

植物生态学报 ›› 2023, Vol. 47 ›› Issue (12): 1718-1727.DOI: 10.17521/cjpe.2022.0448
所属专题: 光合作用
刘海燕1, 臧纱纱1,*( ), 张春霞2, 左进城1, 阮祚禧3, 吴红艳1
), 张春霞2, 左进城1, 阮祚禧3, 吴红艳1
收稿日期:2022-11-07
接受日期:2023-06-06
出版日期:2023-12-20
发布日期:2023-12-22
通讯作者:
*(daidai4918@126.com)
基金资助:
LIU Hai-Yan1, ZANG Sha-Sha1,*( ), ZHANG Chun-Xia2, ZUO Jin-Cheng1, RUAN Zuo-Xi3, WU Hong-Yan1
), ZHANG Chun-Xia2, ZUO Jin-Cheng1, RUAN Zuo-Xi3, WU Hong-Yan1
Received:2022-11-07
Accepted:2023-06-06
Online:2023-12-20
Published:2023-12-22
Contact:
*(daidai4918@126.com)
Supported by:摘要:
海洋暖化导致海水上部混合层变浅, 使营养盐限制情况加剧, 也导致硅藻接收到的阳光辐射量增加。高光及营养盐限制的双重胁迫会影响硅藻的光合产量。该研究主要探索硅藻应对磷饥饿及高光胁迫的光合生理调控, 以进一步了解海洋环境变化对硅藻光合作用的影响。将体积不同的假微型海链藻(Thalassiosira pseudonana)和威氏海链藻(T. weissflogii)在磷饥饿条件下培养, 监测其光系统II (PSII)功能变化, 并将其置于高光强下, 研究其光合生理响应。结果表明: 磷饥饿条件下, 体积较小的假微型海链藻PSII活性逐渐下降, 从与D2蛋白结合的质体醌QA-到与D1蛋白结合的质体醌QB的电子传递效率降低, 单位反应中心捕获的用于电子传递的能量下降, 并诱导产生非光化学淬灭, 而体积较大的威氏海链藻则能够维持较长时间的PSII活性; 假微型海链藻在磷充足条件下相比威氏海链藻具有较高的PSII光失活功能截面(σi)值, 易发生光抑制, 但同时也具有高的PSII修复速率, 磷饥饿对其光抑制敏感度无显著影响, 而威氏海链藻在磷饥饿条件下的σi显著升高, 其对高光强的耐受能力显著降低。在营养盐限制及辐射水平增强情况下, 体积较大的威氏海链藻可能倾向于分布在真光层的下部海域。该研究表明海洋环境变化可能会改变不同粒径硅藻的生态位, 并影响其初级生产力。
刘海燕, 臧纱纱, 张春霞, 左进城, 阮祚禧, 吴红艳. 磷饥饿下硅藻光系统II光化学反应及其对高光强的响应. 植物生态学报, 2023, 47(12): 1718-1727. DOI: 10.17521/cjpe.2022.0448
LIU Hai-Yan, ZANG Sha-Sha, ZHANG Chun-Xia, ZUO Jin-Cheng, RUAN Zuo-Xi, WU Hong-Yan. Photochemical reaction of photosystem II in diatoms under phosphorus starvation and its response to high light intensity. Chinese Journal of Plant Ecology, 2023, 47(12): 1718-1727. DOI: 10.17521/cjpe.2022.0448
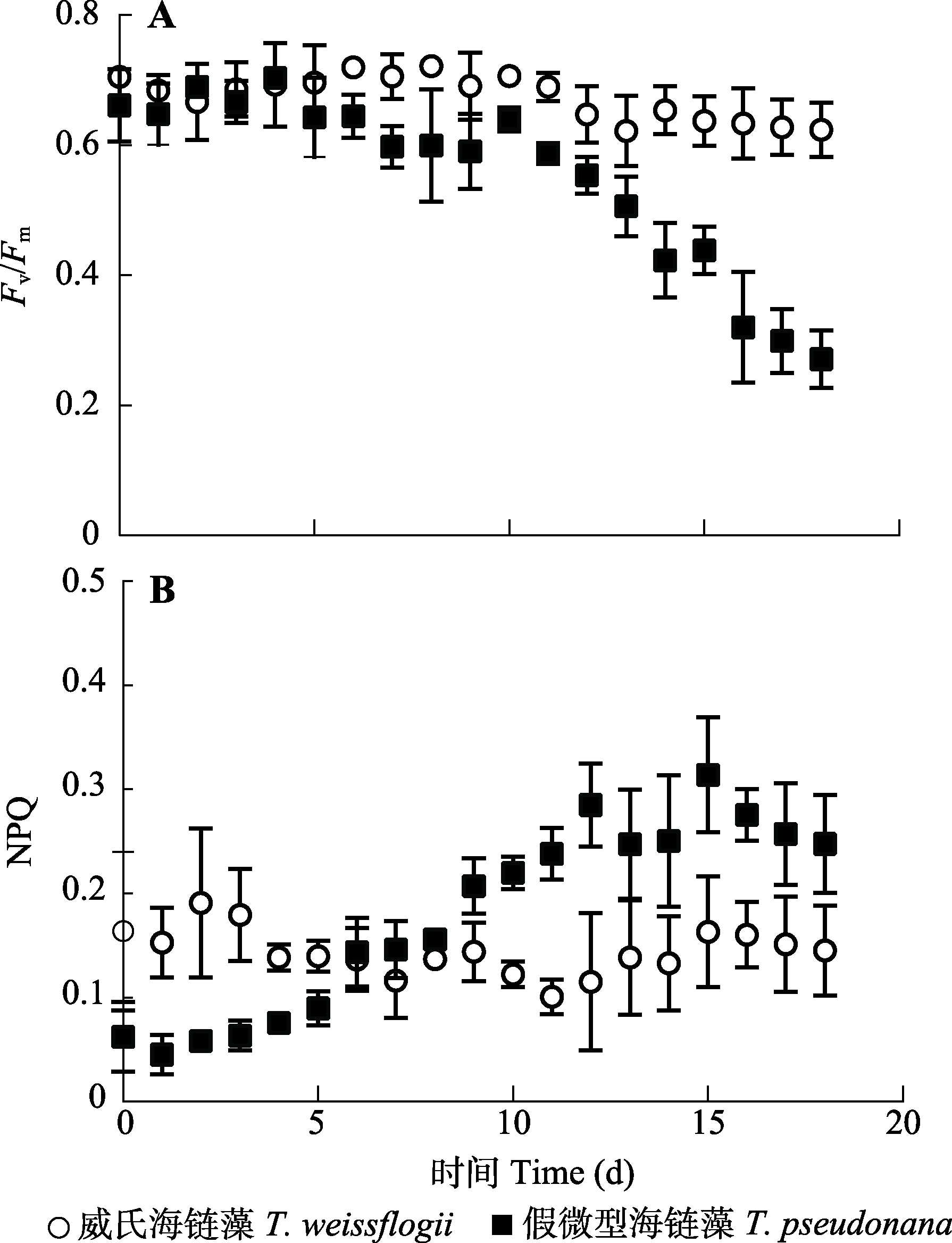
图1 磷饥饿条件下威氏海链藻和假微型海链藻光系统II最大光化学效率(Fv/Fm) (A)及非光化学淬灭(NPQ) (B)的变化(平均值±标准差)。
Fig. 1 Changes of PSII maximum photochemical efficiency (Fv/Fm) (A) and non-photochemical quenching (NPQ) (B) in Thalassiosira weissflogii and T. pseudonana cultures under phosphorus starvation (mean ± SD).
