

Chin J Plant Ecol ›› 2019, Vol. 43 ›› Issue (8): 635-647.DOI: 10.17521/cjpe.2019.0076
• Reviews • Next Articles
Received:2019-04-07
Revised:2019-08-02
Online:2019-08-20
Published:2020-01-03
Contact:
WANG Chuan-Kuan ORCID:0000-0003-3513-5426
Supported by:LI Zhi-Min, WANG Chuan-Kuan. Research progress on responses of xylem of woody plants to freeze-thaw embolism[J]. Chin J Plant Ecol, 2019, 43(8): 635-647.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.plant-ecology.com/EN/10.17521/cjpe.2019.0076
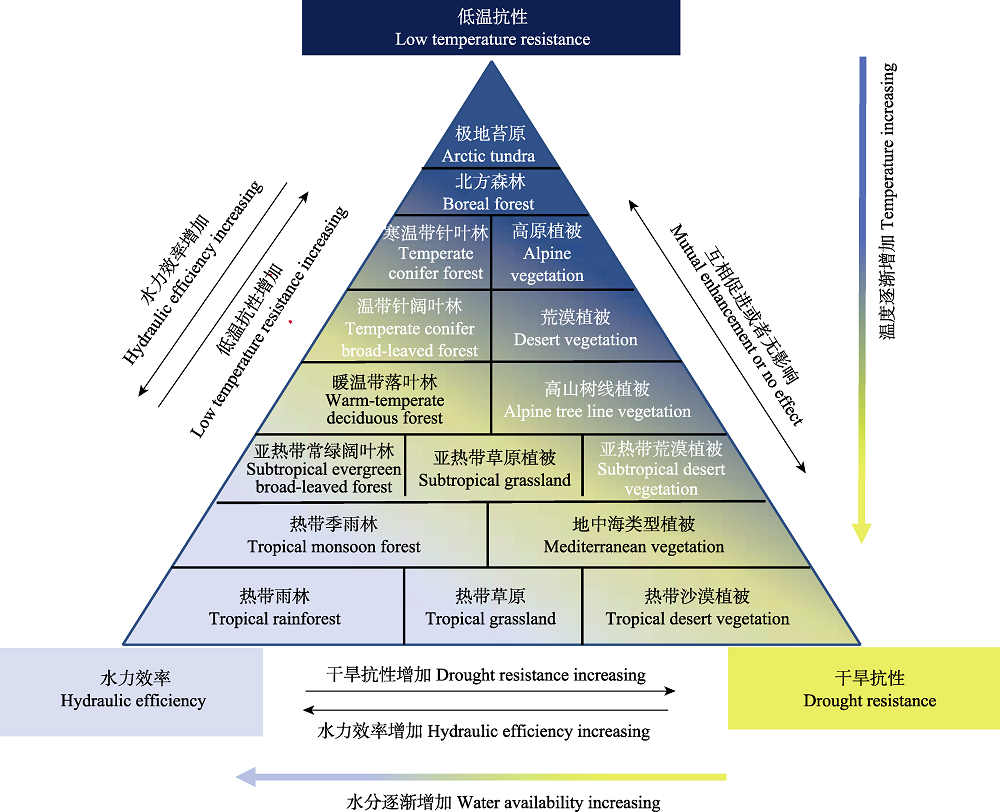
Fig. 2 A conceptual diagram of multiple trade-offs among low-temperature resistance, drought resistance and hydraulic efficiency of the major vegetation types in the world. High (low) hydraulic efficiency is associated with weak (strong) low-temperature resistance, but the capacity of positive refilling embolism may influence the trade-off between hydraulic efficiency and low-temperature resistance, and low-temperature also affects the plant’s frost fatigue. High (low) hydraulic efficiency is associated with weak (strong) drought resistance, but many species have both low hydraulic efficiency and drought resistance rather than high hydraulic efficiency and drought resistance. Drought stress may increase low-temperature resistance (asynchronous), and vice versa; concomitant drought and low-temperature stresses aggravate the loss of hydraulic conductance, but there are no related impacts of drought and low-temperature stresses on the hydraulic system.
