3724/SP.J.1258.2012.01172
| 氮肥种类 N fertilizer type | 处理 Treatment | 氮肥浓度 Nitrogen concentration (mmol·L-1) | 施用方式 Applications | 每周期处理时间 Treatment time per period (h) |
|---|---|---|---|---|
| NO3- | A | 0.2/18.0 | 脉冲 Pulse | 62/10 |
| B | 1.0/18.0 | 脉冲 Pulse | 62/10 | |
| C | 0.2 | 稳定 Stable | 72 | |
| D | 1.0 | 稳定 Stable | 72 | |
| E | 18.0 | 稳定 Stable | 72 | |
| NH4+ | A | 0.2/20.0 | 脉冲 Pulse | 62/10 |
| B | 4.0/20.0 | 脉冲 Pulse | 62/10 | |
| C | 0.2 | 稳定 Stable | 72 | |
| D | 4.0 | 稳定 Stable | 72 | |
| E | 20.0 | 稳定 Stable | 72 |
表1 翠菊施氮浓度及浓度变换脉冲处理
Table 1 A list of N fertilization treatments (concentration and concentration pulse change) applied to Callistephus chinensis
| 氮肥种类 N fertilizer type | 处理 Treatment | 氮肥浓度 Nitrogen concentration (mmol·L-1) | 施用方式 Applications | 每周期处理时间 Treatment time per period (h) |
|---|---|---|---|---|
| NO3- | A | 0.2/18.0 | 脉冲 Pulse | 62/10 |
| B | 1.0/18.0 | 脉冲 Pulse | 62/10 | |
| C | 0.2 | 稳定 Stable | 72 | |
| D | 1.0 | 稳定 Stable | 72 | |
| E | 18.0 | 稳定 Stable | 72 | |
| NH4+ | A | 0.2/20.0 | 脉冲 Pulse | 62/10 |
| B | 4.0/20.0 | 脉冲 Pulse | 62/10 | |
| C | 0.2 | 稳定 Stable | 72 | |
| D | 4.0 | 稳定 Stable | 72 | |
| E | 20.0 | 稳定 Stable | 72 |
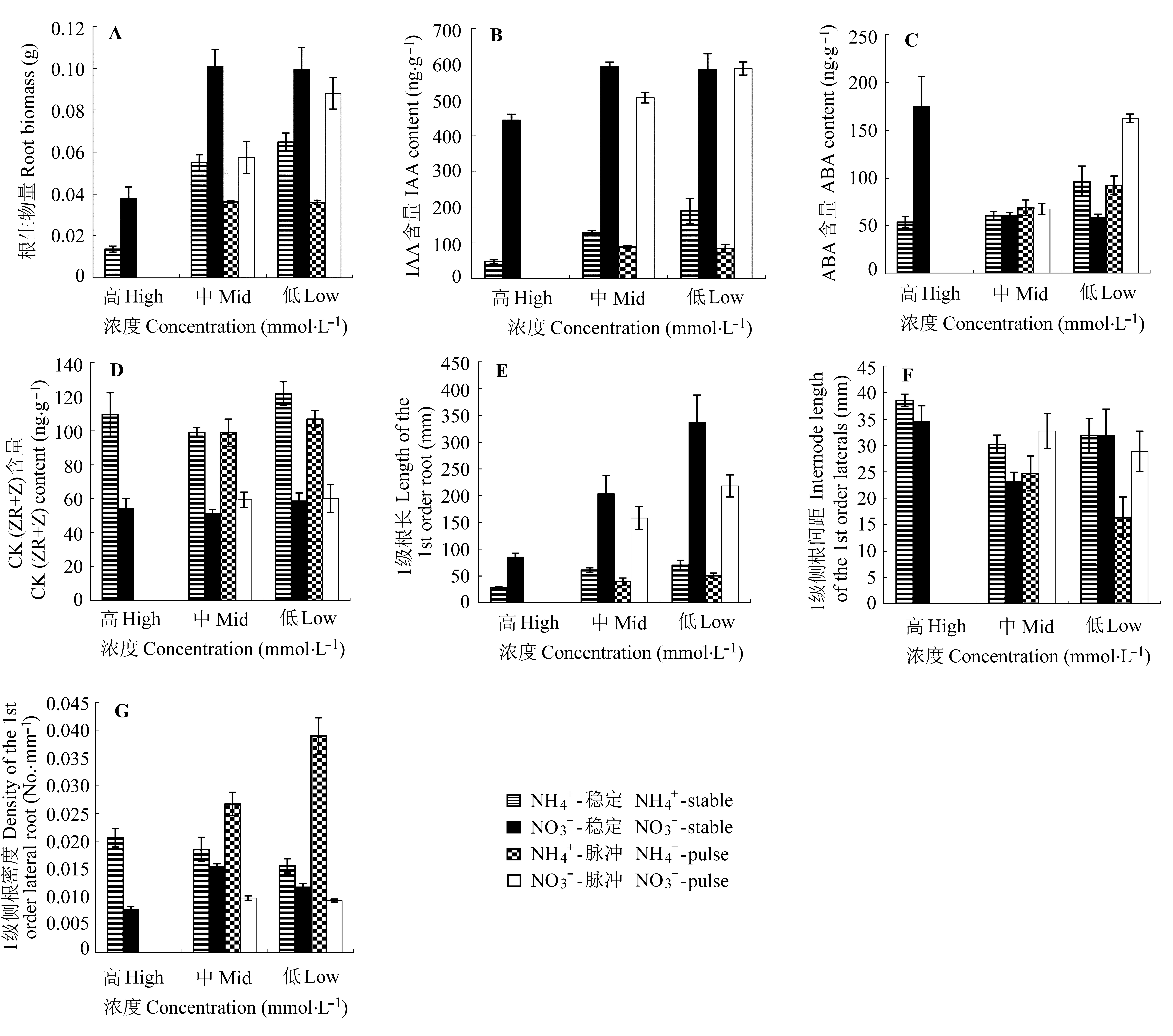
图1 氮肥种类、施用方式和浓度对根生物量(A)、生长素(IAA) (B)、脱落酸(ABA) (C)、细胞分裂素(CK (ZR + Z)) (D)含量、1级根长(E)、1级侧根间距(F)和1级侧根密度(G)的影响(平均值±标准误差)。
Fig. 1 Effects of N fertilizer type, application method and concentration on root biomass (A), contents of auxin (IAA) (B), abscisic acid (ABA) (C), cytokinins (CK(ZR + Z)) (D) in roots, length of the 1st order roots (E), internode length of the 1st order laterals (F), and the density of the 1st order lateral roots (G) (mean ± SE).
| 来源 Source | 自由度df | log(根生物量 Root biomass) | log (IAA) | log (ABA) | Log (CK (ZR + Z)) | 1级根长 Length of 1st order root | 1级侧根密度 Density of 1st order lateral roots | 1级侧根间距 Internode length of the 1st order laterals | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | ||
| 模型修正 Model corr. | 9 | 0.270 | 44.64 | *** | 0.850 | 106.40 | *** | 0.140 | 11.39 | *** | 0.100 | 18.11 | *** | 5.030 | 23.15 | *** | 0.016 | 3.31 | ** | 3.360 | 25.85 | *** |
| 施氮种类 N-type (A) | 1 | 0.720 | 119.00 | *** | 6.770 | 844.19 | *** | 0.250 | 19.86 | *** | 0.810 | 142.90 | *** | 19.170 | 88.22 | *** | 0.013 | 2.67 | 18.280 | 140.63 | *** | |
| 施氮方式 Application (B) | 1 | 0.300 | 50.05 | *** | 0.180 | 22.66 | *** | 0.140 | 11.22 | *** | 0.003 | 0.00 | 3.140 | 14.44 | *** | 0.004 | 0.88 | 2.430 | 18.67 | *** | ||
