

植物生态学报 ›› 2016, Vol. 40 ›› Issue (9): 933-941.DOI: 10.17521/cjpe.2015.0261
收稿日期:2015-07-08
接受日期:2016-04-25
出版日期:2016-09-10
发布日期:2016-09-29
通讯作者:
孙雪
基金资助:
Jia SHEN, Ya-He LI, Lin ZHANG, Xue SUN*( )
)
Received:2015-07-08
Accepted:2016-04-25
Online:2016-09-10
Published:2016-09-29
Contact:
Xue SUN
摘要:
为了探讨光照强度和CO2浓度对蛋白核小球藻(Chlorella pyrenoidosa)生长、无机碳利用的复合效应, 丰富绿藻中无机碳浓缩机制的资料, 该文设置两种光照强度(40和120 µmol photons•m-2•s-1)和两种CO2浓度(0.04%和0.16%)组合成4种条件, 比较了蛋白核小球藻生长、无机碳浓度、pH补偿点、光合放氧速率、碳酸酐酶(CA)活性和α-CA基因转录表达对这4种培养条件的响应。结果发现: 蛋白核小球藻在高光强高CO2浓度组生长最快; 低光强高CO2浓度组培养体系中总无机碳浓度为1163.3 µmol•L-1, 显著高于其他3组; 高光强低CO2浓度组藻的pH补偿点最高(9.8), 而低光强高CO2浓度组藻的pH补偿点最低(8.6); 低光强高CO2浓度组藻的最大光合速率(Vmax)和最大光合速率一半时的无机碳浓度(K0.5)最高, 分别是其他3组的1.28-1.91倍和1.61-2.00倍; 高光强低CO2浓度组藻的胞外CA活性最高; 而低光强低CO2浓度组藻的胞外α-CA基因表达量显著高于其他3组。以上结果表明低CO2浓度可促进蛋白核小球藻的pH补偿点和无机碳亲和力的提高, 诱导胞外CA活性及α-CA基因的表达; 该藻主要以HCO3-为无机碳源, 其对无机碳的利用受光照的调节。
沈佳, 李亚鹤, 张琳, 孙雪. 蛋白核小球藻生长和无机碳利用对不同光照强度和CO2浓度的响应. 植物生态学报, 2016, 40(9): 933-941. DOI: 10.17521/cjpe.2015.0261
Jia SHEN, Ya-He LI, Lin ZHANG, Xue SUN. Response of growth and inorganic carbon utilization to different light and CO2 levels in Chlorella pyrenoidosa. Chinese Journal of Plant Ecology, 2016, 40(9): 933-941. DOI: 10.17521/cjpe.2015.0261
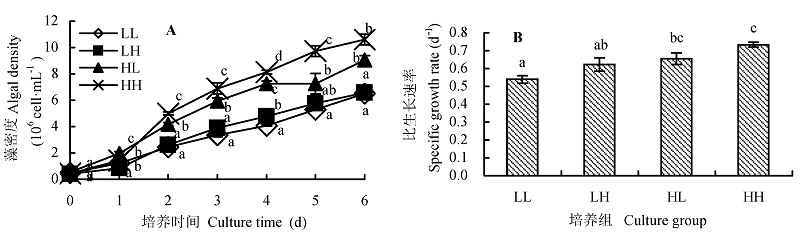
图1 不同光照强度和CO2浓度对蛋白核小球藻生长的影响(平均值±标准偏差)。A, 生长曲线。B, 比生长速率。LL、LH、HL和HH分别表示低光强低CO2浓度、低光强高CO2浓度、高光强低CO2浓度、高光强高CO2浓度条件。不同小写字母表示差异显著(p < 0.05)。
Fig. 1 Effect of different light intensity and CO2 concentration conditions on the growth of Chlorella pyrenoidosa (mean ± SD). A, Growth curve. B, Specific growth rate. LL, LH, HL and HH represent low-light intensity and low-CO2 concentration, low-light intensity and high-CO2 concentration, high-light intensity and low-CO2 concentration, high-light intensity and high-CO2 concentration conditions, respectively. Different lowercase letters indicate significant difference (p < 0.05).
