

Chin J Plant Ecol ›› 2019, Vol. 43 ›› Issue (8): 718-728.DOI: 10.17521/cjpe.2019.0036
Special Issue: 微生物生态学
• Research Articles • Previous Articles
CUI Li1,2,3,GUO Feng1,2,3,ZHANG Jia-Lei1,2,3,YANG Sha1,2,3,WANG Jian-Guo1,2,3,MENG Jing-Jing1,2,3,GENG Yun1,2,3,LI Xin-Guo1,2,3,*( ),WAN Shu-Bo2,3,4,*(
),WAN Shu-Bo2,3,4,*( )
)
Received:2019-02-21
Revised:2019-08-05
Online:2019-08-20
Published:2020-01-03
Contact:
LI Xin-Guo ORCID:0000-0003-3277-9808,WAN Shu-Bo
Supported by:CUI Li, GUO Feng, ZHANG Jia-Lei, YANG Sha, WANG Jian-Guo, MENG Jing-Jing, GENG Yun, LI Xin-Guo, WAN Shu-Bo. Improvement of continuous microbial environment in peanut rhizosphere soil by Funneliformis mosseae[J]. Chin J Plant Ecol, 2019, 43(8): 718-728.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.17521/cjpe.2019.0036
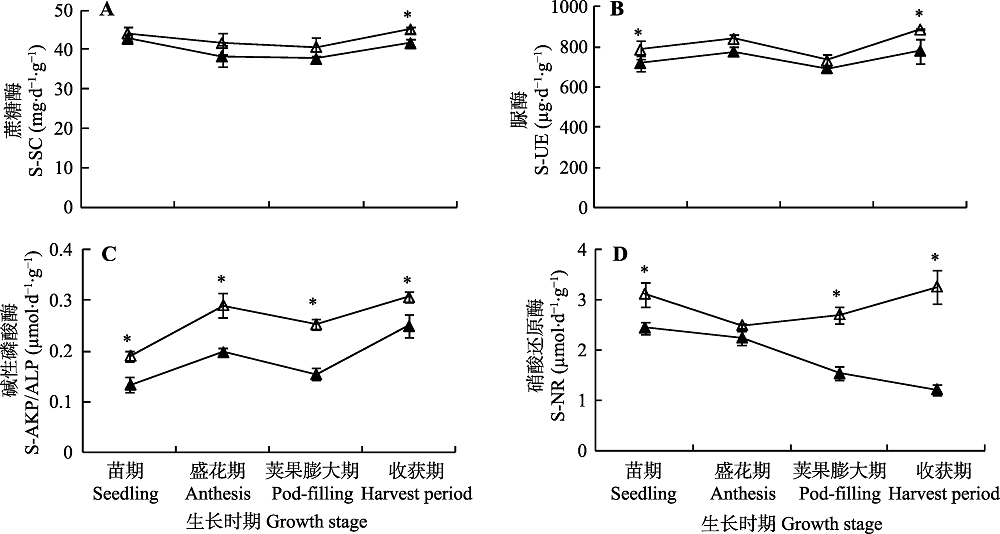
Fig. 1 Effects of inoculation with Funneliformis mosseae on enzyme activities of solid-sucrase (S-SC)(A), solid-urease (S-UE)(B), solid alkaline-phosphatase (S-AKP/ALP)(C) and solid-nitrate reductase (S-NR)(D) in rhizosphere soil of continuing cropping peanut at different growth stages (mean ± SE). * indicates significant difference between the two treatments (p < 0.05). ▲, without F. mosseae inoculation; △, F. mosseae inoculation.