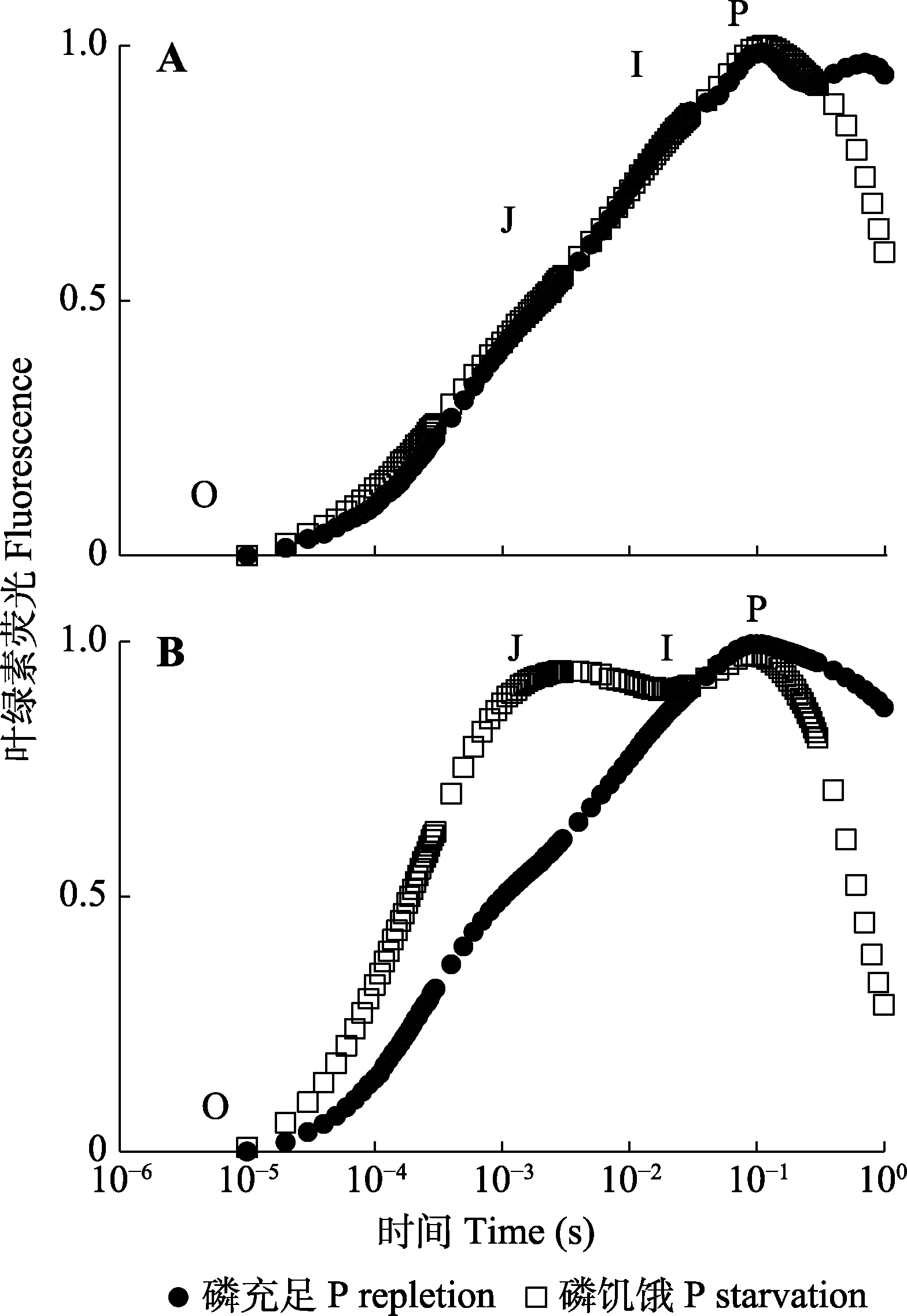
图2 磷充足和磷饥饿条件下威氏海链藻(A)和假微型海链藻(B)的叶绿素荧光诱导曲线(OJIP)。
Fig. 2 Chlorophyll a fluorescence transients (OJIP) in Thalassiosira weissflogii (A) and T. pseudonana (B) grown under phosphorus (P) repletion or starvation conditions.