| 处理组 Treatment group | 总碱度 Total alkalinity (µmol•L-1) | pH | DIC (µmol•L-1) | CO2 (µmol•L-1) | HCO3- (µmol•L-1) | CO32- (µmol•L-1) |
|---|---|---|---|---|---|---|
| LL | 1 387.8 ± 55.1b | 9.35 ± 0.00c | 765.3 ± 36.8ab | 0.22 ± 0.18a | 383.6 ± 18.44a | 381.5 ± 18.3c |
| LH | 1 388.4 ± 57.3b | 8.48 ± 0.03a | 1 163.3 ± 58.8c | 4.37 ± 0.46b | 1 022.1 ± 57.4c | 136.8 ± 3.6a |
| HL | 1 275.0 ± 16.5ab | 9.41 ± 0.04c | 665.8 ± 22.6a | 0.16 ± 0.44a | 311.7 ± 24.2a | 354.0 ± 5.8c |
| HH | 1 190.1 ± 0.0a | 8.95 ± 0.04b | 803.4 ± 17.2b | 0.84 ± 0.12a | 574.9 ± 27.2b | 227.6 ± 10.2b |
表1 不同光照强度和CO2浓度条件下蛋白核小球藻培养体系中各碳酸盐参数的比较(平均值±标准偏差, n = 3)
Table 1 Comparison of the parameters of carbonate system under different light intensity and CO2 concentration conditions in Chlorella pyrenoidosa (mean ± SD, n = 3)
| 处理组 Treatment group | 总碱度 Total alkalinity (µmol•L-1) | pH | DIC (µmol•L-1) | CO2 (µmol•L-1) | HCO3- (µmol•L-1) | CO32- (µmol•L-1) |
|---|---|---|---|---|---|---|
| LL | 1 387.8 ± 55.1b | 9.35 ± 0.00c | 765.3 ± 36.8ab | 0.22 ± 0.18a | 383.6 ± 18.44a | 381.5 ± 18.3c |
| LH | 1 388.4 ± 57.3b | 8.48 ± 0.03a | 1 163.3 ± 58.8c | 4.37 ± 0.46b | 1 022.1 ± 57.4c | 136.8 ± 3.6a |
| HL | 1 275.0 ± 16.5ab | 9.41 ± 0.04c | 665.8 ± 22.6a | 0.16 ± 0.44a | 311.7 ± 24.2a | 354.0 ± 5.8c |
| HH | 1 190.1 ± 0.0a | 8.95 ± 0.04b | 803.4 ± 17.2b | 0.84 ± 0.12a | 574.9 ± 27.2b | 227.6 ± 10.2b |
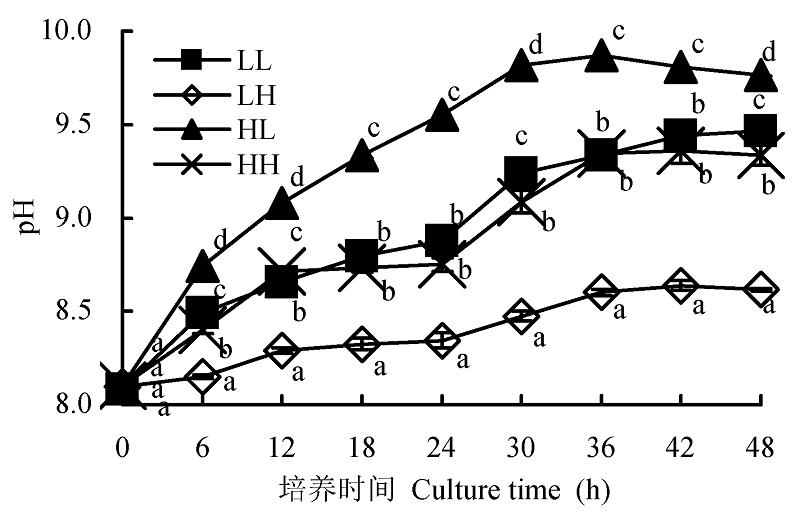
图2 不同光照强度和CO2浓度下蛋白核小球藻的pH补偿点(平均值±标准偏差)。LL、LH、HL和HH分别表示低光强低CO2浓度、低光强高CO2浓度、高光强低CO2浓度、高光强高CO2浓度条件。不同小写字母表示差异显著(p < 0.05)。
Fig. 2 Effect of different light intensity and CO2 concentration conditions on pH compensation point of Chlorella pyrenoidosa (mean ± SD). LL, LH, HL and HH represent low-light intensity and low-CO2 concentration, low-light intensity and high-CO2 concentration, high-light intensity and low-CO2 concentration, high-light intensity and high-CO2 concentration conditions, respectively. Different lowercase letters indicate significant difference (p < 0.05).