| [1] | Améglio T, Bodet C, Lacointe A, Cochard H (2002). Winter embolism, mechanisms of xylem hydraulic conductivity recovery and springtime growth patterns in walnut and peach trees. Tree Physiology, 22, 1211-1220. |
| [2] | Améglio T, Cochard H, Ewers FW (2001). Stem diameter variations and cold hardiness in walnut trees. Journal of Experimental Botany, 52, 2135-2142. |
| [3] | Améglio T, Decourteix M, Alves G, Valentin V, Sakr S, Julien JL, Petel G, Guilliot A, Lacointe A (2004). Temperature effects on xylem sap osmolarity in walnut trees: Evidence for a vitalistic model of winter embolism repair. Tree Physiology, 24, 785-793. |
| [4] | Arias NS, Bucci SJ, Scholz FG, Goldstein G (2015). Freezing avoidance by supercooling in Olea europaea cultivars: The role of apoplastic water, solute content and cell wall rigidity. Plant, Cell & Environment, 38, 2061-2070. |
| [5] | Arias NS, Scholz FG, Goldstein G, Bucci SJ (2017). The cost of avoiding freezing in stems: Trade-off between xylem resistance to cavitation and supercooling capacity in woody plants. Tree Physiology, 37, 1251-1262. |
| [6] | Bachofen C, Wohlgemuth T, Ghazoul J, Moser B (2016). Cold temperature extremes during spring do not limit the range shift of Mediterranean pines into regions with intermittent frost. Functional Ecology, 30, 856-865. |
| [7] | Ball MC, Canny MJ, Huang CX, Heady RD (2004). Structural changes in acclimated and unacclimated leaves during freezing and thawing. Functional Plant Biology, 31, 29-40. |
| [8] | Blackman CJ, Brodribb TJ, Jordan GJ (2010). Leaf hydraulic vulnerability is related to conduit dimensions and drought resistance across a diverse range of woody angiosperms. New Phytologist, 188, 1113-1123. |
| [9] | Bowling DR, Logan BA, Hufkens K, Aubrecht DM, Richardson AD, Burns SP, Anderegg WRL, Blanken PD, Eiriksson DP (2018). Limitations to winter and spring photosynthesis of a Rocky Mountain subalpine forest. Agricultural and Forest Meteorology, 252, 241-255. |
| [10] | Brodersen CR, McElrone AJ (2013). Maintenance of xylem network transport capacity: A review of embolism repair in vascular plants. Frontiers in Plant Science, 4, 108. DOI: 10.3389/fpls.2013.00108. |
| [11] | Brodribb TJ, Feild TS, Jordan GJ (2007). Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiology, 144, 1890-1898. |
| [12] | Buchner O, Neuner G (2011). Winter frost resistance of Pinus cembra measured in situ at the alpine timberline as affected by temperature conditions. Tree Physiology, 31, 1217-1227. |
| [13] | Cao KF, Yang SJ, Zhang YJ, Brodribb TJ (2012). The maximum height of grasses is determined by roots. Ecology Letters, 15, 666-672. |
| [14] | Carles S, Lamhamedi MS, Stowe DC, Bernier PY, Veilleux L, Margolis HA (2011). Relationships between frost hardiness, root growth potential, and photosynthesis of nursery- grown white spruce seedlings. Annals of Forest Science, 68, 1303-1313. |
| [15] | Charra-Vaskou K, Badel E, Charrier G, Ponomarenko A, Bonhomme M, Foucat L, Mayr S, Améglio T (2016). Cavitation and water fluxes driven by ice water potential in Juglans regia during freeze-thaw cycles. Journal of Experimental Botany, 67, 739-750. |
| [16] | Charrier G, Charra-Vaskou K, Kasuga J, Cochard H, Mayr S, Améglio T (2014). Freeze-thaw stress: Effects of temperature on hydraulic conductivity and ultrasonic activity in ten woody angiosperms. Plant Physiology, 164, 992-998. |
| [17] | Charrier G, Cochard H, Améglio T (2013). Evaluation of the impact of frost resistances on potential altitudinal limit of trees. Tree Physiology, 33, 891-902. |
| [18] | Charrier G, Ngao J, Saudreau M, Améglio T (2015). Effects of environmental factors and management practices on microclimate, winter physiology, and frost resistance in trees. Frontiers in Plant Science, 6, 259. DOI: 10.3389/fpls.2015.00259. |
| [19] | Charrier G, Nolf M, Leitinger G, Charra-Vaskou K, Losso A, Tappeiner U, Améglio T, Mayr S (2017). Monitoring of freezing dynamics in trees: A simple phase shift causes complexity. Plant Physiology, 173, 2196-2207. |
| [20] | Choat B, Brodie TW, Cobb AR, Zwieniecki MA, Holbrook NM (2006). Direct measurements of intervessel pit membrane hydraulic resistance in two angiosperm tree species. American Journal of Botany, 93, 993-1000. |