| 施氮浓度 Concentration (C) | 2 | 0.740 | 122.62 | *** | 0.300 | 37.83 | *** | 0.190 | 14.55 | *** | 0.009 | 1.64 | 5.400 | 24.85 | *** | 0.012 | 2.64 | 0.090 | 0.70 | |||
| AB | 1 | 0.000 | 0.06 | 0.110 | 13.55 | ** | 0.100 | 7.76 | 0.008 | 1.44 | 1.220 | 5.62 | * | 0.040 | 8.60 | ** | 7.040 | 54.15 | *** | |||
| AC | 2 | 0.022 | 3.64 | * | 0.110 | 13.87 | *** | 0.260 | 20.73 | * | 0.001 | 0.26 | 2.860 | 13.15 | *** | 0.005 | 1.03 | 0.900 | 6.89 | ** | ||
| BC | 1 | 0.008 | 1.34 | 0.007 | 0.82 | 0.069 | 5.45 | 0.007 | 1.25 | 0.300 | 1.36 | 0.016 | 3.35 | 1.520 | 11.66 | ** | ||||||
| ABC | 1 | 0.023 | 3.83 | 0.038 | 4.71 | * | 0.120 | 9.30 | * | 0.000 | 0.01 | 0.200 | 0.94 | <0.001 | 0.00 | 0.640 | 4.93 | * | ||||
| 误差 Error | 36 | 0.001 | 0.008 | 0.013 | 0.006 | 0.220 | 0.005 | 0.130 | ||||||||||||||
表2 多元方差分析结果表示氮肥种类(NO3-、NH4+)、施用方式(稳定、脉冲)和浓度对翠菊根生物量、根内生长素(IAA)、脱落酸(ABA)和细胞分裂素(CK (ZR + Z))含量和根构型(1级根长、1级侧根间距和1级侧根密度)的影响
Table 2 MANOAV results illustrating the influences of N fertilizer types (NO3-, NH4+), application methods (stable & pulse) and concentration on root biomass, content of auxin (IAA), abscisic acid (ABA) and cytokinins (CK (ZR + Z)) in roots, and root structure (length of the 1st order roots, internode length of the 1st order laterals and density of the 1st order laterals) for Callistephus chinensis
| 来源 Source | 自由度df | log(根生物量 Root biomass) | log (IAA) | log (ABA) | Log (CK (ZR + Z)) | 1级根长 Length of 1st order root | 1级侧根密度 Density of 1st order lateral roots | 1级侧根间距 Internode length of the 1st order laterals | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | 均方 MS | F | 显著性 sig. | ||
| 模型修正 Model corr. | 9 | 0.270 | 44.64 | *** | 0.850 | 106.40 | *** | 0.140 | 11.39 | *** | 0.100 | 18.11 | *** | 5.030 | 23.15 | *** | 0.016 | 3.31 | ** | 3.360 | 25.85 | *** |
| 施氮种类 N-type (A) | 1 | 0.720 | 119.00 | *** | 6.770 | 844.19 | *** | 0.250 | 19.86 | *** | 0.810 | 142.90 | *** | 19.170 | 88.22 | *** | 0.013 | 2.67 | 18.280 | 140.63 | *** | |
| 施氮方式 Application (B) | 1 | 0.300 | 50.05 | *** | 0.180 | 22.66 | *** | 0.140 | 11.22 | *** | 0.003 | 0.00 | 3.140 | 14.44 | *** | 0.004 | 0.88 | 2.430 | 18.67 | *** | ||
| 施氮浓度 Concentration (C) | 2 | 0.740 | 122.62 | *** | 0.300 | 37.83 | *** | 0.190 | 14.55 | *** | 0.009 | 1.64 | 5.400 | 24.85 | *** | 0.012 | 2.64 | 0.090 | 0.70 | |||
| AB | 1 | 0.000 | 0.06 | 0.110 | 13.55 | ** | 0.100 | 7.76 | 0.008 | 1.44 | 1.220 | 5.62 | * | 0.040 | 8.60 | ** | 7.040 | 54.15 | *** | |||