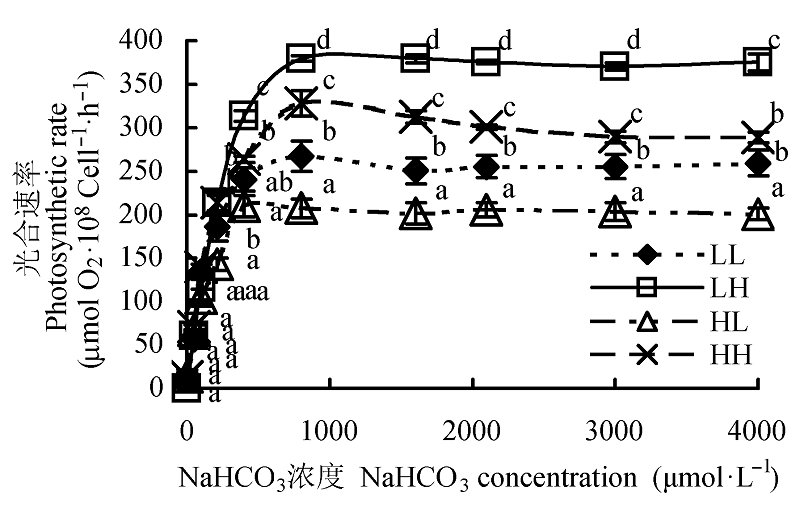
图3 不同光照强度和CO2浓度下蛋白核小球藻的P-C曲线(平均值±标准偏差)。LL、LH、HL和HH分别表示低光强低CO2浓度、低光强高CO2浓度、高光强低CO2浓度、高光强高CO2浓度条件。不同小写字母表示差异显著(p < 0.05)。
Fig. 3 Effect of different light intensity and CO2 concentration conditions on P-C curve of Chlorella pyrenoidosa (mean ± SD). LL, LH, HL and HH represent low-light intensity and low-CO2 concentration, low-light intensity and high-CO2 concentration, high-light intensity and low-CO2 concentration, high-light intensity and high-CO2 concentration conditions, respectively. Different lowercase letters indicate significant difference (p < 0.05).