| [21] | Choat B, Brodribb TJ, Brodersen CR, Duursma RA, López R, Medlyn BE (2018). Triggers of tree mortality under drought. Nature, 558, 531-539. |
| [22] | Choat B, Jansen S, Brodribb TJ, Cochard H, Delzon S, Bhaskar R, Bucci SJ, Feild TS, Gleason SM, Hacke UG, Jacobsen AL, Lens F, Maherali H, Martínez-Vilalta J, Mayr S, Mencuccini M, Mitchell PJ, Nardini A, Pittermann J, Pratt RB, Sperry JS, Westoby M, Wright IJ, Zanne AE (2012). Global convergence in the vulnerability of forests to drought. Nature, 491, 752-755. |
| [23] | Choat B, Medek DE, Stuart SA, Pasquet-Kok J, Egerton JJG, Salari H, Sack L, Ball MC (2011). Xylem traits mediate a trade-off between resistance to freeze-thaw-induced embolism and photosynthetic capacity in overwintering evergreens. New Phytologist, 191, 996-1005. |
| [24] | Christensen-Dalsgaard KK, Tyree MT (2013). Does freezing and dynamic flexing of frozen branches impact the cavitation resistance of Malus domestica and the Populus clone walker? Oecologia, 173, 665-674. |
| [25] | Christensen-Dalsgaard KK, Tyree MT (2014). Frost fatigue and spring recovery of xylem vessels in three diffuse-porous trees in situ. Plant, Cell & Environment, 37, 1074-1085. |
| [26] | Climent J, Costa e Silva F, Chambel MR, Pardos M, Almeida MH (2009). Freezing injury in primary and secondary needles of Mediterranean pine species of contrasting ecological niches. Annals of Forest Science, 66, 407. DOI: 10.1051/forest/2009016. |
| [27] | Cochard H, Tyree MT (1990). Xylem dysfunction in Quercus: Vessel sizes, tyloses, cavitation and seasonal changes in embolism. Tree Physiology, 6, 393-407. |
| [28] | Davis SD, Sperry JS, Hacke UG (1999). The relationship between xylem conduit diameter and cavitation caused by freezing. American Journal of Botany, 86, 1367-1372. |
| [29] | de Swaef T, Hanssens J, Cornelis A, Steppe K (2013). Non-destructive estimation of root pressure using sap flow, stem diameter measurements and mechanistic modelling. Annals of Botany, 111, 271-282. |
| [30] | Domec JC (2011). Let’s not forget the critical role of surface tension in xylem water relations. Tree Physiology, 31, 359-360. |
| [31] | Feng F, Ding F, Tyree MT (2015). Investigations concerning cavitation and frost fatigue in clonal 84K poplar using high-resolution cavitron measurements. Plant Physiology, 168, 144-155. |
| [32] | Fernández-Pérez L, Villar-Salvador P, Martínez-Villata J, Toca A, Zavala MA (2018). Distribution of pines in the Iberian Peninsula agrees with species differences in foliage frost tolerance, not with vulnerability to freezing-induced xylem embolism. Tree Physiology, 38, 507-516. |
| [33] | Feild TS, Brodribb T (2001). Stem water transport and freeze-thaw xylem embolism in conifers and angiosperms in a Tasmanian treeline heath. Oecologia, 127, 314-320. |
| [34] | Gleason SM, Blackman CJ, Gleason ST, McCulloh KA, Ocheltree TW, Westoby M (2018). Vessel scaling in evergreen angiosperm leaves conforms with Murray’s law and area-filling assumptions: Implications for plant size, leaf size and cold tolerance. New Phytologist, 218, 1360-1370. |
| [35] | Gleason SM, Westoby M, Jansen S, Choat B, Hacke UG, Pratt RB, Bhaskar R, Brodribb TJ, Bucci SJ, Cao KF, Cochard H, Delzon S, Domec JC, Fan ZX, Feild TS, Jacobsen AL, Johnson DM, Lens F, Maherali H, Martínez-Vilalta J, Mayr S, McCulloh KA, Mencuccini M, Mitchell PJ, Morris H, Nardini A, Pittermann J, Plavcová L, Schreiber SG, Sperry JS, Wright IJ, Zanne AE (2016). Weak tradeoff between xylem safety and xylem-specific hydraulic efficiency across the world’s woody plant species. New Phytologist, 209, 123-136. |
| [36] | Granda E, Scoffoni C, Rubio-Casal AE, Sack L, Valladares F (2014). Leaf and stem physiological responses to summer and winter extremes of woody species across temperate ecosystems. Oikos, 123, 1281-1290. |
| [37] | Hacke UG, Lachenbruch B, Pittermann J, Mayr S, Domec JC, Schulte PJ (2015). The hydraulic architecture of conifers. In: Hacke U ed. Functional and Ecological Xylem Anatomy. Springer, Cham, Switzerland. 39-75. |
| [38] | Hacke UG, Sperry JS (2001). Functional and ecological xylem anatomy. Perspectives in Plant Ecology, Evolution and Systematics, 4, 97-115. |