| AC | 2 | 0.022 | 3.64 | * | 0.110 | 13.87 | *** | 0.260 | 20.73 | * | 0.001 | 0.26 | 2.860 | 13.15 | *** | 0.005 | 1.03 | 0.900 | 6.89 | ** | ||
| BC | 1 | 0.008 | 1.34 | 0.007 | 0.82 | 0.069 | 5.45 | 0.007 | 1.25 | 0.300 | 1.36 | 0.016 | 3.35 | 1.520 | 11.66 | ** | ||||||
| ABC | 1 | 0.023 | 3.83 | 0.038 | 4.71 | * | 0.120 | 9.30 | * | 0.000 | 0.01 | 0.200 | 0.94 | <0.001 | 0.00 | 0.640 | 4.93 | * | ||||
| 误差 Error | 36 | 0.001 | 0.008 | 0.013 | 0.006 | 0.220 | 0.005 | 0.130 | ||||||||||||||
| 变量间相互关系 Relationship between variables | 响应变量 Dependent variable (Y) | 因变量(X) Independent variable | 回归关系 Regression | R2 | p |
|---|---|---|---|---|---|
| 根生长和根构型关系 Relationships between root growth and root architecture parameters | 根干重 Root dry mass (g) | 1级根长 Length of 1st order roots (mm) | Y = 0.02162 + 0.03696X - 0.00348X2 | 0.693 | p < 0.0001 |
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 0.0801 - 0.01265X | 0.156 | p = 0.017 | ||
| 根生长和激素关系 Relationships between root growth and hormones | 根干重 Root dry mass (g) | IAA | Y = 125.434 + 1.5829X | 0.633 | p < 0.0001 |
| ABA | 关系复杂 Complex relationship | ||||
| CK (ZR + Z) | Y = 110.3072e(-5.9906X) | 0.257 | p < 0.0001 | ||
| 根构型和激素关系 Relationships between root architecture parameters and hormones | IAA (ng·g-1) | 1级根长 Length of 1st order roots (mm) | Y = 0.319895e(0.003427X) | 0.845 | p < 0.0001 |
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 3.2525 - 0.00975X + 1.04299(e-005X2) | 0.515 | p < 0.0001 | ||
| ABA (ng·g-1) | 1级根长 Length of 1st order roots (mm) | 无显著回归关系 Insignificant regression relation | |||
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 2.3737e(-0.0045X) | 0.171 | p = 0.012 | ||
| CK (ZR + Z) ( ng·g-1) | 1级根长 Length of 1st order roots (mm) | Y = 5.2567e(-0.02124X) | 0.527 | p < 0.0001 | |
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 2.0851 + 0.07907X - 0.000345X2 | 0.402 | p < 0.0001 | ||
| 激素间相互关系 Relationships among hormones | IAA | ABA | Y = 48.3898 + 0.3154X - 0.0004X2 | 0.117 | p = 0.069 |
| CK (ZR + Z) | Y = 116.1967 - 0.103X | 0.647 | p < 0.0001 | ||
| ABA | CK (ZR + Z) | 无显著回归关系 Insignificant regression relation |
表3 翠菊根干重、根构型和根内激素相互之间的回归关系
Table 3 Regression relationships among root mass, root architecture parameters, root hormones for Callistephus chinensis
| 变量间相互关系 Relationship between variables | 响应变量 Dependent variable (Y) | 因变量(X) Independent variable | 回归关系 Regression | R2 | p |
|---|---|---|---|---|---|
| 根生长和根构型关系 Relationships between root growth and root architecture parameters | 根干重 Root dry mass (g) | 1级根长 Length of 1st order roots (mm) | Y = 0.02162 + 0.03696X - 0.00348X2 | 0.693 | p < 0.0001 |