| 处理组 Treatmeat group | Vmax (µmol O2• 108cell·h-1) | K0.5 (µmol•L-1) | ||
|---|---|---|---|---|
| DIC | CO2 | HCO3- | ||
| LL | 275.93 ± 16.83b | 107.20 ± 2.49a | 2.43 ± 0.06a | 104.78 ± 2.43a |
| LH | 415.44 ± 4.23d | 188.33 ± 2.04b | 4.26 ± 0.45b | 184.07 ± 19.59b |
| HL | 218.03 ± 10.16a | 94.20 ± 7.03a | 2.13 ± 0.16a | 92.07 ± 6.87a |
| HH | 324.05 ± 1.34c | 116.83 ± 6.89a | 2.64 ± 0.16a | 114.19 ± 6.73a |
表2 不同光照强度和CO2浓度下蛋白核小球藻的Vmax和K0.5 (平均值±标准偏差, n = 3)
Table 2 Effect of different light intensity and CO2 concentration conditions on Vmax and K0.5 of Chlorella pyrenoidosa (mean ± SD, n = 3)
| 处理组 Treatmeat group | Vmax (µmol O2• 108cell·h-1) | K0.5 (µmol•L-1) | ||
|---|---|---|---|---|
| DIC | CO2 | HCO3- | ||
| LL | 275.93 ± 16.83b | 107.20 ± 2.49a | 2.43 ± 0.06a | 104.78 ± 2.43a |
| LH | 415.44 ± 4.23d | 188.33 ± 2.04b | 4.26 ± 0.45b | 184.07 ± 19.59b |
| HL | 218.03 ± 10.16a | 94.20 ± 7.03a | 2.13 ± 0.16a | 92.07 ± 6.87a |
| HH | 324.05 ± 1.34c | 116.83 ± 6.89a | 2.64 ± 0.16a | 114.19 ± 6.73a |
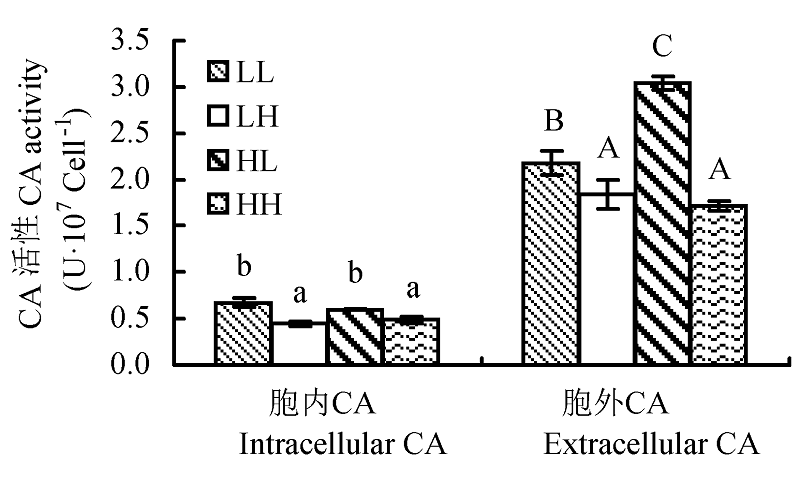
图4 不同光照强度和CO2浓度下蛋白核小球藻的碳酸酐酶(CA)活性(平均值±标准偏差)。LL、LH、HL和HH分别表示低光强低CO2浓度、低光强高CO2浓度、高光强低CO2浓度、高光强高CO2浓度条件。不同字母表示差异显著(p < 0.05)。
Fig. 4 Effect of different light intensity and CO2 concentration conditions on carbonic anhydrase (CA) activity of Chlorella pyrenoidosa (mean ± SD). LL, LH, HL and HH represent low-light intensity and low-CO2 concentration, low-light intensity and high-CO2 concentration, high-light intensity and low-CO2 concentration, high-light intensity and high-CO2 concentration conditions, respectively. Different letters indicate significant difference (p < 0.05).