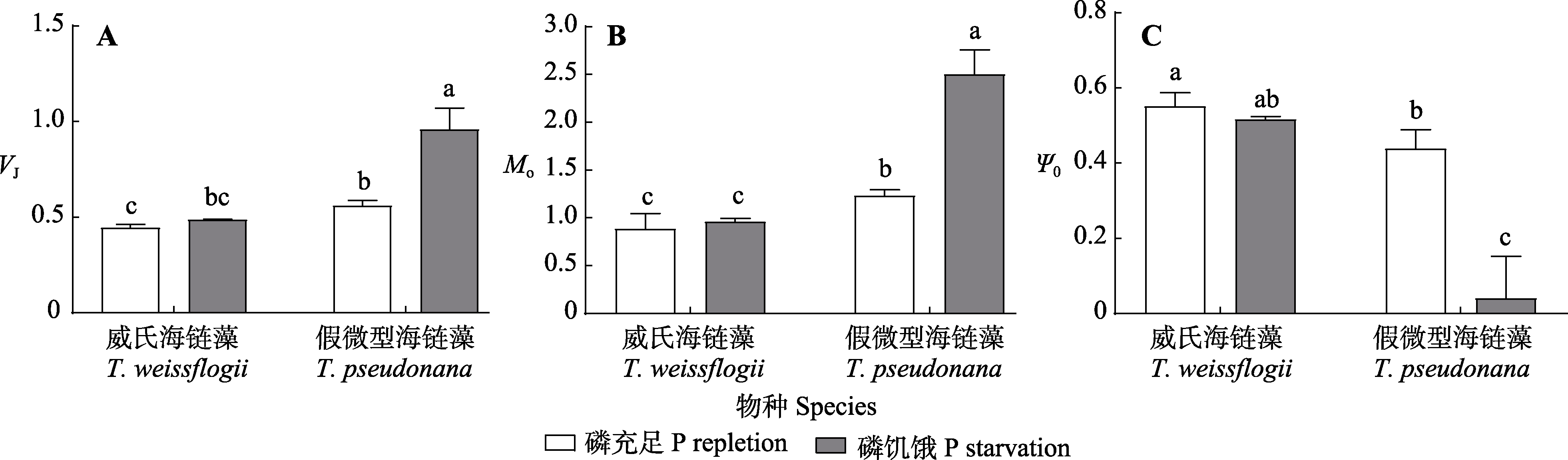
图3 磷充足和磷饥饿条件下威氏海链藻和假微型海链藻的J点的相对可变荧光(VJ)、瞬时荧光开始时的初始斜率(Mo)和QA-到QB的电子传递效率(Ψ0) (平均值±标准差)。不同小写字母表示不同物种、处理之间的差异显著(p < 0.05)。
Fig. 3 Relatively variable fluorescence at J-point (VJ), initial slope at the beginning of instantaneous fluorescence (Mo) and QA- electron transfer efficiency to QB (Ψ0) in Thalassiosira weissflogii and T. pseudonana cultures grown under phosphorus (P) repletion or starvation conditions (mean ± SD). Different lowercase letters indicate differences between different species and treatments (p < 0.05).
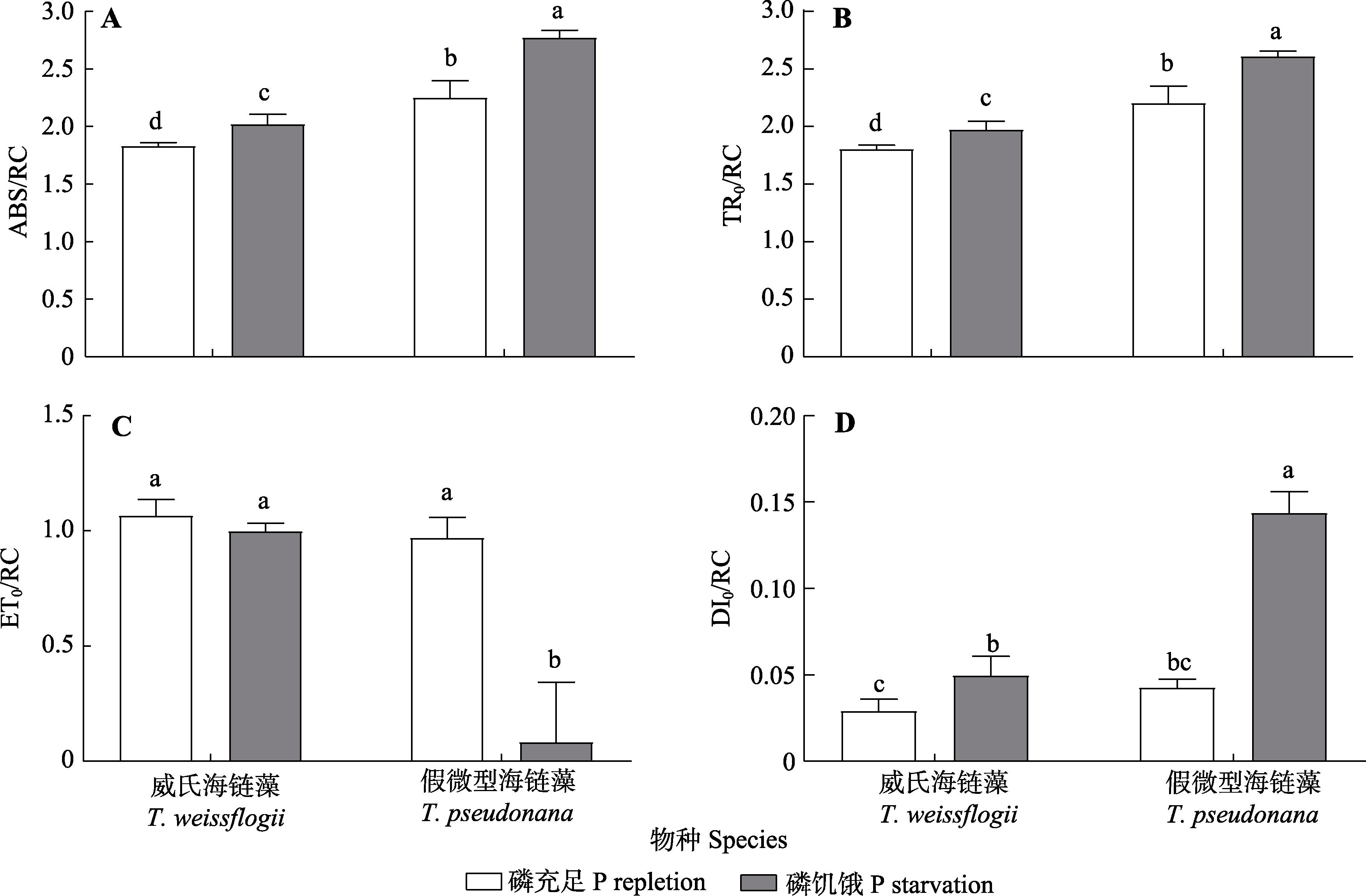
图4 磷充足和磷饥饿条件下威氏海链藻和假微型海链藻的单位反应中心吸收的光能(ABS/RC)、单位反应中心耗散掉的能量(DI0/RC)、单位反应中心捕获的用于还原QA的能量(TR0/RC)和单位反应中心捕获的用于电子传递的能量(ET0/RC)变化(平均值±标准差)。不同小写字母表示不同物种、处理之间差异显著(p < 0.05)。