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 0.0801 - 0.01265X | 0.156 | p = 0.017 | ||
| 根生长和激素关系 Relationships between root growth and hormones | 根干重 Root dry mass (g) | IAA | Y = 125.434 + 1.5829X | 0.633 | p < 0.0001 |
| ABA | 关系复杂 Complex relationship | ||||
| CK (ZR + Z) | Y = 110.3072e(-5.9906X) | 0.257 | p < 0.0001 | ||
| 根构型和激素关系 Relationships between root architecture parameters and hormones | IAA (ng·g-1) | 1级根长 Length of 1st order roots (mm) | Y = 0.319895e(0.003427X) | 0.845 | p < 0.0001 |
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 3.2525 - 0.00975X + 1.04299(e-005X2) | 0.515 | p < 0.0001 | ||
| ABA (ng·g-1) | 1级根长 Length of 1st order roots (mm) | 无显著回归关系 Insignificant regression relation | |||
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 2.3737e(-0.0045X) | 0.171 | p = 0.012 | ||
| CK (ZR + Z) ( ng·g-1) | 1级根长 Length of 1st order roots (mm) | Y = 5.2567e(-0.02124X) | 0.527 | p < 0.0001 | |
| 1级侧根间距 Internode length of the 1st order laterals (mm) | 无显著回归关系 Insignificant regression relation | ||||
| 1级侧根密度 Density of 1st order lateral roots (no.·mm-1) | Y = 2.0851 + 0.07907X - 0.000345X2 | 0.402 | p < 0.0001 | ||
| 激素间相互关系 Relationships among hormones | IAA | ABA | Y = 48.3898 + 0.3154X - 0.0004X2 | 0.117 | p = 0.069 |
| CK (ZR + Z) | Y = 116.1967 - 0.103X | 0.647 | p < 0.0001 | ||
| ABA | CK (ZR + Z) | 无显著回归关系 Insignificant regression relation |
| 营养元素 Nutrient element | 化学式 Chemical formula | 浓度 Concentration (mg·L-1) |
|---|---|---|
| 铁盐 Ferric salt | FeSO4·7H2O | 27.850 |
| EDTA·2Na | 37.250 | |
| 微量元素 Micro-nutrient element | H3BO4 | 3.090 |
| MnSO4·H2O | 0.340 | |
| ZnSO4·7H2O | 0.290 | |
| CuSO4·5H2O | 0.075 | |
| KI | 0.830 | |
| Na2Mo4·2H2O | 0.024 | |
| CoCl2·6H2O | 0.048 | |
| 大量元素 Macro-nutrient element | KH2PO4 | 169.880 |
| MgSO4·7H2O | 369.000 | |
| CaCl2·2H2O | 294.000 |
附录I 营养液配方
Appendix I Recipe of the nutrient solution
| 营养元素 Nutrient element | 化学式 Chemical formula | 浓度 Concentration (mg·L-1) |
|---|---|---|
| 铁盐 Ferric salt | FeSO4·7H2O | 27.850 |
| EDTA·2Na | 37.250 | |
| 微量元素 Micro-nutrient element | H3BO4 | 3.090 |
| MnSO4·H2O | 0.340 | |
| ZnSO4·7H2O | 0.290 | |
| CuSO4·5H2O | 0.075 | |
| KI | 0.830 | |
| Na2Mo4·2H2O | 0.024 | |
| CoCl2·6H2O | 0.048 | |
| 大量元素 Macro-nutrient element | KH2PO4 | 169.880 |
| MgSO4·7H2O | 369.000 | |
| CaCl2·2H2O | 294.000 |
| 1 |