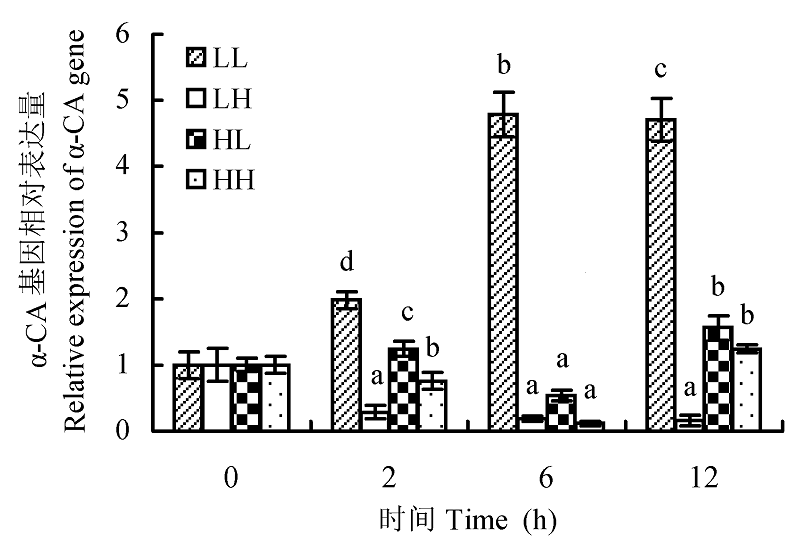
图5 不同光照强度和CO2浓度对蛋白核小球藻α-CA基因表达的影响(平均值±标准偏差)。LL、LH、HL和HH分别表示低光强低CO2浓度、低光强高CO2浓度、高光强低CO2浓度、高光强高CO2浓度条件。不同小写字母表示差异显著(p < 0.05)。
Fig. 5 The effect of different light intensity and CO2 concentration conditions on α-CA gene expression of Chlorella pyrenoidosa (mean ± SD). LL, LH, HL and HH represent low-light intensity and low-CO2 concentration, low-light intensity and high-CO2 concentration, high-light intensity and low-CO2 concentration, high-light intensity and high-CO2 concentration conditions, respectively. Different lowercase letters indicate significant difference (p < 0.05).
| 1 | Badger MR (1980). Internal inorganic carbon pool of Chlamydomonas reinhardtii: Evidence for a carbon dioxide- concentrating mechanism.Plant Physiology, 66, 407-413. |
| 2 | Badger MR (1987). The CO2-concentrating mechanism in aquatic phototrophs. In: Hatch MD, Boardman NK eds. The Biochemistry of Plants: A Comprehensive Treatise, Vol 10, Photosynthesis. Academic Press, San Diego, USA. 219-274. |
| 3 | Bozzo GG, Colman B, Matsuda Y (2000). Active transport of CO2 and bicarbonate is induced in response to external CO2 concentration in the green alga Chlorella kessleri.Journal of Experimental Botany, 51, 1341-1348. |
| 4 | Chen XW, Gao KS (2003). Effect of CO2 concentrations on the activity of photosynthetic CO2 fixation and extracellular carbonic anhydrase in the marine diatom Skeletonema costatum.Chinese Science Bulletin, 48, 2275-2279. (in Chinese with English abstract)[陈雄文, 高坤山 (2003). CO2浓度对中肋骨条藻的光合无机碳吸收和胞外碳酸酐酶活性的影响. 科学通报, 48, 2275-2279.] |
| 5 | Elrad D, Niyogi KK, Grossman AR (2002). A major light- harvesting polypeptide of photosystem II functions in thermal dissipation.Plant Cell, 14, 1801-1816. |
| 6 | Gao K, Aruga Y, Asada K, Kiyohara M (1993). Influence of enhanced CO2 on growth and photosynthesis of the red algae Gracilaria sp. and G. chilensis.Journal of Applied Phycology, 5, 563-571. |
| 7 | Gao KS (1999). Research techniques and methods in characterizing photosynthetic carbon fixation by algae.Marine Sciences, 23, 37-41. (in Chinese with English abstract)[高坤山 (1999). 藻类光合固碳的研究技术与解析方法. 海洋科学, 23, 37-41.] |
| 8 | Goyal A, Shiraiwa Y, Husic HD, Tolbert NE (1992). External and internal carbonic anhydrases in Dunaliella species.Marine Biology, 113, 349-355. |
| 9 | Guillard RRL, Ryther JH (1962). Studies of marine planktonic diatoms: I. Cyclotella nana Hustedt, and Detonula confer- vacea (Cleve) Gran.Canadian Journal of Microbiology, 8, 229-240. |