| 处理 Treatment | 全氮 Total N (g·kg-1) | 全磷 Total P (g·kg-1) | 全钾 Total K (mg·g-1) | |||
|---|---|---|---|---|---|---|
| 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | |
| -AMF | 1.23 ± 0.06 | 0.74 ± 0.02 | 0.59 ± 0.06 | 0.37 ± 0.04 | 6.67 ± 0.22 | 3.57 ± 0.67 |
| +AMF | 1.44 ± 0.06* | 0.82 ± 0.03* | 0.70 ± 0.03* | 0.47 ± 0.07* | 7.41 ± 0.57 | 5.39 ± 0.73* |
Table 1 Comparison of soil contents of total nitrogen, total phosphorus and total potassium between the treatments with and without Funneliformis mosseae inoculation under continuing cropping of peanuts (mean ± SE, n = 3)
| 处理 Treatment | 全氮 Total N (g·kg-1) | 全磷 Total P (g·kg-1) | 全钾 Total K (mg·g-1) | |||
|---|---|---|---|---|---|---|
| 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | |
| -AMF | 1.23 ± 0.06 | 0.74 ± 0.02 | 0.59 ± 0.06 | 0.37 ± 0.04 | 6.67 ± 0.22 | 3.57 ± 0.67 |
| +AMF | 1.44 ± 0.06* | 0.82 ± 0.03* | 0.70 ± 0.03* | 0.47 ± 0.07* | 7.41 ± 0.57 | 5.39 ± 0.73* |
| 处理 Treatment | 碱解氮 Alkaline N (mg·kg-1) | 速效磷 Effective P (g·kg-1) | 速效钾 Effective K (mg·kg-1) | |||
|---|---|---|---|---|---|---|
| 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | |
| -AMF | 73.39 ± 3.66 | 75.77 ± 3.66 | 0.18 ± 0.01 | 0.15 ± 0.01 | 15.40 ± 0.56 | 11.03 ± 0.64 |
| +AMF | 74.86 ± 5.54 | 81.67 ± 7.00 | 0.21 ± 0.02* | 0.18 ± 0.00* | 16.17 ± 0.47 | 13.20 ± 0.66* |
Table 2 Effects of Funneliformis mosseae inoculation on the soil contents of alkaline nitrogen, available phosphorus and available potassium (mean ± SE, n = 3)
| 处理 Treatment | 碱解氮 Alkaline N (mg·kg-1) | 速效磷 Effective P (g·kg-1) | 速效钾 Effective K (mg·kg-1) | |||
|---|---|---|---|---|---|---|
| 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | 盛花期 Anthesis | 收获期 Harvest period | |
| -AMF | 73.39 ± 3.66 | 75.77 ± 3.66 | 0.18 ± 0.01 | 0.15 ± 0.01 | 15.40 ± 0.56 | 11.03 ± 0.64 |
| +AMF | 74.86 ± 5.54 | 81.67 ± 7.00 | 0.21 ± 0.02* | 0.18 ± 0.00* | 16.17 ± 0.47 | 13.20 ± 0.66* |
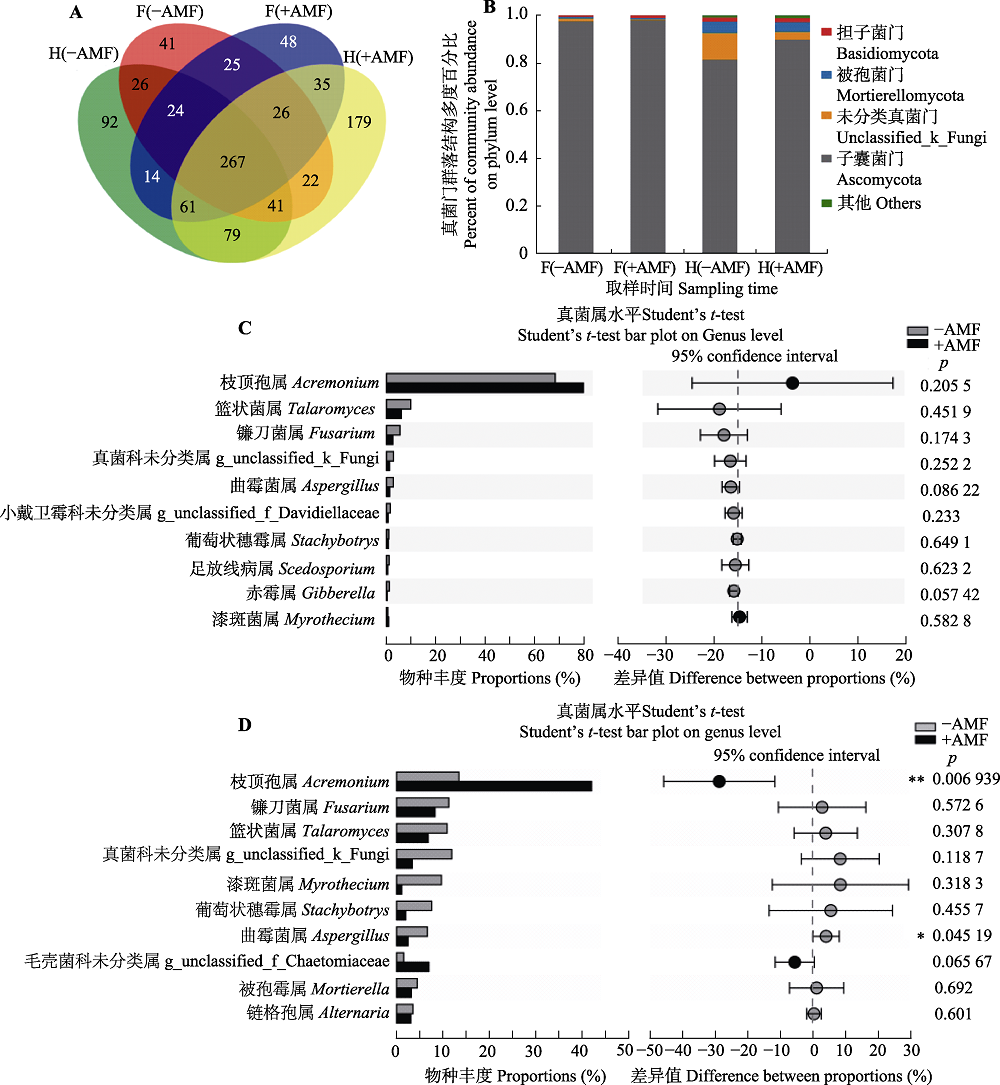