Benková E, Hejátko J ( 2009). Hormone interactions at the root apical meristem. Plant Molecular Biology, 69, 383-396.
DOI URL |
| 2 | Blilou I, Xu J, Wildwater M, Willemsen V, Paponov I, Friml J, Heidstra R, Aida M, Palme K, Scheres B ( 2005). The PIN auxin efflux facilitator network controls growth and patterning in Arabidopsis roots. Nature, 433, 39-44. |
| 3 | Bradshaw AD ( 1965). Evolutionary significance of phenotypic plasticity in plants. Advances in Genetics, 13, 115-155. |
| 4 | Brewitz E, Larsson CM, Larsson M ( 1995). Influence of nitrate supply on concentrations and translocation of abscisic acid in barley ( Hordeum vulgare). Physiologia Plantarum, 95, 499-506. |
| 5 | Britto TD, Kronzucker HJ ( 2002). NH4 + toxicity in higher plants: a critical review . Journal of Plant Physiology, 159, 567-584. |
| 6 | Cao Y, Class ADM, Crawford NM ( 1993). Ammonium inhibition of Arabidopsis root growth can be reversed by potassium and by auxin resistance mutations aux1, axr1, and axr2. Plant Physiology, 102, 983-989. |
| 7 | Collier MD, Fotelli MN, Nahm M, Kopriva S, Rennenberg H, Hanke DE, Geßler A ( 2003). Regulation of nitrogen uptake by Fagus sylvatica on a whole plant level- interactions between cytokinins and soluble N compounds. Plant, Cell & Environment, 26, 1549-1560. |
| 8 | de Smet I, Zhang HM, Inzé D, Beeckman T ( 2006). A novel role for abscisic acid emerges from underground. Trends in Plant Science, 11, 434-439. |
| 9 | Drew MC, Saker LR ( 1978). Nutrient supply and the growth of the seminal root system in barley. Journal of Experimental Botany, 29, 435-451. |
| 10 | Drew MC, Saker LR, Ashley TW ( 1973). Nutrient supply and the growth of the seminal root system in barley. Journal of Experimental Botany, 24, 1189-1202. |
| 11 | Editorial Committee for Flora of China of Chinese Academy of Sciences ( 中国科学院中国植物志编辑委员会) ( 1985). The Flora of China (中国植物志), Tomus 74. Science Press, Beijing. 109-110. (in Chinese) |
| 12 | Eghball B, Maranvill JW ( 1993). Root development and nitrogen influx of corn genotypes grown under combined drought and nitrogen stresses. Agronomy Journal, 85, 147-152. |
| 13 | Einsmann JC, Jones RH, Mou P, Mitchell RJ ( 1999). Nutrient foraging traits in 10 co-occurring plant species of contrasting life forms. Journal of Ecology, 87, 609-619. |
| 14 | Eissenstat DM ( 1992). Costs and benefits of constructing roots of small diameter. Journal of Plant Nutrition, 15, 763-782. |
| 15 |
Ettema CH, Wardle DA ( 2002). Spatial soil ecology. Trends in Ecology and Evolution, 17, 177-183.