Fig. 4 Changes of the energy absorbed per unit reaction center (ABS/RC), energy dissipated from the reaction center (TR0/RC), energy captured by the unit reaction center for reducing QA (ET0/RC) and energy captured by the reaction center for electron transport (DI0/RC) in Thalassiosira weissflogii and T. pseudonana grown under phosphorus (P) repletion or starvation conditions (mean ± SD). Different lowercase letters indicate significant differences between species and treatments (p < 0.05).
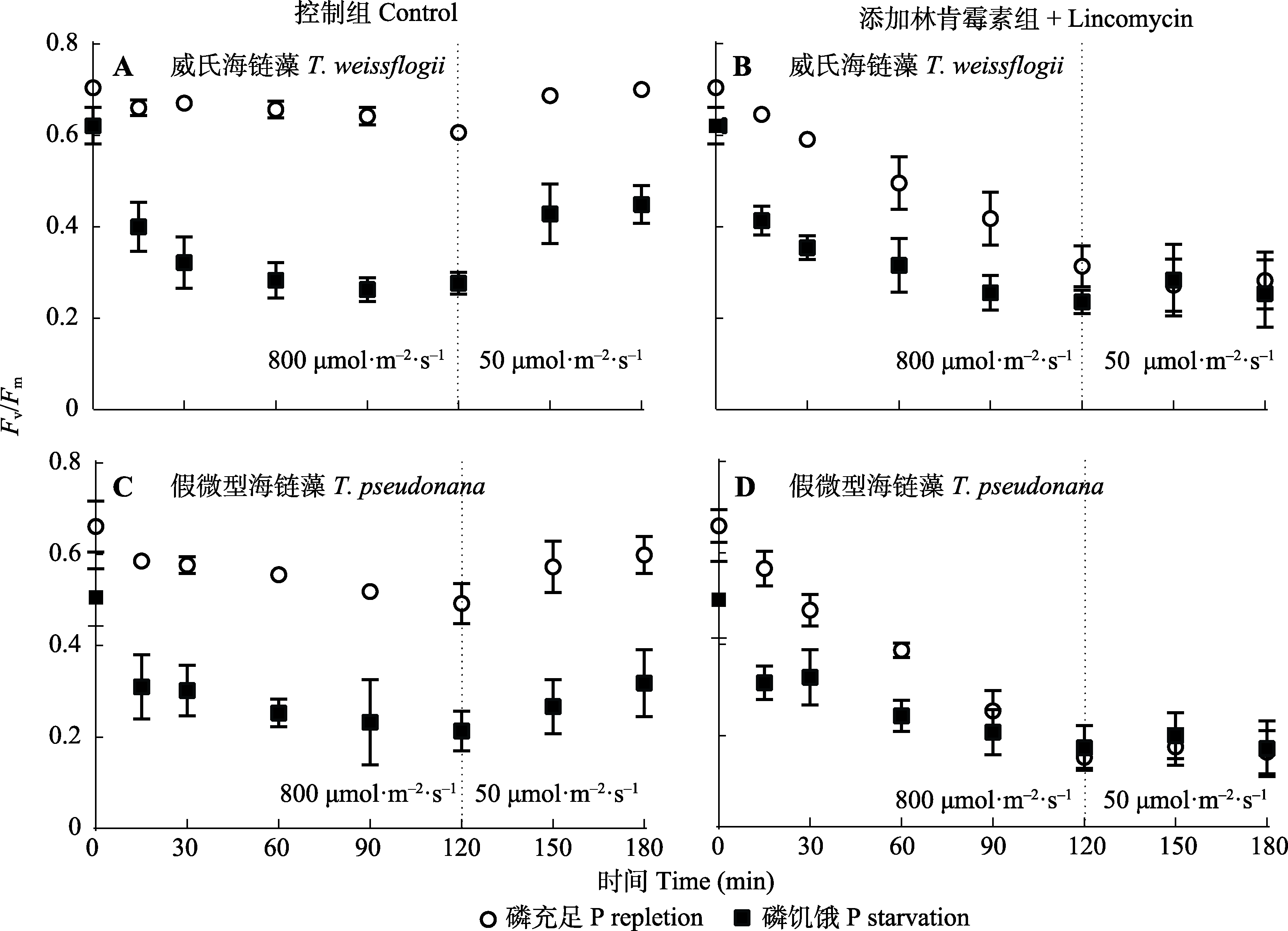
图5 磷充足及磷饥饿条件下威氏海链藻和假微型海链藻响应高光强的光系统II (PSII)最大光化学效率(Fv/Fm)变化(平均值±标准差)。
Fig. 5 Response of photosystem II (PSII) maximum photochemical efficiency (Fv/Fm) to high light in Thalassiosira weissflogii and T. pseudonana grown under phosphorus (P) repletion or starvation conditions (mean ± SD).