Fig. 2 Effects of with and without Funneliformis mosseae inoculation on the structure and abundance of soil fungi community under continuing cropping of peanut. A, The Venn figure shows the number of fungal operational taxonomic units in different treatments. B, Abundance of soil fungi in continuing cropping of peanut was changed by F. mosseae. C, Abundance of fungal genera were not significantly different between with and without F. mosseae inoculation in the flowering period of continuing cropping peanuts (mean ± SE, n = 3). D, Abundance of some fungal genera were significantly different between with and without F. mosseae inoculation at harvest period (mean ± SE, n = 3). -AMF, without F. mosseae inoculation; +AMF, F. mosseae inoculation. F(-AMF), peanut rhizosphere soil without F. mosseae inoculation during the flowering period; F(+AMF), rhizosphere soil of peanut with F. mosseae inoculation; H(-AMF), peanut rhizosphere soil without F. mosseae inoculation during the harvest period; H(+AMF), peanut rhizosphere soil with F. mosseae inoculation during the harvest period. *, p < 0.05; **, p < 0.01.
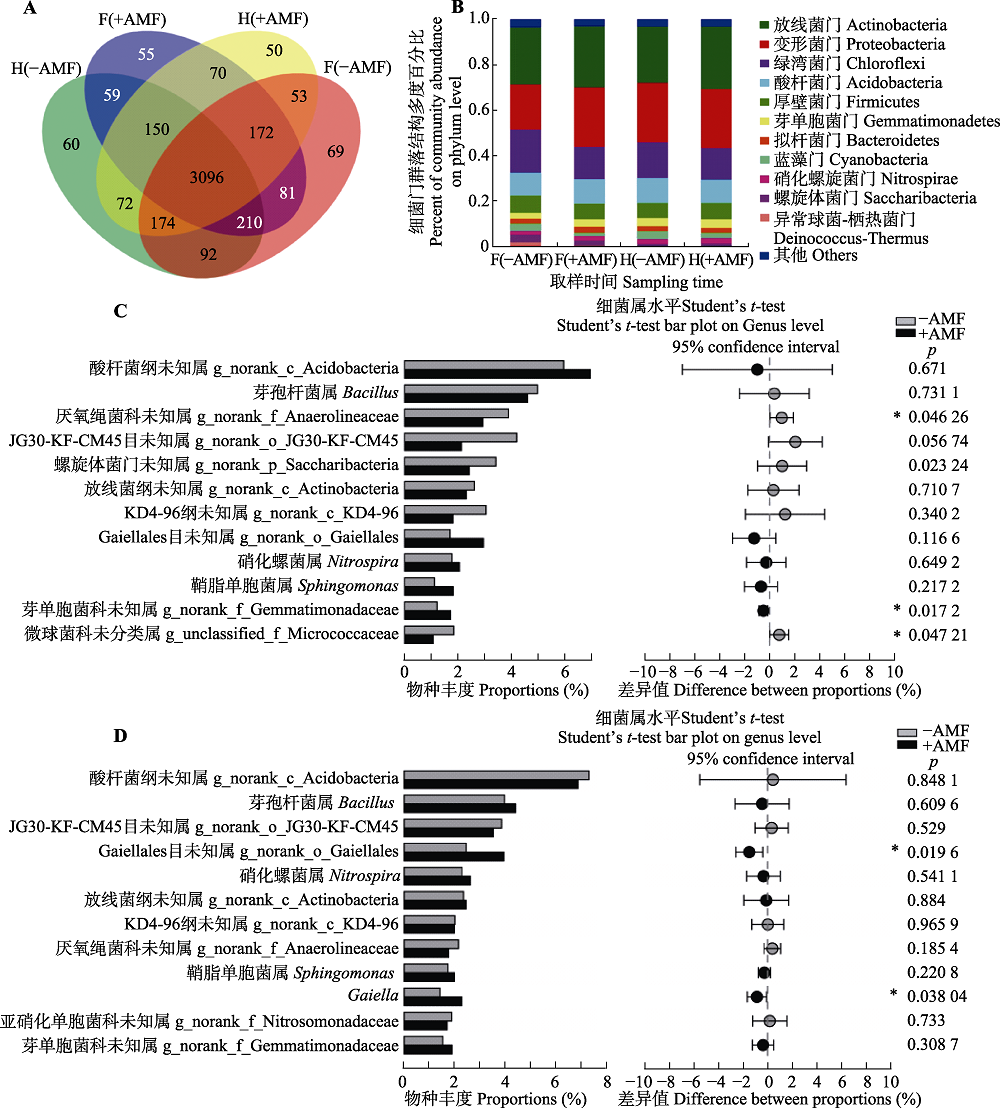
Fig. 3 Effects of Funneliformis mosseae on the structure and abundance of soil bacterial community in rhizosphere soil of peanut under continuing cropping. A, The Venn figure shows the number of bacterial operational taxonomic units in the two treatments. B, Abundance of soil bacteria in continuous cropping of peanut changed in the F. mosseae inoculation tratement C, Abundance of bacterial genera were significantly different between with and without F. mosseae inoculation in the flowering period of continuing cropping peanuts (mean ± SE, n = 3). D, Abundance of bacterial genera were significantly different between with and without F. mosseae inoculation in the harvesting period of continuous cropping peanuts (mean ± SE, n = 3). -AMF, without F. mosseae inoculation; +AMF, F. mosseae inoculation. F(-AMF), peanut rhizosphere soil without F. mosseae inoculation during the flowering period; F(+AMF), rhizosphere soil of peanut with F. mosseae inoculation; H(-AMF), peanut rhizosphere soil without F. mosseae inoculation during the harvest period; H(+AMF), peanut rhizosphere soil with F. mosseae inoculation during the harvest period. *, p < 0.05.