| [39] | Hacke UG, Spicer R, Schreiber SG, Plavcová L (2017). An ecophysiological and developmental perspective on variation in vessel diameter. Plant, Cell & Environment, 40, 831-845. |
| [40] | Hajek P, Leuschner C, Hertel D, Delzon S, Schuldt B (2014). Trade-offs between xylem hydraulic properties, wood anatomy and yield in Populus. Tree Physiology, 34, 744-756. |
| [41] | Hao GY, Hoffmann WA, Scholz FG, Bucci SJ, Meinzer FC, Franco AC, Cao KF, Goldstein G (2008). Stem and leaf hydraulics of congeneric tree species from adjacent tropical savanna and forest ecosystems. Oecologia, 155, 405-415. |
| [42] | Hao GY, Wheeler JK, Holbrook NM, Goldstein G (2013). Investigating xylem embolism formation, refilling and water storage in tree trunks using frequency domain reflectometry. Journal of Experimental Botany, 64, 2321-2332. |
| [43] | Hillabrand RM, Hacke UG, Lieffers VJ (2016). Drought-induced xylem pit membrane damage in aspen and balsam poplar. Plant, Cell & Environment, 39, 2210-2220. |
| [44] | Hoermiller II, Ruschhaupt M, Heyer AG (2018). Mechanisms of frost resistance in Arabidopsis thaliana. Planta, 248, 827-835. |
| [45] | Hoffman L, DaCosta M, Ebdon JS, Zhao JZ (2012). Effects of drought preconditioning on freezing tolerance of perennial ryegrass. Environmental and Experimental Botany, 79, 11-20. |
| [46] | Hoffmann WA, Marchin RM, Abit P, Lau OL (2011). Hydraulic failure and tree dieback are associated with high wood density in a temperate forest under extreme drought. Global Change Biology, 17, 2731-2742. |
| [47] | Hölttä T, Dominguez Carrasco MDR, Salmon Y, Aalto J, Vanhatalo A, Bäck J, Lintunen A (2018). Water relations in silver birch during springtime: How is sap pressurised? Plant Biology, 20, 834-847. |
| [48] | Jiménez-Castillo M, Lusk CH (2013). Vascular performance of woody plants in a temperate rain forest: Lianas suffer higher levels of freeze-thaw embolism than associated trees. Functional Ecology, 27, 403-412. |
| [49] | Jin Y, Wang CK, Zhou ZH, Li ZM (2016). Co-ordinated performance of leaf hydraulics and economics in 10 Chinese temperate tree species. Functional Plant Biology, 43, 1082-1090. |
| [50] | Jin Y, Wang CK (2016). Leaf hydraulic traits and their trade-offs for nine Chinese temperate tree species with different wood properties. Chinese Journal of Plant Ecology, 40, 702-710. |
| [ 金鹰, 王传宽 (2016). 九种不同材性的温带树种叶水力性状及其权衡关系. 植物生态学报, 40, 702-710.] | |
| [51] | Jin Y, Wang CK, Zhou ZH (2016). Mechanisms of xylem embolism repair in woody plants: Research progress and questions. Chinese Journal of Plant Ecology, 40, 834-846. |
| [ 金鹰, 王传宽, 周正虎 (2016). 木本植物木质部栓塞修复机制: 研究进展与问题. 植物生态学报, 40, 834-846.] | |
| [52] | Johnson DM, McCulloh KA, Woodruff DR, Meinzer FC (2012). Hydraulic safety margins and embolism reversal in stems and leaves: Why are conifers and angiosperms so different? Plant Science, 195, 48-53. |
| [53] | Kong RS, Henry HAL (2016). Prior exposure to freezing stress enhances the survival and recovery of Poa pratensis exposed to severe drought. American Journal of Botany, 103, 1890-1896. |
| [54] | Kreyling J, Schmid S, Aas G (2015). Cold tolerance of tree species is related to the climate of their native ranges. Journal of Biogeography, 42, 156-166. |
| [55] | Kreyling J, Wiesenberg GLB, Thiel D, Wohlfart C, Huber G, Walter J, Jentsch A, Konnert M, Beierkuhnlein C (2012). Cold hardiness of Pinus nigra Arnold as influenced by geographic origin, warming, and extreme summer drought. Environmental and Experimental Botany, 78, 99-108. |
| [56] | Lemoine D, Granier A, Cochard H (1999). Mechanism of freeze-induced embolism in Fagus sylvatica L. Trees, 13, 206-210. |
| [57] | Lens F, Sperry JS, Christman MA, Choat B, Rabaey D, Jansen S (2011). Testing hypotheses that link wood anatomy to cavitation resistance and hydraulic conductivity in the genus Acer. New Phytologist, 190, 709-723. |
| [58] | Lens F, Tixier A, Cochard H, Sperry JS, Jansen S, Herbette S (2013). Embolism resistance as a key mechanism to understand adaptive plant strategies. Current Opinion in Plant Biology, 16, 287-292. |