DOI URL |
| 16 | Farley RA, Fitter AH ( 1999). The responses of seven co-occurring woodland herbaceous perennials to localized nutrient-rich patches. Journal of Ecology, 87, 849-859. |
| 17 | Fitter AH (1994). Architecture and biomass allocation as components of the plastic response of root systems to soil heterogeneity. In: Caldwell MM, Pearcy RW eds. Exploitation of Environmental Heterogeneity by Plants. Academic Press, San Diego, USA. 305-323. |
| 18 | Fitter AH, Stickland TR, Harvey ML, Wilson GW ( 1991). Architectural analysis of plant root systems 1. Architectural correlates of exploitation efficiency. New Phytologist, 118, 375-382. |
| 19 |
Friml J, Vieten A, Sauer M, Weijers D, Schwarz H, Hamann T, Offringa R, Jürgens G ( 2003). Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature, 426, 147-153.
PMID |
| 20 |
Fukaki H, Tasaka M ( 2009). Hormone interactions during lateral root formation. Plant Molecular Biology, 69, 437-449.
DOI URL |
| 21 | He ZP ( 何钟佩 ) ( 1993). Guidance to Experiment on Chemical Control in Crop Plants (农作物化学控制试验指导). Beijing Agricultural University Publishers, Beijing. 60-68. (in Chinese) |
| 22 | Hodge A ( 2004). The plastic plant: root responses to heterogeneous supplies of nutrients. New Phytologist, 162, 9-24. |
| 23 | Hodge A ( 2006). Plastic plants and patchy soils. Journal of Experimental Botany, 57, 401-411. |
| 24 | Jackson RB, Caldwell MM ( 1993). The scale of nutrient heterogeneity around individual plants and its quantification with geostatistics. Ecology, 74, 612-614. |
| 25 | Jackson RB, Manwaring JH, Caldwell MM ( 1990). Rapid physiological adjustment of roots to localized soil enrichment. Nature, 344, 58-60. |
| 26 | Kume T, Sekiya N, Yano K ( 2006). Heterogeneity in spatial P-distribution and foraging capability by Zea mays: effect of patch size and barriers to restrict root proliferation within a patch. Annals of Botany, 98, 1271-1277. |
| 27 |
Kutz A, Müller A, Henning P, Kaiser WM, Piotrowski M, Weiler EW ( 2002). A role for nitrilase 3 in the regulation of root morphology in sulphur-starving Arabidopsis thaliana. The Plant Journal, 30, 95-106.