| 处理 Treatment | 单株结果数 Pod number per plant | 单株果质量 Pod mass per plant (g) | 饱果率 Full fruit rate (%) | 蛋白质 Protein (%) | 总氨基酸 Total amino acid (%) | 油酸 Oleic (%) | 亚油酸 Linoleic (%) |
|---|---|---|---|---|---|---|---|
| -AMF | 34.67 ± 2.08 | 41.85 ± 2.87 | 60.82 ± 0.02 | 18.34 ± 0.17 | 18.29 ± 1.68 | 52.46 ± 1.16 | 24.12 ± 1.37 |
| +AMF | 39.33 ± 0.58* | 52.05 ± 0.79* | 70.33 ± 0.04* | 21,89 ± 0.22* | 21.30 ± 0.97* | 57.38 ± 1.32* | 27.20 ± 1.19* |
Table 3 Effects of Funneliformis mosseae inoculation on the yield and quality of continuous cropping peanut
| 处理 Treatment | 单株结果数 Pod number per plant | 单株果质量 Pod mass per plant (g) | 饱果率 Full fruit rate (%) | 蛋白质 Protein (%) | 总氨基酸 Total amino acid (%) | 油酸 Oleic (%) | 亚油酸 Linoleic (%) |
|---|---|---|---|---|---|---|---|
| -AMF | 34.67 ± 2.08 | 41.85 ± 2.87 | 60.82 ± 0.02 | 18.34 ± 0.17 | 18.29 ± 1.68 | 52.46 ± 1.16 | 24.12 ± 1.37 |
| +AMF | 39.33 ± 0.58* | 52.05 ± 0.79* | 70.33 ± 0.04* | 21,89 ± 0.22* | 21.30 ± 0.97* | 57.38 ± 1.32* | 27.20 ± 1.19* |
| [1] | Adolfsson L, Nziengui H, Abreu IN, Šimura J, Beebo A, Herdean A, Aboalizadeh J, Široká J, Moritz T, Novák O, Ljung K, Schoefs B, Spetea C (2017). Enhanced secondary- and hormone metabolism in leaves of arbuscular mcorrhizal Medicago truncatula. Plant Physiology, 175, 392-411. |
| [2] | Azcón-Aguilar C, Barea JM (1997). Arbuscular mycorrhizas and biological control of soil-borne plant pathogens—An overview of the mechanisms involved. Mycorrhiza, 6, 457-464. |
| [3] | Bagyaraj DJ, Manjunath A, Patil RB (1979). Interaction between a vesicular-arbuscular mycorrhiza and rhizobium and their effects on soybean in the field. New Phytologist, 82, 141-145. |
| [4] | Bao SD (1999). Soil Agrochemical Analysis. 3rd edn. China Agricultural Press, Beijing. 42-56, 71-80, 100-108. |
| [ 鲍士旦 (1999). 土壤农化分析. 第三版. 中国农业出版社, 北京. 42-56, 71-80, 100-108.] | |
| [5] | Chen MN, Li X, Yang QL, Chi XY, Pan LJ, Chen N, Yang Z, Wang T, Wang M, Yu SL (2012). Soil eukaryotic microorganism succession as affected by continuous cropping of peanut-pathogenic and beneficial fungi were selected. PLOS ONE, 7, e40659. DOI: 10.1371/journal.pone.0040659. |
| [6] | Chen MN, Li X, Yang QL, Chi XY, Pan LJ, Chen N, Yang Z, Wang T, Wang M, Yu SL (2014). Dynamic succession of soil bacterial community during continuous cropping of peanut ( Arachis hypogaea L.). PLOS ONE, 9, e101355. DOI: 10.1371/journal.pone.0101355. |
| [7] | Cui L, Guo F, Zhang JL, Yang S, Meng JJ, Geng Y, Wang Q, Li XG, Wan SB (2019). Arbuscular mycorrhizal fungi combined with exogenous calcium improves the growth of peanut ( Arachis hypogaea L.) seedlings under continuous cropping. Journal of Integrative Agriculture, 18, 407-416. |
| [8] | Dehne H, Schonbeck F, Baltruschat H (1978). The influence of endotrophic mycorrhiza on plant disease. 3. Chitinase-activity and ornithine-cylce. Journal of Plant Disease Protection, 85, 666-678. |
| [9] | Desjardins AE (2003). Gibberella from A (venaceae) to Z (eae). Annual Review of Phytopathology, 41, 177-198. |
| [10] | Feng HS, Wan SB, Zuo XQ, Cheng B (1999). Changes of main microbial groups in soil and rhizosphere of peanut continuous cropping and their correlation with yield. Peanut Science and Technology, (Suppl. 1), 277-283. |