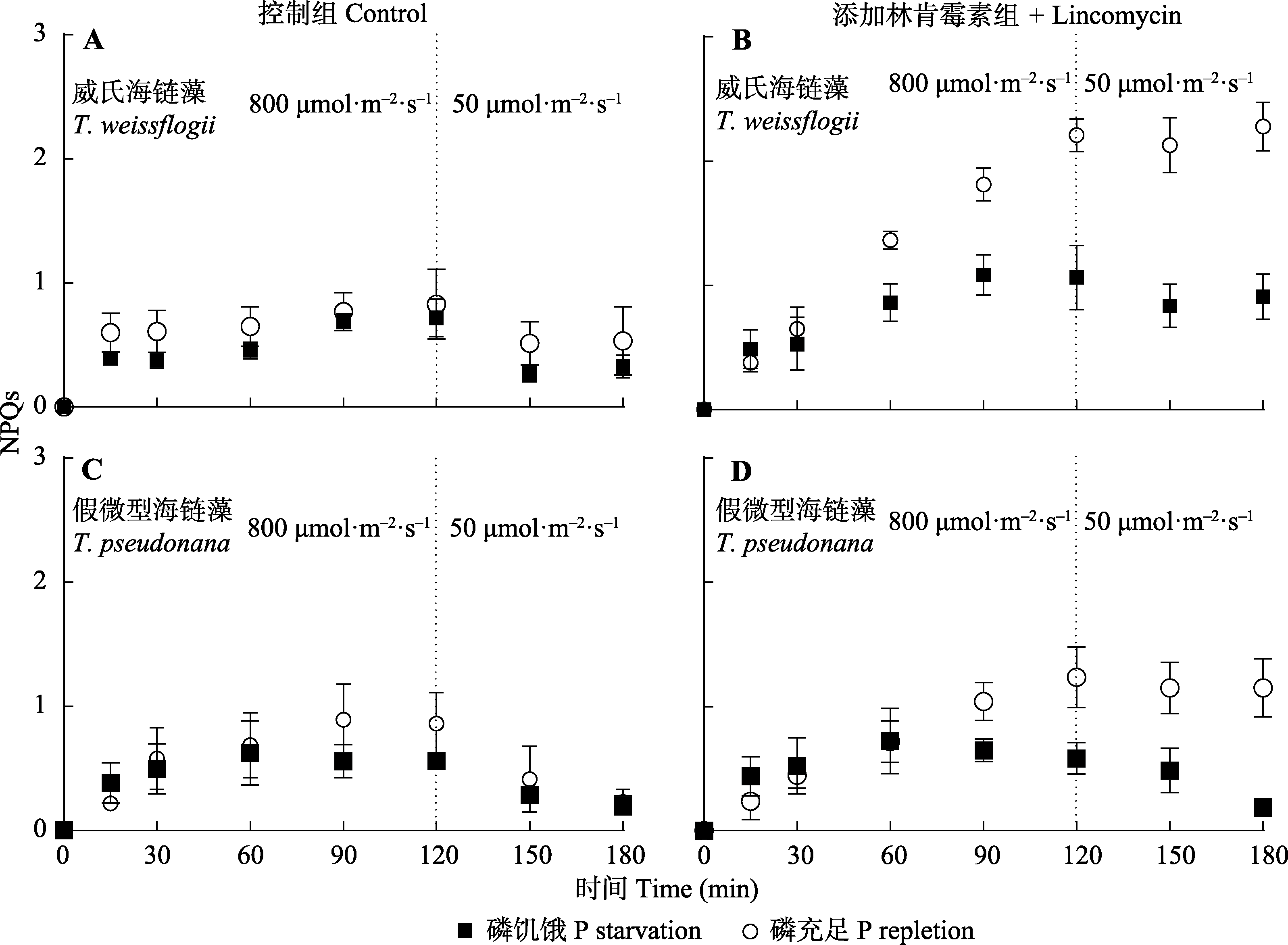
图6 磷充足及磷饥饿条件下威氏海链藻和假微型海链藻响应高光强的非光化学淬灭(NPQs)变化(平均值±标准差)。
Fig. 6 Response of non-photochemical quenching (NPQs) to high light in Thalassiosira weissflogii and T. pseudonana grown under phosphorus (P) repletion or starvation (mean ± SD).