| [59] | Lindfors L, Hölttä T, Lintunen A, Porcar-Castell A, Nikinmaa E, Juurola E (2015). Dynamics of leaf gas exchange, chlorophyll fluorescence and stem diameter changes during freezing and thawing of Scots pine seedlings. Tree Physiology, 35, 1314-1324. |
| [60] | Lintunen A, Hölttä T, Kulmala M (2013). Anatomical regulation of ice nucleation and cavitation helps trees to survive freezing and drought stress. Scientific Reports, 3, 2031. DOI: 10.1038/srep02031. |
| [61] | Lintunen A, Lindfors L, Kolari P, Juurola E, Nikinmaa E, Hölttä T (2014). Bursts of CO2 released during freezing offer a new perspective on avoidance of winter embolism in trees. Annals of Botany, 114, 1711-1718. |
| [62] | Lintunen A, Lindfors L, Nikinmaa E, Hölttä T (2016). Xylem diameter changes during osmotic stress, desiccation and freezing in Pinus sylvestris and Populus tremula. Tree Physiology, 37, 491-500. |
| [63] | Lintunen A, Mayr S, Salmon Y, Cochard H, Hölttä T (2018). Drivers of apoplastic freezing in gymnosperm and angiosperm branches. Ecology and Evolution, 8, 333-343. |
| [64] | Losso A, Nardini A, Dämon B, Mayr S (2018). Xylem sap chemistry: Seasonal changes in timberline conifers Pinus cembra, Picea abies, and Larix decidua. Biologia Plantarum, 62, 157-165. |
| [65] | Martínez-Vilalta J, Sala AN, Asensio D, Galiano L, Hoch G, Palacio S, Piper FI, Lloret F (2016). Dynamics of non-structural carbohydrates in terrestrial plants: A global synthesis. Ecological Monographs, 86, 495-516. |
| [66] | Mayr S, Améglio T (2016). Freezing stress in tree xylem. Progress in Botany, 77, 381-414. |
| [67] | Mayr S, Cochard H, Améglio T, Kikuta SB (2007). Embolism formation during freezing in the wood of Picea abies. Plant Physiology, 143, 60-67. |
| [68] | Mayr S, Hacke U, Schmid P, Schwienbacher F, Gruber A (2006). Frost drought in conifers at the alpine timberline: Xylem dysfunction and adaptations. Ecology, 87, 3175-3185. |
| [69] | Mayr S, Schmid P, Laur J, Rosner S, Charra-Vaskou K, Dämon B, Hacke UG (2014). Uptake of water via branches helps timberline conifers refill embolized xylem in late winter. Plant Physiology, 164, 1731-1740. |
| [70] | Mayr S, Schwienbacher F, Bauer H (2003). Winter at the alpine timberline. Why does embolism occur in Norway spruce but not in stone pine? Plant Physiology, 131, 780-792. |
| [71] | Mayr S, Sperry JS (2010). Freeze-thaw-induced embolism in Pinus contorta: Centrifuge experiments validate the ‘thaw-expansion hypothesis’ but conflict with ultrasonic emission data. New Phytologist, 185, 1016-1024. |
| [72] | Mayr S, Wolfschwenger M, Bauer H (2002). Winter-drought induced embolism in Norway spruce (Picea abies) at the Alpine timberline. Physiologia Plantarum, 115, 74-80. |
| [73] | McCully ME, Canny MJ, Huang CX (2004). The management of extracellular ice by petioles of frost-resistant herbaceous plants. Annals of Botany, 94, 665-674. |
| [74] | McDowell NG (2011). Mechanisms linking drought, hydraulic, carbon metabolism, and vegetation mortality. Plant Physiology, 155, 1051-1059. |
| [75] | McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, Yepez EA (2008). Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytologist, 178, 719-739. |
| [76] | Medeiros JS, Danielson SC (2018). Renewed interest in whole-plant physiology sheds light on the complexity of plant stress response architecture. Tree Physiology, 38, 503-506. |
| [77] | Medeiros JS, Pockman WT (2011). Drought increases freezing tolerance of both leaves and xylem of Larrea tridentate. Plant, Cell & Environments, 34, 43-51. |
| [78] | Nardini A, Lo Gullo MA, Salleo S (2011). Refilling embolized xylem conduits: Is it a matter of phloem unloading? Plant Science, 180, 604-611. |
| [79] | Nardini A, Luglio J (2014). Leaf hydraulic capacity and drought vulnerability: Possible trade-offs and correlations with climate across three major biomes. Functional Ecology, 28, 810-818. |
| [80] | Nardini A, Pedà G, La Rocca N (2012). Trade-offs between leaf hydraulic capacity and drought vulnerability: Morpho-anatomical bases, carbon costs and ecological consequences. New Phytologist, 196, 788-798. |
| [81] | Niu CY, Meinzer FC, Hao GY (2017). Divergence in strategies for coping with winter embolism among co-occurring temperate tree species: The role of positive xylem pressure, wood type and tree stature. Functional Ecology, 31, 1550-1560. |