| [ 封海胜, 万书波, 左学青, 成波 (1999). 花生连作土壤及根际主要微生物类群的变化及与产量的相关. 花生科技, (增刊1), 277-283.] | |
| [11] | Feng Y (2017). Effects of arbuscular mycorrhizal fungi (AMF) on soil microbes and nutrients in rhizosphere of Salvia miltiorrhiza. China Agriculture Information, (10), 57-58. |
| [ 封晔 (2017). 丛枝菌根真菌(AMF)对丹参根际土壤微生物及养分的影响. 中国农业信息, (10), 57-58.] | |
| [12] | Fu XF, Zhang GP, Zhang XW, Ren JH (2016). Effects of PSB and AMF on growth, microorganisms and soil enzyme activities in the rhizosphere of Taxus chinensis var. mairei seedlings. Acta Botanica Boreali-Occidentalia Sinica, 36, 353-360. |
| [ 付晓峰, 张桂萍, 张小伟, 任嘉红 (2016). 溶磷细菌和丛枝菌根真菌接种对南方红豆杉生长及根际微生物和土壤酶活性的影响. 西北植物学报, 36, 353-360.] | |
| [13] | Gao P, Li F, Guo YE, Duan TY (2017). Advances in AM fungi and rhizobium to control plant fungal diseases. Acta Agrestia Sinica, 25, 236-242. |
| [ 高萍, 李芳, 郭艳娥, 段廷玉 (2017). 丛枝菌根真菌和根瘤菌防控植物真菌病害的研究进展. 草地学报, 25, 236-242.] | |
| [14] | Garcia K, Chasman D, Roy S, Ané JM (2017). Physiological responses and gene co-expression network of mycorrhizal roots under K+ deprivation . Plant Physiology, 173, 1811-1823. |
| [15] | Guan SY (1986). Soil Enzyme and Its Research Method. China Agricultural Press, Beijing. 274-276, 294-297, 309-313, 332-333. |
| [ 关松荫 (1986). 土壤酶及其研究方法. 中国农业出版社, 北京. 274-276, 294-297, 309-313, 332-333.] | |
| [16] | Han B, Guo SR, He CX, Yan Y, Yu XC (2012). Effects of arbuscular mycorrhiza fungi on the plant growth, fruit yield and fruit quality of cucumber under salt stress. Chinese Journal of Applied Ecology, 23, 154-158. |
| [ 韩冰, 郭世荣, 贺超兴, 闫妍, 于贤昌 (2012). 丛枝菌根真菌对盐胁迫下黄瓜植株生长果实产量和品质的影响. 应用生态学报, 23, 154-158. ] | |
| [17] | He ZQ, Li HX, Tang HR (2010). Effect of arbuscular mycorrhizal fungi on cucumber rhizoctonia rot. Journal of Sichuan Agricultural University, 28, 200-204. |
| [ 贺忠群, 李焕秀, 汤浩茹 (2010). 丛枝菌根真菌对黄瓜立枯病的影响. 四川农业大学学报, 28, 200-204.] | |
| [18] | Huang YQ, Han XR, Yang JF, Liu N, Liu XH (2011). Studies on the changes of soil microbial communities under peanuts continuous cropping. Chinese Journal of Soil Science, 42, 552-556. |
| [ 黄玉茜, 韩晓日, 杨劲峰, 刘宁, 刘小虎 (2011). 花生连作土壤微生物区系变化研究. 土壤通报, 42, 552-556.] | |
| [19] | Jakobsen I, Gazey C, Abbott LK (2001). Phosphate transport by communities of arbuscular mycorrhizal fungi in intact soil cores. New Phytologist, 149, 95-103. |
| [20] | Kunoh H (2002). Endophytic actinomycetes: Attractive biocontrol agents. Journal of General Plant Pathology, 68, 249-252. |
| [21] | LeBlanc N, Kinkel L, Kistler HC (2017). Plant diversity and plant identity influence Fusarium communities in soil. Mycologia, 109, 128-139. |
| [22] | Lehmann A, Rillig MC (2015). Arbuscular mycorrhizal contribution to copper, manganese and iron nutrient concentrations in crops—A meta-analysis. Soil Biology & Biochemistry, 81, 147-158. |
| [23] | Lei H, Wang JZ, Tan Y, Zeng M (2019). Effects of reduced fertilization and inoculation with AMF on growth and soil enzyme activities of potted citrus. South China Fruits, 48(2), 11-17. |
| [ 雷卉, 王久照, 谭嫣, 曾明 (2019). 减量施肥和接种AMF对盆栽柑橘生长及土壤酶活性的影响. 中国南方果树, 48(2), 11-17.] | |