| 10 | Harada H, Nakatsuma D, Ishida M, Matsuda Y (2005). Regulation of the expression of intracellular β-carbonic anhydrase in response to CO2 and light in the marine diatom Phaeodactylum tricornutum.Plant Physiology, 139, 1041-1050. |
| 11 | Hu HH, Gao KS (2003). Optimization of growth and fatty acid composition of a unicellular marine picoplankton, Nanno chloropsis sp., with enriched carbon sources.Biotechnology Letters, 25, 421-425. |
| 12 | Kucho K, Yoshioka S, Taniguchi F, Ohyama K, Fukuzawa H (2003). Cis-acting elements and DNA-binding proteins involved in CO2-responsive transcriptional activation of Cah1 encoding a periplasmic carbonic anhydrase in Chlamydomonas reinhardtii.Plant Physiology, 133, 783-793. |
| 13 | Li L, Fu ML, Zhao YH, Zhu YT (2012). Characterization of carbonic anhydrase II from Chlorella vulgaris in bio-CO2 capture.Environmental Science and Pollution Research, 19, 4227-4232. |
| 14 | Li N, Bi YH, Gao DW, Hu ZY, Ren NQ (2011). Effects of elevated CO2 concentration on growth of microcystis aeruginosa.Acta Hydrobiologica Sinica, 35, 698-702. (in Chinese with English abstract)[李娜, 毕永红, 高大文, 胡征宇, 任南琪 (2011). 大气CO2浓度变化对铜绿微囊藻生长的影响. 水生生物学报, 35, 698-702.] |
| 15 | Livak KJ, Schmittgen TD (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method.Methods, 25, 402-408. |
| 16 | Maberly SC (1990). Exogenous sources of inorganic carbon for photosynthesis by marine macroalgae.Journal of Phycology, 26, 439-499. |
| 17 | Millero FJ, Graham TB, Huang F, Bustos-Serrano H, Pierrot D (2006). Dissociation constants of carbonic acid in seawater as a function of salinity and temperature.Marine Chemistry, 100, 80-94. |
| 18 | Miura K, Kohinata T, Yoshioka S, Ohyama K, Fukuzawa H (2002). Regulation of a carbon concentrating mechanism through CCM1 in Chlamydomonas reinhardtii.Functional Plant Biology, 29, 211-219. |
| 19 | Moroney JV, Ynalvez RA (2007). Proposed carbon dioxide concentrating mechanism in Chlamydomonas reinhardtii.Eukaryotic Cell, 6, 1251-1259. |
| 20 | Moroney JV, Ma Y, Frey WD, Fusilier KA, Pham TT, Simms TA, DiMario RJ, Yang J, Mukherjee B (2011). The car- bonic anhydrase isoforms of Chlamydomonas reinhardtii: Intracellular location, expression, and physiological roles.Photosynthesis Research, 109, 133-149. |
| 21 | Ochiai T, Colman B, Matsuda Y (2007). Acclimation of wild-type cells and CO2-insensitive mutants of the green alga Chlorella ellipsoidea to elevated [CO2].Plant, Cell & Environment, 30, 944-951. |