DOI URL |
| 28 | Laskowski MJ, Williams ME, Nusbaum HC, Sussex IM ( 1995). Formation of lateral root meristems is a two-stage process. Development, 121, 3303-3310. |
| 29 | Leopold LB ( 1971). Trees and streams: the efficiency of branching patterns. Journal of Theoretical Biology, 31, 339-354. |
| 30 | Li X, Mo XR, Shou HX, Wu P ( 2006). Cytokinin-mediated cell cycling arrest of pericycle founder cells in lateral root initiation of Arabidopsis. Plant Cell Physiology, 47, 1112-1123. |
| 31 | López-Bucio J, Cruz-Ramírez A, Herrera-Estrella H ( 2003). The role of nutrient availability in regulating root architecture. Current Opinion in Plant Biology, 6, 280-287. |
| 32 | Lu YL, Xu YC, Shen QR, Dong CX ( 2009). Effects of different nitrogen forms on the growth and cytokinin content in xylem sap of tomato ( Lycopersicon esculentum Mill.) seedlings. Plant and Soil, 315, 67-77. |
| 33 | Lynch J ( 1995). Root architecture and plant productivity. Plant Physiology, 109, 7-13. |
| 34 | Marschner H (1995). Mineral Nutrition of Higher Plants 2nd edn. Academic Press, New York. |
| 35 | Mi GH, Chen FJ, Zhang FS ( 2008). Multiple signaling pathways control nitrogen-mediated root elongation in maize. Plant Signaling and Behavior, 3, 1030-1032. |
| 36 | Mou P, Jones RH, Tan ZQ, Bao Z, Chen HM ( 2012). Morphological and physiological plasticity of plant roots when nutrients are both spatially and temporally heterogeneous. Plant and Soils, DOI:10.1007/S11104-012-1336-y. |
| 37 | Mou P, Michell RJ, Jones RH ( 1997). Root distribution of two tree species under a heterogeneous nutrient environment. Journal of Applied Ecology, 34, 645-656. |
| 38 | Nordström A, Tarkowski P, Tarkowska D, Norbaek R, Åstot C, Dolezal K, Sandberg G ( 2004). Auxin regulation of cytokinin biosynthesis in Arabidopsis thaliana: a factor of potential importance for auxin-cytokinin- regulated development. Proceedings of the National Academy of Sciences of the United States of America, 101, 8039-8044. |
| 39 | Pregitzer KS, Hendrick RL, Fogel R ( 1993). The demography of fine roots in response to patches of water and nitrogen. New Phytologist, 125, 575-580. |
| 40 | Rahayu YS, Pia WL, Neumann G, Römheld V, von Wirén N, Bangerth F ( 2005). Root-derived cytokinins as long-distance signals for NO3 --induced stimulation of leaf growth . Journal of Experimental Botany, 56, 1143-1152. |
| 41 | Razem FA, El-Kereamy A, Abrams SR, Hill RD ( 2006). The RNA-binding protein FCA is an abscisic acid receptor. Nature, 439, 290-294. |
| 42 | Reed RC, Brady SR, Muday GK ( 1998). Inhibition of auxin movement from the shoot into the root inhibits lateral root development in Arabidopsis. Plant Physiology, 118, 1369-1378. |
| 43 | Robinson D ( 1994). The responses of plants to non-uniform supplies of nutrients. New Phytologist, 127, 635-674. |
| 44 | Sánchez-Calderón L, López-Bucio J, Chacón-López L, Cruz-Ramírez A, Nieto-Jacobo F, Dubrovsky JG, Herrera-Estrella L ( 2005). Phosphate starvation induces a determinate developmental program in the roots of Arabidopsis thaliana. Plant Cell Physiology, 46, 174-184. |
| 45 | Scheiner SM (2001). MANOVA: multiple response variables and multispecies interactions. In: Schneiner SM, Gurevitch J eds. Design and Analysis of Ecological Experiments. Oxford University Press, New York. 99-115. |