| [24] | Li H, Chen XW, Wong MH (2016). Arbuscular mycorrhizal fungi reduced the ratios of inorganic/organic arsenic in rice grains. Chemosphere, 145, 224-230. |
| [25] | Li XG, Ding CF, Zhang TL, Wang XX (2014). Fungal pathogen accumulation at the expense of plant-beneficial fungi as a consequence of consecutive peanut monoculturing. Soil Biology & Biochemistry, 72, 11-18. |
| [26] | Li XG, Zhang TL, Wang XX (2015). Advances in mechanism of peanut continuous cropping obstacle. Soils, 47, 266-271. |
| [ 李孝刚, 张桃林, 王兴祥 (2015). 花生连作土壤障碍机制研究进展. 土壤, 47, 266-271.] | |
| [27] | Li Z, Jiang LG, Tang RH, Guo WF (2018). Effects of long-term continuous peanut cropping on dry matter weight of different peanut varieties, soil nutrient contents and enzyme activities. Soils, 50, 491-497. |
| [ 李忠, 江立庚, 唐荣华, 郭文峰 (2018). 连作对花生土壤酶活性、养分含量和植株产量的影响. 土壤, 50, 491-497.] | |
| [28] | Liu XL, Xi XY, Shen H, Liu B, Guo T (2014). Influences of arbuscular mycorrhizal (AM) fungi inoculation on resistance of tobacco to bacterial wilt. Tobacco Science & Technology, 47(5), 94-98. |
| [ 刘先良, 习向银, 申鸿, 刘斌, 郭涛 (2014). 接种丛枝菌根真菌对烟草青枯病抗性的影响. 烟草科技, 47(5), 94-98.] | |
| [29] | Luo WQ, Li J, Ma XN, Niu H, Hou SW, Wu FY (2019). Effect of arbuscular mycorrhizal fungi on uptake of selenate, selenite, and selenomethionine by roots of winter wheat. Plant and Soil, 438, 71-83. |
| [30] | Marschner P, Crowley D, Lieberei R (2001). Arbuscular mycorrhizal infection changes the bacterial 16S rDNA community composition in the rhizosphere of maize. Mycorrhiza, 11, 297-302. |
| [31] | Ning CH, Li WB, Liu RJ (2019). Research advances in plant symbiotic Actinomyces. Chinese Journal of Ecology, 38, 256-266. |
| [ 宁楚涵, 李文彬, 刘润进 (2019). 植物共生放线菌研究进展. 生态学杂志, 38, 256-266.] | |
| [32] | Ozgonen H, Akgul DS, Erkili A (2010). The effects of arbuscular mycorrhizal fungi on yield and stem rot caused by Sclerotium rolfsii Sacc. in peanut. African Journal of Agricultural Research, 5(2), 128-132. |
| [33] | Pozo MJ, López-Ráez JA, Azcón-Aguilar C, García-Garrido JM (2015). Phytohormones as integrators of environmental signals in the regulation of mycorrhizal symbioses. New Phytologist, 205, 1431-1436. |
| [34] | Qu MH, Yu YC, Li S, Zhang JC (2019). Advances in research on activation of mineral nutrients by arbuscular mycorrhizal fungi. Journal of Zhejiang A&F University, 36, 394-405. |
| [ 屈明华, 俞元春, 李生, 张金池 (2019). 丛枝菌根真菌对矿质养分活化作用研究进展. 浙江农林大学学报, 36, 394-405.] | |
| [35] | Rouphael Y, Franken P, Schneider C, Schwarz D, Giovannetti M, Agnolucci M, De Pascale S, Bonini P, Colla G (2015). Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Scientia Horticulturae, 196, 91-108. |
| [36] | Shen ML, Zhao C, Liao P, Li J, Cheng XF, Li CC, Zhang Q, Li YB, Zhang LL, Zhao K (2018). The isolation and identification of endophytic actinobacteria from Glycyrrhiza glabra in the Tarim basin and their stress resistance and ability to promote plant growth. Pratacultural Science, 35, 1624-1633. |
| [ 申枚灵, 赵翀, 廖萍, 李静, 程雪芬, 李成成, 张琴, 李艳宾, 张利莉, 赵珂 (2018). 塔里木盆地光果甘草内生放线菌的分离鉴定及抗逆、促生特性. 草业科学, 35, 1624-1633.] | |