| [82] | Pittermann J, Sperry J (2003). Tracheid diameter is the key trait determining the extent of freezing-induced embolism in conifers. Tree Physiology, 23, 907-914. |
| [83] | Pittermann J, Sperry JS (2006). Analysis of freeze-thaw embolism in conifers. The interaction between cavitation pressure and tracheid size. Plant Physiology, 140, 374-382. |
| [84] | Pivovaroff AL, Sack L, Santiago LS (2014). Coordination of stem and leaf hydraulic conductance in southern California shrubs: A test of the hydraulic segmentation hypothesis. New Phytologist, 203, 842-850. |
| [85] | Ployet R, Soler M, Carocha V, Ladouce N, Alves A, Rodrigues J, Harvengt L, Marque C, Teulières C, Grima-Pettenati J, Mounet F (2018). Long cold exposure induces transcriptional and biochemical remodelling of xylem secondary cell wall in Eucalyptus. Tree Physiology, 38, 409-422. |
| [86] | Prendin AL, Mayr S, Beikircher B, von Arx G, Petit G (2018). Xylem anatomical adjustments prioritize hydraulic efficiency over safety as Norway spruce trees grow taller. Tree Physiology, 38, 1088-1097. |
| [87] | Preston JC, Sandve SR (2013). Adaptation to seasonality and the winter freeze. Frontiers in Plant Science, 4, 167. DOI: 10.3389/fpls.2013.00167. |
| [88] | Rasmussen DH, AP DH (1972). Effect of solute on ice-solution interfacial free energy; calculation from measured homogeneous nucleation temperatures. In: Jellinek HHG ed. Water Structure at the Water-polymer Interface. Springer, Boston. 126-145. |
| [89] | Reich PB (2014). The world-wide “fast-slow” plant economics spectrum: A traits manifesto. Journal of Ecology, 102, 275-301. |
| [90] | Reich PB, Sendall KM, Stefanski A, Wei XR, Rich RL, Montgomery RA (2016). Boreal and temperate trees show strong acclimation of respiration to warming. Nature, 531, 633-636. |
| [91] | Reyes-Díaz M, Ulloa N, Zúñiga-Feest A, Gutiérrez A, Gidekel M, Alberdi M, Corcuera LJ, Bravo LA (2006). Arabidopsis thaliana avoids freezing by supercooling. Journal of Experimental Botany, 57, 3687-3696. |
| [92] | Rorato DG, Araujo MM, Tabaldi LA, Turchetto F, Griebeler AM, Berghetti ÁLP, Barbosa FM (2018). Tolerance and resilience of forest species to frost in restoration planting in southern Brazil. Restoration Ecology, 26, 537-542. |
| [93] | Salleo S, Lo Gullo MA, Trifilò P, Nardini A (2004). New evidence for a role of vessel-associated cells and phloem in the rapid xylem refilling of cavitated stems of Laurus nobilis L. Plant, Cell & Environment, 27, 1065-1076. |
| [94] | Santiago LS, De Guzman ME, Baraloto C, Vogenberg JE, Brodie M, Hérault B, Fortunel C, Bonal D (2018). Coordination and trade-offs among hydraulic safety, efficiency and drought avoidance traits in Amazonian rainforest canopy tree species. New Phytologist, 218, 1015-1024. |
| [95] | Schreiber SG, Hamann A, Hacke UG, Thomas BR (2013). Sixteen years of winter stress: An assessment of cold hardiness, growth performance and survival of hybrid poplar clones at a boreal planting site. Plant, Cell & Environment, 36, 419-428. |
| [96] | Sevanto S, Suni T, Pumpanen J, Grönholm T, Kolari P, Nikinmaa E, Hari P, Vesala T (2006). Wintertime photosynthesis and water uptake in a boreal forest. Tree Physiology, 26, 749-757. |
| [97] | Sierra-Almeida A, Reyes-Bahamonde C, Cavieres LA (2016). Drought increases the freezing resistance of high-elevation plants of the Central Chilean Andes. Oecologia, 181, 1011-1023. |
| [98] | Sperling O, Secchi F, Godfrey J, Zwieniecki MA (2017). Acclimation of Pistacia integerrima trees to frost in semi-arid environments depends on autumn’s drought. Planta, 245, 671-679. |
| [99] | Sperry JS, Hacke UG, Pittermann J (2006). Size and function in conifer tracheids and angiosperm vessels. American Journal of Botany, 93, 1490-1500. |
| [100] | Sperry JS, Love DM (2015). What plant hydraulics can tell us about responses to climate-change droughts. New Phytologist, 207, 14-27. |
| [101] | Sperry JS, Sullivan JEM (1992). Xylem embolism in response to freeze-thaw cycles and water stress in ring-porous, diffuse-porous, and conifer species. Plant Physiology, 100, 605-613. |