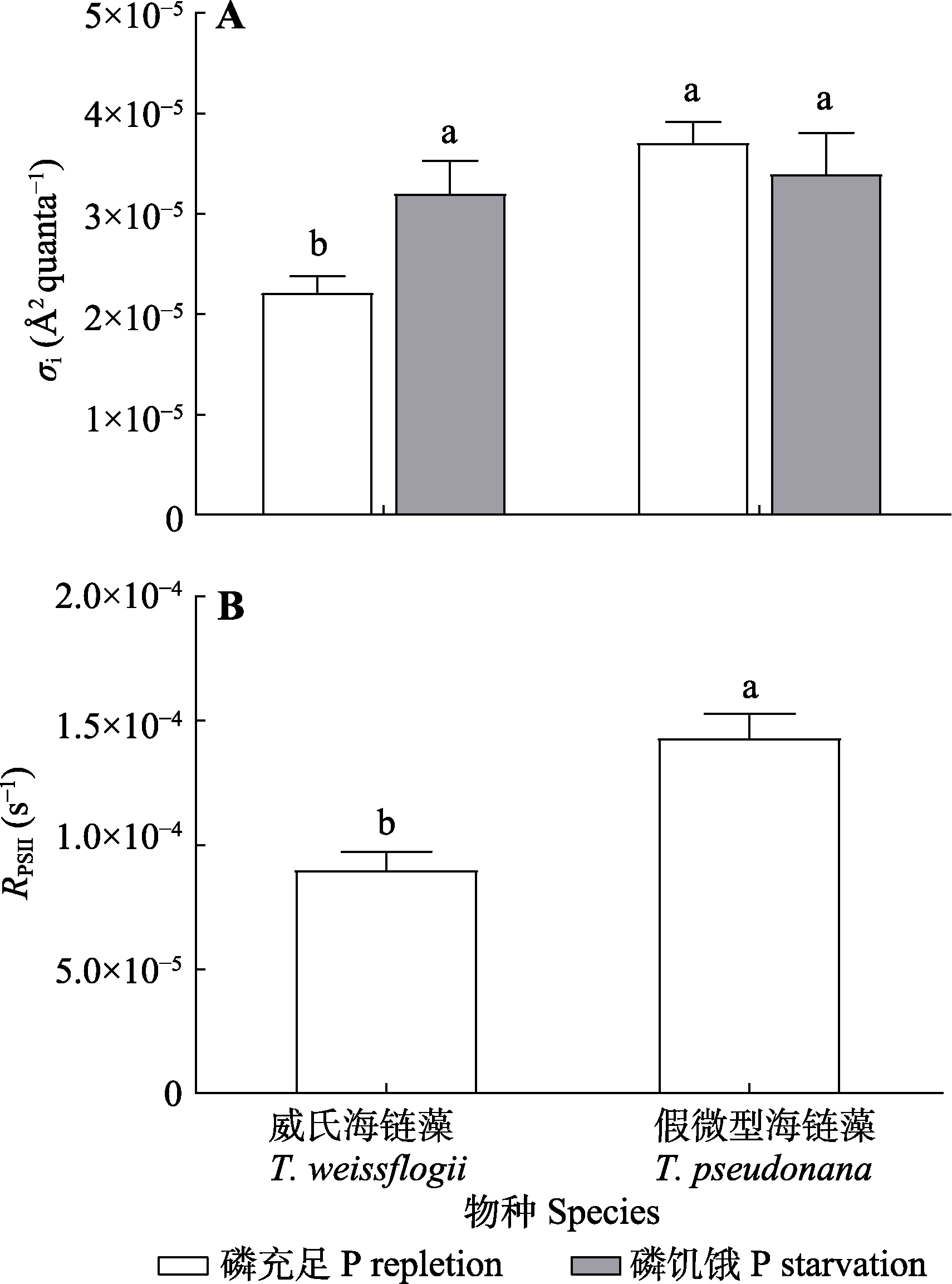
图7 磷充足和磷饥饿条件下, 威氏海链藻和假微型海链藻的PSII光失活功能截面(σi)和光系统II (PSII)修复速率(RPSII)的变化(平均值±标准差)。不同小写字母表示不同物种、处理之间差异显著(p < 0.05)。
Fig. 7 Changes of PSII photo deactivation function cross section (σi) and photosystem II (PSII) repair rate (RPSII) in Thalassiosira weissflogii and T. pseudonana grown under phosphorus (P) repletion or starvation conditions (mean ± SD). Different lowercase letters indicate significant differences between species and treatments (p < 0.05).
| [1] |
Armbrust EV (2009). The life of diatoms in the world’s oceans. Nature, 459, 185-192.
DOI |
| [2] |
Arsenieff L, Le Gall F, Rigaut-Jalabert F, Mahé F, Sarno D, Gouhier L, Baudoux AC, Simon N (2020). Diversity and dynamics of relevant nanoplanktonic diatoms in the Western English Channel. The ISME Journal, 14, 1966-1981.
DOI URL |
| [3] |
Claassens NJ, Scarinci G, Fischer A, Flamholz AI, Newell W, Frielingsdorf S, Lenz O, Bar-Even A (2020). Phosphoglycolate salvage in a chemolithoautotroph using the Calvin cycle. Proceedings of the National Academy of Sciences of the United States of America, 117, 22452-22461.
DOI PMID |
| [4] |
Diaz J, Ingall E, Benitez-Nelson C, Paterson D, de Jonge MD, McNulty I, Brandes JA (2008). Marine polyphosphate: a key player in geologic phosphorus sequestration. Science, 320, 652-655.
DOI PMID |
| [5] | Dyhrman ST, Jenkins BD, Rynearson TA, Saito MA, Mercier ML, Alexander H, Whitney LP, Drzewianowski A, Bulygin VV, Bertrand EM, Wu Z, Benitez-Nelson C, Heithoff A (2012). The transcriptome and proteome of the diatom Thalassiosira pseudonana reveal a diverse phosphorus stress response. PLoS ONE, 7, e33768. DOI: 10.1371/journal.pone.0033768. |
| [6] |
Falkowski PG, Oliver MJ (2007). Mix and match: How climate selects phytoplankton. Nature Reviews Microbiology, 5, 813-819.
PMID |
| [7] | Feng T, Yang Z, Zheng J, Xie Y, Li D, Murugan SB, Yang W, Liu J, Li H (2015). Examination of metabolic responses to phosphorus limitation via proteomic analyses in the marine diatom Phaeodactylum tricornutum. Scientific Reports, 5, 10373. DOI: 10.1038/srep10373. |
| [8] |
Finkel ZV, Beardall J, Flynn KJ, Quigg A, Rees TAV, Raven JA (2010). Phytoplankton in a changing world: cell size and elemental stoichiometry. Journal of Plankton Research, 32, 119-137.
DOI URL |
| [9] | Ge W, Wang F, Chai C (2012). Effect of nitrogen and phosphorus concentration on interspecific competition between Skeletonema costatum and Scrippsiella trochoidea. Chinese Journal of Plant Ecology, 36, 697-704. |
|
[ 葛蔚, 汪芳, 柴超 (2012). 氮和磷浓度对中肋骨条藻和锥状斯氏藻种间竞争的影响. 植物生态学报, 36, 697-704.]