| [37] | Smith SE, Manjarrez M, Stonor R, McNeill A, Smith FA (2015). Indigenous arbuscular mycorrhizal (AM) fungi contribute to wheat phosphate uptake in a semi-arid field environment, shown by tracking with radioactive phosphorus. Applied Soil Ecology, 96, 68-74. |
| [38] | Sun RL, Zhao BQ, Zhu LS, Xu J, Zhang FD (2008). Dynamic changes of soil enzyme activities in long-term fertilization soil. Ecology and Environment, 17, 2059-2063. |
| [ 孙瑞莲, 赵秉强, 朱鲁生, 徐晶, 张夫道 (2008). 长期定位施肥田土壤酶活性的动态变化特征. 生态环境, 17, 2059-2063.] | |
| [39] | Sun XS, Feng HS, Wan SB, Zuo XQ (2001). Changes of main microbial strains and enzymes activities in peanut continuous cropping soil and their interactions. Acta Agronomica Sinica, 27, 617-621. |
| [ 孙秀山, 封海胜, 万书波, 左学青 (2001). 连作花生田主要微生物类群与土壤酶活性变化及交互作用. 作物学报, 27, 617-621.] | |
| [40] | Sun XX, He CX, Li YS, Yu XC (2017). Effects of arbuscular mycorrhizal fungi on microbial community and function in the rhizosphere soil of cucumber plants. Mycosystema, 36, 892-903. |
| [ 孙秀秀, 贺超兴, 李衍素, 于贤昌 (2017). AM真菌对黄瓜根围土壤微生物群落功能的影响. 菌物学报, 36, 892-903.] | |
| [41] | Teng Y, Ren WJ, Li ZG, Wang XB, Liu WX, Luo YM (2015). Advance in mechanism of peanut continuous cropping obstacle. Soils, 47, 259-265. |
| [ 滕应, 任文杰, 李振高, 王小兵, 刘五星, 骆永明 (2015). 花生连作障碍发生机理研究进展. 土壤, 47, 259-265.] | |
| [42] | Wan SB, Wang CB, Lu JL, Li GM, Lun WZ, Wu ZF, Cheng B (2007). Study on the growth characteristics of continuous cropping peanut. Shandong Agricultural Sciences,(2), 32-36. |
| [ 万书波, 王才斌, 卢俊玲, 李光敏, 伦伟志, 吴正峰, 成波 (2007). 连作花生的生育特性研究. 山东农业科学, (2), 32-36.] | |
| [43] | Wang J, Bi YL, Zhang YX, Hong TC, Qiu L, Chen SL (2014). Effects of arbuscular mycorrhiza on soil microorganisms and enzyme activities in disturbed coal mine areas. Journal of Southern Agriculture, 45, 1417-1423. |
| [ 王瑾, 毕银丽, 张延旭, 洪天才, 裘浪, 陈书琳 (2014). 接种丛枝菌根对矿区扰动土壤微生物群落及酶活性的影响. 南方农业学报, 45, 1417-1423.] | |
| [44] | Yao XD, Li XG, Ding CF, Han ZM, Wang XX (2019). Microzone distribution characteristics of soil microbial community with peanut cropping system, monocropping or rotation. Acta Pedologica Sinica, 56, 975-985. |
| [ 姚小东, 李孝刚, 丁昌峰, 韩正敏, 王兴祥 (2019). 连作和轮作模式下花生土壤微生物群落不同微域分布特征. 土壤学报, 56, 975-985.] | |
| [45] | Zhang CS (2013). Study on the Distribution, Aflatoxin Production and Genetic Diversity of Aspergillus flavus in Soils of Peanut Fields in Four Agroecological Zones of China. PhD dissertation, Chinese Academy of Agricultural Science, Beijing. |
| [ 张初署 (2013). 中国四个生态区花生土壤中黄曲霉菌分布、产毒特征及遗传多样性研究. 博士学位论文, 中国农业科学院, 北京.] | |
| [46] | Zhou BL, Zheng JD, Bi XH, Cai LL, Guo WW (2015). Effects of mycorrhizal fungi on eggplant verticillium wilt and eggplant growth. Chinese Journal of Ecology, 34, 1026-1030. |
| [ 周宝利, 郑继东, 毕晓华, 蔡莲莲, 郭伟伟 (2015). 丛枝菌根真菌对茄子黄萎病的防治效果和茄子植株生长的影响. 生态学杂志, 34, 1026-1030.] |
| [1] | . Effects and metabolites analysis of Penicillium oxalate C11 on Rehmannia glutinosa growth [J]. Chin J Plant Ecol, 2024, 48(预发表): 0-0. |
| [2] | Ke-Yu CHEN Sen Xing Yu Tang Sun JiaHui Shijie Ren Bao-Ming JI. Arbuscular mycorrhizal fungal community characteristics and driving factors in different grassland types [J]. Chin J Plant Ecol, 2024, 48(5): 660-674. |
| [3] | Die Hu Xinqi Jiang DAI Zhicong Daiyi Chen Yu Zhang Shan-Shan Qi. Arbuscular mycorrhizal fungi enhance the herbicide tolerance of an invasive weed Sphagneticola trilobata [J]. Chin J Plant Ecol, 2024, 48(5): 651-659. |