| 22 | Raven JA (2010). Inorganic carbon acquisition by eukaryotic algae: Four current questions.Photosynthesis Research, 106, 123-134. |
| 23 | Tachibana M, Allen AE, Kikutani S, Endo Y, Bowler C, Matsuda Y (2011). Localization of putative carbonic anhy- drases in two marine diatoms, Phaeodactylum tricornutum and Thalassiosira pseudonana.Photosynthesis Research, 109, 205-221. |
| 24 | Wang SG, Yang R, Zhou XQ, Song DD, Sun X, Luo QJ (2013). Utilization of inorganic carbon in Pyropia haitanensis (Rhodophyta) under heat stress.Oceanologia et Limnologia Sinica, 44, 1378-1385. (in Chinese with English abstract)[王淑刚, 杨锐, 周新倩, 宋丹丹, 孙雪, 骆其君 (2013). 高温胁迫下坛紫菜(Pyropia haitanensis)对无机碳的利用. 海洋与湖沼, 44, 1378-1385.] |
| 25 | Wang SS, Liu YD, Zou YD, Li DH (2006). Modulation and adaptation of carbonic anhydrase activity in Microcystis spp. under different environmental factors.Acta Ecologica Sinica, 26, 2443-2448. (in Chinese with English abstract)[王山杉, 刘永定, 邹永东, 李敦海 (2006). 微囊藻碳酸酐酶活性在不同环境因素下的调节与适应. 生态学报, 26, 2443-2448.] |
| 26 | Wang WW, Sun X, Wang DM, Shen J, Xu NJ (2014). Effects of salinity and inorganic carbon on the growth, extracellu- lar carbonic anhydrase activity and ca gene expression of Chlorella pyrenoidosa.Journal of Fisheries of China, 38, 920-928. (in Chinese with English abstract)[王玮蔚, 孙雪, 王冬梅, 沈佳, 徐年军 (2014). 盐度和无机碳对蛋白核小球藻生长、胞外碳酸酐酶活性及其基因表达的影响. 水产学报, 38, 920-928.] |
| 27 | Wang Y, Spalding MH (2006). An inorganic carbon transport system responsible for acclimation specific to air levels of CO2 in Chlamydomonas reinhardtii.Proceedings of the National Academy of Sciences of the United States of America, 103, 10110-10115. |
| 28 | Wang Y, Sun Z, Horken KM, Im CS, Xiang YB, Grossman, Arthur R, Weeks DP (2005). Analyses of CIA5, the master regulator of the carbon-concentrating mechanism in Chlamydomonas reinhardtii, and its control of gene expression.Canadian Journal of Botany, 83, 765-779. |
| 29 | Wilbur KM, Anderson NG (1948). Electrometric and colori- metric determination of carbonic anhydrase.The Journal of Biological Chemistry, 176, 147-154. |
| 30 | Wu Y, Gao K, Riebesell U (2010). CO2 induced seawater acidification affects physiological performance of the marine diatom Phaeodactylyum tricornutum.Biogeosciences, 7, 2915-2923. |
| 31 | Xia JR, Gao KS (2001). Effects of high CO2 concentration on growth and photosynthesis of Spirulina maxima.Acta Hydrobiologica Sinica, 25, 474-480. (in Chinese with English abstract)[夏建荣, 高坤山 (2001). 高浓度CO2对极大螺旋藻生长和光合作用的影响. 水生生物学报, 25, 474-480.] |