DOI |
|
| [10] | Giovagnetti V, Flori S, Tramontano F, Lavaud J, Brunet C (2014). The velocity of light intensity increase modulates the photoprotective response in coastal diatoms. PLoS ONE, 9, e103782. DOI: 10.1371/journal.pone.0103782. |
| [11] | Hao JC, Liu HF, Zhou W, Shi LJ, Li SP, Lian B (2018). Interaction mechanism between growth of Microcystis aeruginosa and sorption-desorption of phosphorus on ferric oxide. Earth and Environment, 46, 505-512. |
| [ 郝建朝, 刘惠芬, 周炜, 石利军, 李少朋, 连宾 (2018). 铜绿微囊藻生长与铁氧化物吸附解吸磷的相互作用机制研究. 地球与环境, 46, 505-512.] | |
| [12] | Hou JJ, Huang BQ, Cao ZR, Chen JX, Hong HS (2007). Effects of nutrient limitation on pigments in Thalassiosira weissflogii and Prorocentrum donghaiense. Journal of Integrative Plant Biology, 49, 686-697. |
| [13] | Jiang MQ, Nakano SI (2022). The crucial influence of trophic status on the relative requirement of nitrogen to phosphorus for phytoplankton growth. Water Research, 222, 118868. DOI: 10.1016/j.watres.2022.118868. |
| [14] |
Key T, McCarthy A, Campbell DA, Six C, Roy S, Finkel ZV (2010). Cell size trade-offs govern light exploitation strategies in marine phytoplankton. Environmental Microbiology, 12, 95-104.
DOI PMID |
| [15] |
Krause GH, Weis E (1991). Chlorophyll fluorescence and photosynthesis: the basics. Annual Review of Plant Physiology and Plant Molecular Biology, 42, 313-349.
DOI URL |
| [16] | Levitus S, Antonov JI, Boyer TP, Locarnini RA, Garcia HE, Mishonov AV (2009). Global Ocean heat content 1955-2008 in light of recently revealed instrumentation problems. Geophysical Research Letters, 36, L07608. DOI: 10.1029/2008GL037155. |
| [17] | Li MZ, Li L, Shi XG, Lin LX, Lin SJ (2015). Effects of phosphorus deficiency and adenosine 5’-triphosphate (ATP) on growth and cell cycle of the dinoflagellate Prorocentrum donghaiense. Harmful Algae, 47, 35-41. |
| [18] | Li X, Zhou YP, Xia QQ, Dai RH (2020). The impacts of phosphorus stress on the growth and metabolism of algae and its response mechanism. Environmental Chemistry, 39, 2074-2083. |
| [ 李璇, 周燕平, 夏琼琼, 代瑞华 (2020). 磷胁迫对藻类生长代谢的影响及藻类对胁迫响应机制的研究进展. 环境化学, 39, 2074-2083.] | |
| [19] |
Li Y, Zhao QL, Lü SH (2013). The genus Thalassiosira off the Guangdong coast, South China Sea. Botanica Marina, 56, 83-110.
DOI URL |
| [20] |
Li Y, Zhao QL, Lü SH (2014). Taxonomy and species diversity of the diatom genus Thalassiosira (Bacillariophyceae) in Zhejiang coastal waters, the East China Sea. Nova Hedwigia, 99, 373-402.
DOI URL |
| [21] |
Marañón E, Cermeño P, López-Sandoval DC, Rodríguez- Ramos T, Sobrino C, Huete-Ortega M, Blanco JM, Rodríguez J (2013). Unimodal size scaling of phytoplankton growth and the size dependence of nutrient uptake and use. Ecology Letters, 16, 371-379.
DOI PMID |
| [22] |
Martin P, Dyhrman ST, Lomas MW, Poulton NJ,van Mooy BAS (2014). Accumulation and enhanced cycling of polyphosphate by Sargasso Sea plankton in response to low phosphorus. Proceedings of the National Academy of Sciences of the United States of America, 111, 8089-8094.
DOI PMID |
| [23] | Martin P, van Mooy BAS, Heithoff A, Dyhrman ST (2011). Phosphorus supply drives rapid turnover of membrane phospholipids in the diatom Thalassiosira pseudonana. The ISME Journal, 5, 1057-1060. |
| [24] | Murata N, Takahashi S, Nishiyama Y, Allakhverdiev SI (2007). Photoinhibition of photosystem II under environmental stress. Biochimica et Biophysica Acta (BBA): Bioenergetics, 1767, 414-421. |
| [25] |
Petrou K, Kranz SA, Doblin MA, Ralph PJ (2012). Photophysiological responses of Fragilariopsis cylindrus (Bacillariophyceae) to nitrogen depletion at two temperatures. Journal of Phycology, 48, 127-136.
DOI URL |
| [26] | Qiao HJ, Zang SS, Yan F, Xu ZG, Wang L, Wu HY (2021). Physiological responses of the diatoms Thalassiosira weissflogii and Thalassiosira pseudonana to nitrogen starvation and high light. Marine Environmental Research, 166, 105276. DOI: 10.1016/j.marenvres.2021.105276. |
| [27] | Raven JA (2012). Protein turnover and plant RNA and phosphorus requirements in relation to nitrogen fixation. Plant Science, 188- 189, 25-35. |
| [28] | Roberts S, Shelly K, Beardall J (2008). Interactions among phosphate uptake, photosynthesis, and chlorophyll fluorescence in nutrient-limited cultures of the chlorophyte microalga Dunaliella tertiolecta. Journal of Phycology, 44, 662-669. |
| [29] |
Toggweiler JR (1999). Oceanography: an ultimate limiting nutrient. Nature, 400, 511-512.
DOI |
| [30] |
Tyrrell T (1999). The relative influences of nitrogen and phosphorus on oceanic primary production. Nature, 400, 525-531.
DOI |
| [31] |
van de Poll WH, Kulk G, Timmermans KR, Brussaard CPD, van der Woerd HJ, Kehoe MJ, Mojica KDA, Visser RJW, Rozema PD, Buma AGJ (2013). Phytoplankton chlorophyll a biomass, composition, and productivity along a temperature and stratification gradient in the northeast Atlantic Ocean. Biogeosciences, 10, 4227-4240.