| [102] | Stiller V, Sperry JS (2002). Cavitation fatigue and its reversal in sunflower ( Helianthus annuus L.). Journal of Experimental Botany, 53, 1155-1161. |
| [103] | Taneda H, Tateno M (2005). Hydraulic conductivity, photosynthesis and leaf water balance in six evergreen woody species from fall to winter. Tree Physiology, 25, 299-306. |
| [104] | Toca A, Oliet JA, Villar-Salvador P, Maroto J, Jacobs DF (2018). Species ecology determines the role of nitrogen nutrition in the frost tolerance of pine seedlings. Tree Physiology, 38, 96-108. |
| [105] | Tyree MT, Ewers FW (1991). The hydraulic architecture of trees and other woody plants. New Phytologist, 119, 345-360. |
| [106] | Venturas MD, Sperry JS, Hacke UG (2017). Plant xylem hydraulics: What we understand, current research, and future challenges. Journal of Integrative Plant Biology, 59, 356-389. |
| [107] | Verhoeven A, García-Plazaola JI, Fernández-Marín B (2018). Shared mechanisms of photoprotection in photosynthetic organisms tolerant to desiccation or to low temperature. Environmental and Experimental Botany, 154, 66-79. |
| [108] | Vitasse Y, Lenz A, Körner C (2014). The interaction between freezing tolerance and phenology in temperate deciduous trees. Frontiers in Plant Science, 5, 541. DOI: 10.3389/fpls.2014.00541. |
| [109] | Walter J, Jentsch A, Beierkuhnlein C, Kreyling J (2013). Ecological stress memory and cross stress tolerance in plants in the face of climate extremes. Environmental and Experimental Botany, 94, 3-8. |
| [110] | Wang AY, Han SJ, Zhang JH, Wang M, Yin XH, Fang LD, Yang D, Hao GY (2018). The interaction between nonstructural carbohydrate reserves and xylem hydraulics in Korean pine trees across an altitudinal gradient. Tree Physiology, 38, 1792-1804. |
| [111] | Westhoff M, Schneider H, Zimmermann D, Mimietz S, Stinzing A, Wegner LH, Kaiser W, Krohne G, Shirley S, Jakob P, Bamberg E, Bentrup FW, Zimmermann U (2008). The mechanisms of refilling of xylem conduits and bleeding of tall birch during spring. Plant Biology, 10, 604-623. |
| [112] | Willick IR, Gusta LV, Fowler DB, Tanino KK (2019). Ice segregation in the crown of winter cereals: Evidence for extraorgan and extratissue freezing. Plant, Cell & Environment, 42, 701-716. |
| [113] | Willick IR, Takahashi D, Fowler DB, Uemura M, Tanino KK (2018). Tissue-specific changes in apoplastic proteins and cell wall structure during cold acclimation of winter wheat crowns. Journal of Experimental Botany, 69, 1221-1234. |
| [114] | Willson CJ, Jackson RB (2006). Xylem cavitation caused by drought and freezing stress in four co-occurring Juniperus species. Physiologia Plantarum, 127, 374-382. |
| [115] | Yin XH, Hao GY (2018). Divergence between ring- and diffuse-porous wood types in broadleaf trees of Changbai Mountains results in substantial differences in hydraulic traits. Chinese Journal of Applied Ecology, 29, 352-360. |
| [ 殷笑寒, 郝广友 (2018). 长白山阔叶树种木质部环孔和散孔结构特征的分化导致其水力学性状的显著差异. 应用生态学报, 29, 352-360.] | |
| [116] | Yin XH, Sterck F, Hao GY (2018). Divergent hydraulic strategies to cope with freezing in co-occurring temperate tree species with special reference to root and stem pressure generation. New Phytologist, 219, 530-541. |
| [117] | Zanne AE, Tank DC, Cornwell WK, Eastman JM, Smith SA, FitzJohn RG, McGlinn DJ, O’Meara BC, Moles AT, Reich PB, Royer DL, Soltis DE, Stevens PF, Westoby M, Wright IJ, Aarssen L, Bertin RI, Calaminus A, Govaerts R, Hemmings F, Leishman MR, Oleksyn J, Soltis PS, Swenson NG, Warman L, Beaulieu JM (2014). Three keys to the radiation of angiosperms into freezing environments. Nature, 506, 89-92. |
| [118] | Zhang HY, Wang CK, Wang XC (2013). Comparison of concentrations of non-structural carbohydrates between new twigs and old branches for 12 temperate species. Acta Ecologica Sinica, 33, 5675-5685. |
| [ 张海燕, 王传宽, 王兴昌 (2013). 温带12个树种新老树枝非结构性碳水化合物浓度比较. 生态学报, 33, 5675-5685.] | |
| [119] | Zhang W, Feng F, Tyree MT (2018). Seasonality of cavitation and frost fatigue in Acer mono Maxim. Plant, Cell & Environment, 41, 1278-1286. |
| [120] | Zhang YJ, Bucci SJ, Arias NS, Scholz FG, Hao GY, Cao KF, Goldstein G (2016). Freezing resistance in Patagonian woody shrubs: The role of cell wall elasticity and stem vessel size. Tree Physiology, 36, 1007-1018. |