| 32 | Xia JR, Gao KS (2002). Advances in research on CO2 con- centrating mechanism of green algae.Chinese Journal of Applied Ecology, 11, 1507-1510. (in Chinese with English abstract)[夏建荣, 高坤山 (2002). 绿藻CO2浓缩机制的研究进展. 应用生态学报, 11, 1507-1510.] |
| 33 | Xia JR, Yu JL (2009). Effects of high CO2 concentration on ex- tracellular carbonic anhydrase activity and photosynthesis in Nitzschia closterium var. minutissima. Journal of Guangzhou University (Natural Science Edition), 8, 49-53. (in Chinese with English abstract)[夏建荣, 余锦兰 (2009). 高浓度CO2对小新月菱形藻胞外碳酸酐酶活性和光合作用的影响. 广州大学学报(自然科学版), 8, 49-53.] |
| 34 | Yamano T, Miura K, Fukuzawa H (2008). Expression analysis of genes associated with the induction of the carbon- concentrating mechanism in Chlamydomonas reinhardtii.Plant Physiology, 147, 340-354. |
| 35 | Yoshioka S, Taniguchi F, Miura K, Inoue T, Yamano T, Fukuzawa H (2004). The novel Myb transcription factor LCR1 regulates the CO2-responsive gene Cah1, encoding a periplasmic carbonic anhydrase in Chlamydomonas reinhardtii.Plant Cell, 16, 1466-1477. |
| [1] | 王晶苑, 魏杰, 温学发. 土壤CO2通量梯度观测技术和方法的理论、假设与应用进展[J]. 植物生态学报, 2022, 46(12): 1523-1536. |
| [2] | 郑周涛, 张扬建. 1982-2018年青藏高原水分利用效率变化及归因分析[J]. 植物生态学报, 2022, 46(12): 1486-1496. |
| [3] | 叶子飘, 于冯, 安婷, 王复标, 康华靖. 植物气孔导度对CO2响应模型的构建[J]. 植物生态学报, 2021, 45(4): 420-428. |
| [4] | 牛书丽, 陈卫楠. 全球变化与生态系统研究现状与展望[J]. 植物生态学报, 2020, 44(5): 449-460. |
| [5] | 冯兆忠, 李品, 张国友, 李征珍, 平琴, 彭金龙, 刘硕. 二氧化碳浓度升高对陆地生态系统的影响: 问题与展望[J]. 植物生态学报, 2020, 44(5): 461-474. |
| [6] | 韩玲, 赵成章, 冯威, 徐婷, 郑慧玲, 段贝贝. 张掖湿地芨芨草叶脉密度和叶脉直径的权衡关系对3种生境的响应[J]. 植物生态学报, 2017, 41(8): 872-881. |
| [7] | 刘波, 吕宪国, 姜明, 张文广, 武海涛. 光照、水深交互作用对松嫩湿地芦苇种子萌发的影响[J]. 植物生态学报, 2015, 39(6): 616-620. |
| [8] | 胡文海, 张斯斯, 肖宜安, 闫小红. 两种杜鹃花属植物对长期遮阴后全光照环境的生理响应及其光保护机制[J]. 植物生态学报, 2015, 39(11): 1093-1100. |
| [9] | 冯汉青, 管东东, 焦青松, 贾凌云, 孙坤. 光照下菜豆叶片抗氰呼吸与光合作用关系的分析[J]. 植物生态学报, 2015, 39(1): 104-109. |
| [10] | 熊慧, 马承恩, 李乐, 曾辉, 郭大立. 不同生境条件下蕨类和被子植物的气孔形态特征及其对光强变化的响应[J]. 植物生态学报, 2014, 38(8): 868-877. |
| [11] | 杨春, 谭太龙, 余佳玲, 廖琼, 张晓龙, 张振华, 宋海星, 官春云. 大气CO2浓度倍增对油菜韧皮部汁液成分及根部氮素积累的影响[J]. 植物生态学报, 2014, 38(7): 776-784. |
| [12] | 康华靖, 陶月良, 权伟, 王伟, 欧阳竹. 植物光合CO2响应模型对光下(暗)呼吸速率拟合的探讨[J]. 植物生态学报, 2014, 38(12): 1356-1363. |
| [13] | 龙凤玲, 李义勇, 方熊, 黄文娟, 刘双娥, 刘菊秀. 大气CO2浓度上升和氮添加对南亚热带模拟森林生态系统土壤碳稳定性的影响[J]. 植物生态学报, 2014, 38(10): 1053-1063. |
| [14] | 孟凡超, 张佳华, 姚凤梅. CO2浓度升高和降水增加协同作用对玉米产量及生长发育的影响[J]. 植物生态学报, 2014, 38(10): 1064-1073. |
| [15] | 唐钢梁, 李向义, 林丽莎, 李磊, 鲁建荣. 骆驼刺在不同遮阴下的水分状况变化及其生理响应[J]. 植物生态学报, 2013, 37(4): 354-364. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2026 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
![]()