| 46 | Signora L, de Smet I, Foyer CH, Zhang H ( 2001). ABA plays a central role in mediating the regulatory effects of nitrate on root branching in Arabidopsis. The Plant Journal, 28, 655-662. |
| 47 | Tian QY, Chen FJ, Liu JX, Zhang FS, Mi GH ( 2008). Inhibition of maize root growth by high nitrate supply is correlated with reduced IAA levels in roots. Journal of Plant Physiology, 165, 942-951. |
| 48 | Wang LX, Mou PP, Jones RH ( 2006). Nutrient foraging via physiological and morphological plasticity in three plant species. Canadian Journal of Forest Research, 36, 164-173. |
| 49 | Wang XT, Below FE (1996). Cytokinins in enhanced growth and tillering of wheat induced by mixed nitrogen source. Crop Science, 36, 121-126. |
| 50 | Weiler EW, Jourdan PS, Conrad W ( 1981). Levels of indole-3-acetic acid in intact and decapitated coleoptiles as determined by a specific and highly sensitive solid-phase enzyme immunoassay. Planta, 153, 561-571. |
| 51 | Werner T, Motyka V, Laucou V, Smets R, van Onckelen H, Schmϋlling T ( 2003). Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. The Plant Cell, 15, 2532-2550. |
| 52 | Werner T, Motyka V, Strnad M, Schmϋlling T ( 2001). Regulation of plant growth by cytokinin. Proceedings of the National Academy of Sciences of the United States of America, 98, 10487-10492. |
| 53 | Yan XL ( 严小龙 ) (2007). Principles and Applications of Root Biology (根系生物学原理与应用). Science Press, Beijing. 30-46. (in Chinese) |
| 54 | Yang JC, Zhang JH, Wang ZQ, Zhu QS, Wang W ( 2001). Hormonal changes in the grains of rice subjected to water stress during grain filling. Plant Physiology, 127, 315-323. |
| 55 | Zhang HM, Jennings A, Barlow PW, Forde BG ( 1999). Dual pathways for regulation of root branching by nitrate. Proceedings of the National Academy of Sciences of the United States of America, 96, 6529-6534. |
| [1] | 魏春雪, 杨璐, 汪金松, 杨家明, 史嘉炜, 田大栓, 周青平, 牛书丽. 实验增温对陆地生态系统根系生物量的影响[J]. 植物生态学报, 2021, 45(11): 1203-1212. |
| [2] | 张鑫, 邢亚娟, 闫国永, 王庆贵. 细根对降水变化响应的meta分析[J]. 植物生态学报, 2018, 42(2): 164-172. |
| [3] | 何为, 杨雪英, 肖娟, 张子良, 蒋铮, 袁远爽, 王东, 刘庆, 尹华军. 氮素富集对青藏高原东缘窄叶鲜卑花灌丛根系分泌物碳输入的影响[J]. 植物生态学报, 2017, 41(6): 610-621. |
| [4] | 陈冠陶, 彭勇, 郑军, 李顺, 彭天驰, 邱细容, 涂利华. 氮添加对亚热带次生常绿阔叶林扁刺栲细根生物量、寿命和形态的短期影响[J]. 植物生态学报, 2017, 41(10): 1041-1050. |
| [5] | 胡凤琴, 牟溥. 植物吸收根的增殖和生长与养分变异的关系——臭椿、翠菊、加拿大一枝黄花分根实验的启示[J]. 植物生态学报, 2013, 37(2): 93-103. |
| [6] | 杨秀云, 韩有志, 武小钢. 华北落叶松林细根生物量对土壤水分、氮营养空间异质性改变的响应[J]. 植物生态学报, 2012, 36(9): 965-972. |
| [7] | 庞勇, 李增元. 基于机载激光雷达的小兴安岭温带森林组分生物量反演[J]. 植物生态学报, 2012, 36(10): 1095-1105. |
| [8] | 高艳, 田秋英, 石凤翎, 李凌浩, 张文浩. 黄花苜蓿与蒺藜苜蓿对土壤低磷胁迫适应策略的比较研究[J]. 植物生态学报, 2011, 35(6): 632-640. |
| [9] | 杨青, 张一, 周志春, 马雪红, 刘伟宏, 丰忠平. 异质低磷胁迫下马尾松家系根构型和磷效率的遗传变异[J]. 植物生态学报, 2011, 35(12): 1226-1235. |
| [10] | 杨秀云, 韩有志, 张芸香. 距树干不同距离处华北落叶松人工林细根生物量分布特征及季节变化[J]. 植物生态学报, 2008, 32(6): 1277-1284. |
| [11] | 贾淑霞, 王政权, 梅莉, 孙王月, 全先奎, 史建伟, 于水强, 孙海龙, 谷加存. 施肥对落叶松和水曲柳人工林土壤呼吸的影响[J]. 植物生态学报, 2007, 31(3): 372-379. |
| [12] | 孙志虎, 牟长城. 采用地统计学方法对落叶松人工纯林表层细根生物量的估计[J]. 植物生态学报, 2006, 30(5): 771-779. |
| [13] | 程云环, 韩有志, 王庆成, 王政权. 落叶松人工林细根动态与土壤资源有效性关系研究[J]. 植物生态学报, 2005, 29(3): 403-410. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2022 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