| [121] | Zhu SD, Li RH, He PC, Siddiq Z, Cao KF, Ye Q (2019). Large branch and leaf hydraulic safety margins in subtropical evergreen broadleaved forest. Tree Physiology, 39, 1405-1415. |
| [122] | Zimmermann MH (1983). Xylem Structure and the Ascent of Sap. Spring, Berlin. |
| [1] | SUN Jia-Hui, SHI Hai-Lan, CHEN Ke-Yu, JI Bao-Ming, ZHANG Jing. Research advances on trade-off relationships of plant fine root functional traits [J]. Chin J Plant Ecol, 2023, 47(8): 1055-1070. |
| [2] | WANG Jia-Yi, WANG Xiang-Ping, XU Cheng-Yang, XIA Xin-Li, XIE Zong-Qiang, FENG Fei, FAN Da-Yong. Response of hydraulic architecture in Fraxinus velutina street trees to the percentage of impervious pavement in Beijing [J]. Chin J Plant Ecol, 2023, 47(7): 998-1009. |
| [3] | LI Yao-Qi, WANG Zhi-Heng. Functional biogeography of plants: research progresses and challenges [J]. Chin J Plant Ecol, 2023, 47(2): 145-169. |
| [4] | HE Lu-Lu, ZHANG Xuan, ZHANG Yu-Wen, WANG Xiao-Xia, LIU Ya-Dong, LIU Yan, FAN Zi-Ying, HE Yuan-Yang, XI Ben-Ye, DUAN Jie. Crown characteristics and its relationship with tree growth on different slope aspects for Larix olgensis var. changbaiensis plantation in eastern Liaoning mountainous area, China [J]. Chin J Plant Ecol, 2023, 47(11): 1523-1539. |
| [5] | WU Min, TIAN Yu, FAN Da-Yong, ZHANG Xiang-Xue. Hydraulic regulation of Populus tomentosa and Acer truncatum under drought stress [J]. Chin J Plant Ecol, 2022, 46(9): 1086-1097. |
| [6] | ZHAI Jiang-Wei, LIN Xin-Hui, WU Rui-Zhe, XU Yi-Xin, JIN Hao-Hao, JIN Guang-Ze, LIU Zhi-Li. Trade-offs between petiole and lamina of different functional plants in Xiao Hinggan Mountains, China [J]. Chin J Plant Ecol, 2022, 46(6): 700-711. |
| [7] | CHENG Si-Qi, JIANG Feng, JIN Guang-Ze. Leaf economics spectrum of broadleaved seedlings and its relationship with defense traits in a temperate forest [J]. Chin J Plant Ecol, 2022, 46(6): 678-686. |
| [8] | HAN Xu-Li, ZHAO Ming-Shui, WANG Zhong-Yuan, YE Lin-Feng, LU Shi-Tong, CHEN Sen, LI Yan, XIE Jiang-Bo. Adaptation of xylem structure and function of three gymnosperms to different habitats [J]. Chin J Plant Ecol, 2022, 46(4): 440-450. |
| [9] | QIN Hui-Jun, JIAO Liang, ZHOU Yi, XUE Ru-Hong, QI Chang-Liang, DU Da-Shi. Effects of altitudes on non-structural carbohydrate allocation in different dominate trees in Qilian Mountains, China [J]. Chin J Plant Ecol, 2022, 46(2): 208-219. |
| [10] | DAI Yuan-Meng, LI Man-Le, XU Ming-Ze, TIAN Yun, ZHAO Hong-Xian, GAO Sheng-Jie, HAO Shao-Rong, LIU Peng, JIA Xin, ZHA Tian-Shan. Leaf traits of Artemisia ordosica at different dune fixation stages in Mau Us Sandy Land [J]. Chin J Plant Ecol, 2022, 46(11): 1376-1387. |
| [11] | FANG Jing, YE Lin-Feng, CHEN Sen, LU Shi-Tong, PAN Tian-Tian, XIE Jiang-Bo, LI Yan, WANG Zhong-Yuan. Differences in anatomical structure and hydraulic function of xylem in branches of angiosperms in field and garden habitats [J]. Chin J Plant Ecol, 2021, 45(6): 650-658. |
| [12] | NI Ming-Yuan, ARITSARA Amy Ny Aina, WANG Yong-Qiang, HUANG Dong-Liu, XIANG Wei, WAN Chun-Yan, ZHU Shi-Dan. Analysis of xylem anatomy and function of representative tree species in a mixed evergreen and deciduous broad-leaved forest of mid-subtropical karst region [J]. Chin J Plant Ecol, 2021, 45(4): 394-403. |
| [13] | WANG Zhao-Ying, CHEN Xiao-Ping, CHENG Ying, WANG Man-Tang, ZHONG Quan-Lin, LI Man, CHENG Dong-Liang. Leaf and fine root economics spectrum across 49 woody plant species in Wuyi Mountains [J]. Chin J Plant Ecol, 2021, 45(3): 242-252. |
| [14] | TAN Yi-Bo, TIAN Hong-Deng, ZENG Chun-Yang, SHEN Hao, SHEN Wen-Hui, YE Jian-Ping, GAN Guo-Juan. Canopy mechanical abrasion between adjacent plants influences twig and leaf traits of Tsuga chinensis assemblage in the Mao’er Mountain [J]. Chin J Plant Ecol, 2021, 45(12): 1281-1291. |
| [15] | LI Hao, MA Ru-Yu, QIANG Bo, HE Cong, HAN Lu, WANG Hai-Zhen. Effect of current-year twig stem configuration on the leaf display efficiency of Populus euphratica [J]. Chin J Plant Ecol, 2021, 45(11): 1251-1262. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2022 Chinese Journal of Plant Ecology
Tel: 010-62836134, 62836138, E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