DOI URL |
| [32] |
Wagner H, Jakob T, Lavaud J, Wilhelm C (2016). Photosystem II cycle activity and alternative electron transport in the diatom Phaeodactylum tricornutum under dynamic light conditions and nitrogen limitation. Photosynthesis Research, 128, 151-161.
DOI PMID |
| [33] |
Wang YZ, Yu J, Wang P, Deng SW, Chang JH, Ran ZX (2018). Response of energy microalgae Chlamydomonas reinhardtii to nitrogen and phosphorus stress. Environmental Science and Pollution Research, 25, 5762-5770.
DOI URL |
| [34] |
Wu HY, Roy S, Alami M, Green BR, Campbell DA (2012). Photosystem II photoinactivation, repair and protection in marine centric diatoms. Plant Physiology, 160, 464-476.
DOI URL |
| [35] | Xing YF, Guo L, Wang Y, Zhao YG, Jin CJ, Gao MC, Ji JY, She ZL (2021). An insight into the phosphorus distribution in extracellular and intracellular cell of Chlorella vulgaris under mixotrophic cultivation. Algal Research, 60, 102482. DOI: 10.1016/j.algal.2021.102482. |
| [36] |
Ye YT, Shi DL (2020). Effects of global change on key processes of primary production in marine ecosystems. Chinese Journal of Plant Ecology, 44, 575-582.
DOI URL |
|
[ 叶幼亭, 史大林 (2020). 全球变化对海洋生态系统初级生产关键过程的影响. 植物生态学报, 44, 575-582.]
DOI |
|
| [37] | Zhou FS, Dong C, Wei F, Guo YS, Wang R (2021). Research progresses on the relationship between PsbS subunits and non-photochemical quenching in plant photosystem II. Molecular Plant Breeding, 19, 1-11. |
| [ 周繁树, 董臣, 位芳, 郭玉双, 王燃 (2021). 植物光系统II中PsbS亚基在非光化学猝灭反应中的研究进展. 分子植物育种, 19, 1-11.] |
| [1] | 李伟斌, 张红霞, 张玉书, 陈妮娜. 昼夜不对称增温对长白山阔叶红松林碳汇能力的影响[J]. 植物生态学报, 2023, 47(9): 1225-1233. |
| [2] | 蒋海港, 曾云鸿, 唐华欣, 刘伟, 李杰林, 何国华, 秦海燕, 王丽超, 姚银安. 三种藓类植物固碳耗水节律调节作用[J]. 植物生态学报, 2023, 47(7): 988-997. |
| [3] | 何敏, 许秋月, 夏允, 杨柳明, 范跃新, 杨玉盛. 植物磷获取机制及其对全球变化的响应[J]. 植物生态学报, 2023, 47(3): 291-305. |
| [4] | 吴霖升, 张永光, 章钊颖, 张小康, 吴云飞. 日光诱导叶绿素荧光遥感及其在陆地生态系统监测中的应用[J]. 植物生态学报, 2022, 46(10): 1167-1199. |
| [5] | 靳川, 李鑫豪, 蒋燕, 徐铭泽, 田赟, 刘鹏, 贾昕, 查天山. 黑沙蒿光合能量分配组分在生长季的相对变化与调控机制[J]. 植物生态学报, 2021, 45(8): 870-879. |
| [6] | 武洪敏, 双升普, 张金燕, 寸竹, 孟珍贵, 李龙根, 沙本才, 陈军文. 短期生长环境光强骤增导致典型阴生植物三七光系统受损的机制[J]. 植物生态学报, 2021, 45(4): 404-419. |
| [7] | 叶子飘, 于冯, 安婷, 王复标, 康华靖. 植物气孔导度对CO2响应模型的构建[J]. 植物生态学报, 2021, 45(4): 420-428. |
| [8] | 李景, 王欣, 王振华, 王斌, 王成章, 邓美凤, 刘玲莉. 臭氧和气溶胶复合污染对杨树叶片光合作用的影响[J]. 植物生态学报, 2020, 44(8): 854-863. |
| [9] | 李旭, 吴婷, 程严, 谭钠丹, 蒋芬, 刘世忠, 褚国伟, 孟泽, 刘菊秀. 南亚热带常绿阔叶林4个树种对增温的生理生态适应能力比较[J]. 植物生态学报, 2020, 44(12): 1203-1214. |
| [10] | 刘校铭, 杨晓芳, 王璇, 张守仁. 暖温带落叶阔叶林辽东栎和五角枫生长和光合生理生态特征对模拟氮沉降的响应[J]. 植物生态学报, 2019, 43(3): 197-207. |
| [11] | 李鑫豪, 闫慧娟, 卫腾宙, 周文君, 贾昕, 查天山. 油蒿资源利用效率在生长季的相对变化及对环境因子的响应[J]. 植物生态学报, 2019, 43(10): 889-898. |
| [12] | 张娜, 朱阳春, 李志强, 卢信, 范如芹, 刘丽珠, 童非, 陈静, 穆春生, 张振华. 淹水和干旱生境下铅对芦苇生长、生物量分配和光合作用的影响[J]. 植物生态学报, 2018, 42(2): 229-239. |
| [13] | 韩吉梅, 张旺锋, 熊栋梁, 张亚黎. 植物光合作用叶肉导度及主要限制因素研究进展[J]. 植物生态学报, 2017, 41(8): 914-924. |
| [14] | 蔡建国, 韦孟琪, 章毅, 魏云龙. 遮阴对绣球光合特性和叶绿素荧光参数的影响[J]. 植物生态学报, 2017, 41(5): 570-576. |
| [15] | 陈良华, 赖娟, 胡相伟, 杨万勤, 张健, 王小军, 谭灵杰. 接种丛枝菌根真菌对受镉胁迫美洲黑杨雌、雄株光合生理的影响[J]. 植物生态学报, 2017, 41(4): 480-488. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2026 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
![]()