| [4] | WANG Ge, HU Shu-Ya, LI Yang, CHEN Xiao-Peng, LI Hong-Yu, DONG Kuan-Hu, HE Nian-Peng, WANG Chang-Hui. Temperature sensitivity of soil net nitrogen mineralization rates across different grassland types [J]. Chin J Plant Ecol, 2024, 48(4): 523-533. |
| [5] | CHEN Bao-Dong, FU Wei, WU Song-Lin, ZHU Yong-Guan. Involvements of mycorrhizal fungi in terrestrial ecosystem carbon cycling [J]. Chin J Plant Ecol, 2024, 48(1): 1-20. |
| [6] | YANG Jia-Rong, DAI Dong, CHEN Jun-Fang, WU Xian, LIU Xiao-Lin, LIU Yu. Insight into recent studies on the diversity of arbuscular mycorrhizal fungi in shaping plant community assembly and maintaining rare species [J]. Chin J Plant Ecol, 2023, 47(6): 745-755. |
| [7] | HE Fei, LI Chuan, Faisal SHAH, LU Xie-Min, WANG Ying, WANG Meng, RUAN Jia, WEI Meng-Lin, MA Xing-Guang, WANG Zhuo, JIANG Hao. Carbon transport and phosphorus uptake in an intercropping system of Robinia pseudoacacia and Amorphophallus konjac mediated by arbuscular mycorrhizal hyphal networks [J]. Chin J Plant Ecol, 2023, 47(6): 782-791. |
| [8] | LI Liu, LIU Qing-Hua, YIN Chun-Ying. Selenium biofortification in plants and application potential of microorganisms in selenium biofortification [J]. Chin J Plant Ecol, 2023, 47(6): 756-769. |
| [9] | HE Min, XU Qiu-Yue, XIA Yun, YANG Liu-Ming, FAN Yue-Xin, YANG Yu-Sheng. Plant phosphorus acquisition mechanisms and their response to global climate changes [J]. Chin J Plant Ecol, 2023, 47(3): 291-305. |
| [10] | LI Wan-Nian, LUO Yi-Min, HUANG Ze-Yue, YANG Mei. Effects of mixed young plantations of Parashorea chinensis on soil microbial functional diversity and carbon source utilization [J]. Chin J Plant Ecol, 2022, 46(9): 1109-1124. |
| [11] | XIE Wei, HAO Zhi-Peng, ZHANG Xin, CHEN Bao-Dong. Research progress and prospect of signal transfer among plants mediated by arbuscular mycorrhizal networks [J]. Chin J Plant Ecol, 2022, 46(5): 493-515. |
| [12] | WU Zan, PENG Yun-Feng, YANG Gui-Biao, LI Qin-Lu, LIU Yang, MA Li-Hua, YANG Yuan-He, JIANG Xian-Jun. Effects of land degradation on soil and microbial stoichiometry in Qingzang Plateau alpine grasslands [J]. Chin J Plant Ecol, 2022, 46(4): 461-472. |
| [13] | MAO Jin, DUO Ying, DENG Jun, CHENG Jie, CHENG Ji-Min, PENG Chang-Hui, GUO Liang. Influences of warming and snow reduction in winter on soil nutrients and bacterial communities composition in a typical grassland of the Loess Plateau [J]. Chin J Plant Ecol, 2021, 45(8): 891-902. |
| [14] | MA Ju-Feng, XIN Min, XU Chen-Chao, ZHU Wan-Ying, MAO Chuan-Zao, CHEN Xin, CHENG Lei. Effects of arbuscular mycorrhizal fungi and nitrogen addition on nitrogen uptake of rice genotypes with different root morphologies [J]. Chin J Plant Ecol, 2021, 45(7): 728-737. |
| [15] | YANG Jian-Qiang, DIAO Hua-Jie, HU Shu-Ya, WANG Chang-Hui. Effects of nitrogen addition at different levels on soil microorganisms in saline-alkaline grassland of northern China [J]. Chin J Plant Ecol, 2021, 45(7): 780-789. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2022 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn