

植物生态学报 ›› 2007, Vol. 31 ›› Issue (4): 599-606.DOI: 10.17521/cjpe.2007.0076
蔡颖1,2, 关保华1,2, 安树青1,2,*( ), 申瑞玲2, 蒋金辉2, 董蕾3
), 申瑞玲2, 蒋金辉2, 董蕾3
收稿日期:2006-07-19
接受日期:2007-01-05
出版日期:2007-07-19
发布日期:2007-07-30
通讯作者:
安树青
作者简介:* E-mail: anshq@nju.edu.cn基金资助:
CAI Ying1,2, GUAN Bao-Hua1,2, AN Shu-Qing1,2,*( ), SHEN Rui-Ling2, JIANG Jin-Hui2, DONG Lei3
), SHEN Rui-Ling2, JIANG Jin-Hui2, DONG Lei3
Received:2006-07-19
Accepted:2007-01-05
Online:2007-07-19
Published:2007-07-30
Contact:
AN Shu-Qing
摘要:
植物,尤其是克隆植物,能够通过表型变化来缓解外界压力,提高对环境的适应能力。该文研究了水生克隆植物乌菱(Trapa bicornis)对底泥磷含量(Sediment phosphorus concentration, SP)、植株密度(Plant density, PD) 及两者间交互作用的可塑性响应,探讨可塑性是否能促进其在富营养化环境中的生长。结果显示,底泥磷含量对乌菱的主菱盘叶数、同化根比根长、吸收根比根长以及叶、茎、同化根、吸收根与植株总磷含量等都有显著影响 (p<0.05),而植株密度对乌菱各生长及生理生态参数均无显著作用 (p>0.05);SP与PD的交互作用弱化了底泥磷含量对乌菱的效应。底泥磷含量和植株密度甚至改变了同化根、吸收根、茎、叶与总生物量之间的异速生长关系。研究结果表明:乌菱的表型可塑性变化主要受底泥磷含量的影响,乌菱通过器官生物量分配、形态结构及生理生态特征的调整来响应底泥磷含量的变化;同时,高的植株密度也可以提高其在富营养化生境下的生态适应性。
蔡颖, 关保华, 安树青, 申瑞玲, 蒋金辉, 董蕾. 克隆植物乌菱对底泥磷含量及植株密度的表型可塑性响应. 植物生态学报, 2007, 31(4): 599-606. DOI: 10.17521/cjpe.2007.0076
CAI Ying, GUAN Bao-Hua, AN Shu-Qing, SHEN Rui-Ling, JIANG Jin-Hui, DONG Lei. PHENOTYPIC PLASTICITY OF THE CLONAL PLANT TRAPA BICORNIS IN RESPONSE TO SEDIMENT PHOSPHORUS CONCENTRATION AND PLANT DENSITY. Chinese Journal of Plant Ecology, 2007, 31(4): 599-606. DOI: 10.17521/cjpe.2007.0076
| 植株密度 Plant density (PD) | 底泥磷含量(SP) Sediment phosphorus concentration (SP) (mg·g-1) | ||
|---|---|---|---|
| 低磷SP Low SP (LSP) 27.56 ± 0.78 | 中磷SP Median SP (MSP) 52.85 ± 1.30 | 高磷SP High SP (HSP) 115.61 ± 2.72 | |
| 4株/箱4 individuals per container (PD4) | LSP-PD4 | MSP-PD4 | HSP-PD4 |
| 8株/箱8 individuals per container (PD8) | LSP-PD8 | MSP-PD8 | HSP-PD8 |
| 12株/箱12 individuals per container (PD12) | LSP-PD12 | MSP-PD12 | HSP-PD12 |
表1 底泥磷含量和植株密度设置
Table 1 Experimental setup of sediment phosphorus concentration and plant density
| 植株密度 Plant density (PD) | 底泥磷含量(SP) Sediment phosphorus concentration (SP) (mg·g-1) | ||
|---|---|---|---|
| 低磷SP Low SP (LSP) 27.56 ± 0.78 | 中磷SP Median SP (MSP) 52.85 ± 1.30 | 高磷SP High SP (HSP) 115.61 ± 2.72 | |
| 4株/箱4 individuals per container (PD4) | LSP-PD4 | MSP-PD4 | HSP-PD4 |
| 8株/箱8 individuals per container (PD8) | LSP-PD8 | MSP-PD8 | HSP-PD8 |
| 12株/箱12 individuals per container (PD12) | LSP-PD12 | MSP-PD12 | HSP-PD12 |
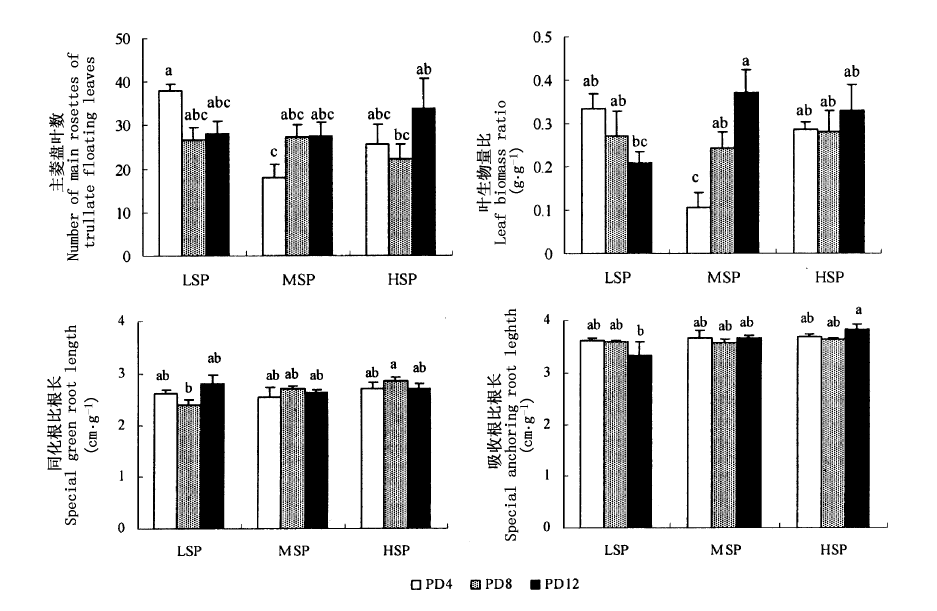
图1 乌菱的主要生长指标对底泥磷含量与植株密度的响应
Fig.1 The responses of the main growth variables of Trapa bicornis to sediment phosphorus concentration and plant density 平均值 ± 标准差Mean ± SE LSP:低底泥磷含量Low sediment phosphorus concentration MSP:中底泥磷含量Median sediment phosphorus concentration HSP:高底泥磷含量High sediment phosphorus concentration PD4:每箱4株4 individuals per container PD8:每箱8株8 individuals per container PD12:每箱12株12 individuals per container 图中上方不同的字母表示不同处理之间通过Duncan法进行多重检验得出的显著性差异 Different upper letters indicate significant differences between treatments according to the results of post-hoc with Duncan test
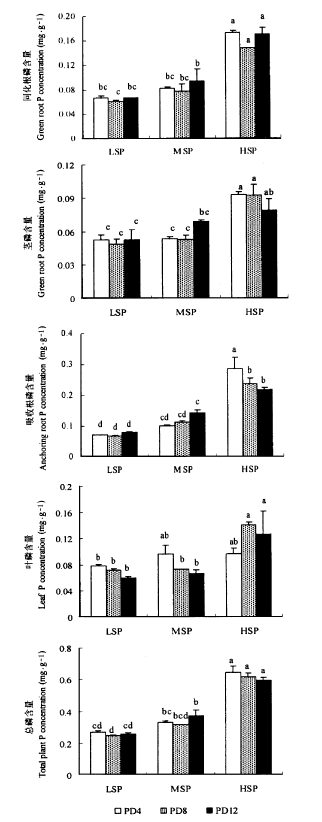
图2 乌菱磷含量对底泥磷含量和植株密度的响应 图注见图1
Fig.2 The responses of plant P concentration of Trapa bicornis to sediment phosphorus concentration and plant density Notes see Fig. 1

图3 乌菱生物量异速生长关系分析 图中排在横行的变量为x轴,纵行排列的为y轴,所有数据都在做相关性分析前采用以10为底数的对数处理。图的下方列出线性回归方程,随后为回归系数及相关显著性p,*: p<0.05; **: p<0.01; ***: p<0.001见图1
Fig.3 The mass-mass allometric relationships of Trapa bicornis Variables listed in the row were viewed as x-axis, while the variables listed in column were viewed as y-axis. Data were logged. Linear regression formulae are shown, followed with regression coefficients and statistical significance values of p. *: p<0.05; **: p<0.01; ***: p<0.001 LSP、MSP、HSP、PD4、PD8、PD12: See Fig. 1
| [1] | Agami M, Waisel Y (2002). Competitive relationships between two water plant species: Najas marina L. and Myriophyllum spicatum L. Najas marina L. and Myriophyllum spicatum L. Hydrobiologia, 482, 197-200. |
| [2] |
Agrawal A, Mohan HYR (1995). In vitro germination and micropropagation of water chestnut (Trapa sp.). Aquatic Botany, 51, 135-146.
DOI URL |
| [3] |
Busch J, Mendelssohn IA, Lorenzen B, Brix H, Miao S (2004). Growth responses of the everglades wet prairie species Eleocharis cellulosa and Rhynchospora tracyi to water level and phosphate availability. Aquatic Botany, 78, 37-54.
DOI URL |
| [4] |
Chapin FS Ⅲ (1980). The mineral nutrition of wild plants. Annual Review of Ecology and Systematics, 11, 233-260.
DOI URL |
| [5] |
Chen XY (2000). Effects of plant density and age on the mating system of Kandelia candel Druce (Rhizophoraceae), a viviparous mangrove species. Hydrobiologia, 432, 189-193.
DOI URL |
| [6] |
Cipollini DF, Bergelson J (2001). Plant density and nutrient availability constrain constitutive and wound-induced expression of trypsin inhibitors in Brassica napus. Journal of Chemical Ecology, 27, 593-610.
DOI URL |
| [7] | Ciro AR, Joao PTW, Silvelena V, Valdir JR (1999). The significance of root growth on cotton nutrition in an acidic low-P soil. Plant and Soil, 212, 185-190. |
| [8] | Dewitt TJ, Andrew S, David SW (1998). Costs and limits of phenotypic plasticity. Trees, 13(2), 77-81. |
| [9] | Dong M (董鸣) (1996). Plant clonal growth in heterogeneous habitats: risk-spreading. Acta Phytoecologica Sinica (植物生态学报), 20, 543-548. (in Chinese with English abstract) |
| [10] |
Foyer C, Spencer C (1986). The relationship between phosphate status and photosynthesis in leaves. Planta, 167, 369-375.
DOI URL PMID |
| [11] |
Fransen BH, Kroon D, Berendse F (1998). Root morphological plasticity and nutrient acquisition of perennial grass species from habitats of different nutrient availability. Oecologia, 115, 351-358.
DOI URL PMID |
| [12] |
Fransen B, Blijjenberg J, de Kroon H (1999). Root morphological and physiological plasticity of perennial grass species and the exploitation of spatial and temporal heterogeneous nutrient patches. Plant and Soil, 211, 179-189.
DOI URL |
| [13] |
Fredeen AL, Raab TK, Rao IM, Terry N (1990). Effects of phosphorus nutrition on photosynthesis in Glycine max (L.) Merr. Planta, 181, 399-405.
DOI URL PMID |
| [14] |
Geber MA, Watson MA (1997). Morphological plasticity in clonal plants: the foraging concept reconsidered. Journal of Ecology, 83, 143-152.
DOI URL |
| [15] |
Greulich S, Barrat-Segretain M, Bornette G (2001). Basal rosette or floating leaf canopy—an example of plasticity in a rare aquatic macrophyte. Hydrobiologia, 448, 53-59.
DOI URL |
| [16] |
Groth AG, Lovett-Doust L, Lovett-Doust J (1996). Population density and module demography in Trapa natans (Trapaceae), an annual, clonal aquatic macrophyte. American Journal of Botany, 83, 1406-1415.
DOI URL |
| [17] |
He WM, Dong M (2003). Plasticity in physiology and growth of Salix matsudana in response to simulated atmospheric temperature rise in the Mu Us Sandland. Photosynthetica, 41, 297-300.
DOI URL |
| [18] | Hunt R (1978). Plant Growth Analysis. Edwand Amold, Landon. |
| [19] | Hutchings MJ, de Kroon H (1994). Foraging in plants: the role of morphological plasticity in resource acquisition. Advances in Ecological Research, 25, 159-238. |
| [20] |
Idestam-Almquist J, Kautsky L (1995). Plastic responses in morphology of Potamogeton pectinatus L. to sediment conditions at two sites in the northern Baltic proper. Aquatic Botany, 52, 205-216.
DOI URL |
| [21] |
Jacob J, Lawlor DW (1992). Dependence of photosynthesis of sunflower and maize leaves on phosphate supply, ribulose-1, 5-bisphosphate carboxylase/oxygenase activity, and ribulose-1, 5-bisphosphate pool size. Plant Physiology, 98, 801-807.
DOI URL PMID |
| [22] | Jian YX (简永兴), Wang JB (王建波), He GQ (何国庆), Li J (李今), Chen JK (陈家宽) (2001). A comparative study of aquatic plant diversity of Haikou, Taibai and Wushan Lake in Hubei Province of China. Acta Ecologica Sinica (生态学报), 21, 1815-1824. (in Chinese with English abstract) |
| [23] |
Kondracka A, Rychter AM (1997). The role of P-irecycling processes during photosynthesis in phosphated efficient bean plants. Journal of Experimental Botany, 48, 1461-1468.
DOI URL |
| [24] |
Linkohr BI, Williamson LC, Fitter AH, Leyser HMO (2002). Nitrate and phosphate availability and distribution have different effects on root system architecture of Arabidopsis. Plant Journal, 29, 751-760.
DOI URL |
| [25] | Lorenzen B, Brix H, Mendelssohn IA, McKee KL, Miao S (2001). Gowth, biomass allocation and nutrientuse efficiency in Cladium jamaicense and Typha domingensis as affected by phosphorus and oxygen availability. Aquatic Botony, 70, 117-133. |
| [26] | Luther H (1983). On life forms, and above-ground and underground biomass of aquatic macrophytes. Acta Botanica Fennica, 123, 1-23. |
| [27] | Marschner H (1995). Mineral Nutrition of Higher Plants. Academic Press, London, 231-255. |
| [28] | Miao SL, DeBusk WF (1999). Effects of phosphorus enrichment on structure and function of sawgrass and cattail communities in the everglades. In: Reddy KR, O'Connor GA, Schelske CL eds. Phosphorus Biogeochemistry in Subtropical Ecosystems. CRC Press, Boca Raton, 275-299. |
| [29] |
Nagashima H, Terashima I, Katon S (1995). Effects of plant density on frequency distributions of plant height in Chenopodium album stands: analysis based on continuous monitoring of height-growth of individual plants. Annals of Botany, 75, 173-180.
DOI URL |
| [30] | Perez M, Duarte CM, Romero J, Sand-Jensen K, Alcoverro T (1994). Growth plasticity in Cymodocea nodosa stands: the importance of nutrient supply. Aquatic Botony, 47, 249-264. |
| [31] |
Plesnicar M, Kastori R, Petrovic N, Pankovic D (1994). Photosynthesis and chlorophyll fluorescence in sunflower (Helianthus-annus L.) leaves as affected by phosphorus nutrition. Journal of Experimental Botany, 45, 919-924.
DOI URL |
| [32] |
Robert WP (1999). Natural enemies of Trapa spp. in Northeast Asia and Europe. Biological Control, 14, 168-180.
DOI URL |
| [33] | Rosolem CA, Witacker JPT, Vanzolini S, Ramos VJ (1999). The significance of root growth on cotton nutrition in an acidic low-P soil. Plant and Soil, 212, 185-190. |
| [34] |
Ryser P, Lambers H (1995). Root and leaf attributes accounting for the performance of fast- and slow-growing grasses at different nutrient supply. Plant and Soil, 170, 251-265.
DOI URL |
| [35] | Sas H (1989). Lake Restoration by Reduction of Nutrient Loading: Expectations, Experiences, Extrapolations. Academia-Verlag Richarz, St. Augustin, Germany, 497. |
| [36] |
Schachtman DP, Reid RJ, Ayling SM (1998). Phosphorus uptake by plants: from soil to cell. Plant Physiology, 116, 447-453.
DOI URL PMID |
| [37] |
Schlichting CD (1986). The evolution of phenotypic plasticity in plants. Annual Review of Ecology and Systematics, 17, 667-693.
DOI URL |
| [38] |
Schroeder MS, Janos DP (2004). Phosphorus and intraspecific density alter plant responses to Arbuscular mycorrhizas. Plant and Soil, 264, 335-348.
DOI URL |
| [39] |
Spector T, Putz FE (2006). Biomechanical plasticity facilitates invasion of maritime forest in the southern USA by Brazilian pepper ( Schinus terebinthifolius). Biological Invasions, 8, 255-260.
DOI URL |
| [40] | Sultan SE (1987). Evolutionary implications of phenotypic plasticity in plant. Evolutionary Biology, 21, 127-176. |
| [41] |
Sultan SE (2001). Phenotypic plasticity for plant development, function and life history. Trends in Plant Science, 5, 537-542.
DOI URL PMID |
| [42] |
Turkington R, Maze J (1982). Patterns of dry-matter distribution in transplanted populations of Trifolium repens and its bearing on ecological interpretations. Canadian Journal of Botany, 60, 2014-2018.
DOI URL |
| [43] |
Vojtísková L, Munzarová E, Votrubová O, Řihová A, Juřicová B (2004). Growth and biomass allocation of sweet flag (Acorus calamus L.) under different nutrient conditions. Hydrobiologia, 518, 9-22.
DOI URL |
| [44] | Wang QR (王庆仁), Li JY (李继云), Li ZS (李振声) (1999). Studies on plant nutrition of efficient utility for soil phosphorus. Acta Ecologica Sinica (生态学报), 19, 417-421. (in Chinese with English abstract) |
| [45] |
Wetzel PR, van der Valk AG (1998). Effects of nutrient and soil moisture on competition between Carex stricta, Phalaris arundinacea and Typha latifolia. Plant Ecology, 138, 179-190.
DOI URL |
| [46] |
Xie YH, An SQ, Wu BF (2005). Resource allocation in the submerged plant Vallisneria natans related to sediment type, rather than water-column nutrients. Freshwater Biology, 50, 391-402.
DOI URL |
| [47] | Xu KY (许凯扬), Ye WH (叶万辉), Li J (李静), Huang ZL (黄忠良), Li GM (李国民) (2005). Phenotypic plasticity in response to soil water content in the invasive species Alternanth-era philoxeroides. Journal of Central China Normal University (Naturalt Science Edition)(华中师范大学学报(自然科学版)), 39, 100-103. (in Chinese with English abstract) |
| [48] | Zeng XF (曾宪锋), Zhou Y (周仪)(1996). The debate about adventitive root on the submerged stem of Trapa sp. Bulletin of Biology (生物学通报), 31(9), 28. (in Chinese) |
| [1] | 马常钦, 黄海龙, 彭政淋, 吴纯泽, 韦庆钰, 贾红涛, 卫星. 水曲柳雌雄株复叶类型及光合功能对不同生境的响应[J]. 植物生态学报, 2023, 47(9): 1287-1297. |
| [2] | 冯珊珊, 黄春晖, 唐梦云, 蒋维昕, 白天道. 细叶云南松针叶形态和显微性状地理变异及其环境解释[J]. 植物生态学报, 2023, 47(8): 1116-1130. |
| [3] | 杨丽婷, 谢燕燕, 左珂怡, 徐森, 谷瑞, 陈双林, 郭子武. 分株比例对异质光环境下美丽箬竹克隆系统光合生理的影响[J]. 植物生态学报, 2022, 46(1): 88-101. |
| [4] | 杜军, 王文, 何志斌, 陈龙飞, 蔺鹏飞, 朱喜, 田全彦. 祁连山青海云杉物候表型的空间分异及其内在机制[J]. 植物生态学报, 2021, 45(8): 834-843. |
| [5] | 叶学华, 薛建国, 谢秀芳, 黄振英. 外部干扰对根茎型克隆植物甘草自然种群植株生长及主要药用成分含量的影响[J]. 植物生态学报, 2020, 44(9): 951-961. |
| [6] | 白天道, 余春兰, 甘泽朝, 赖海荣, 杨隐超, 黄厚宸, 蒋维昕. 细叶云南松种实性状变异与地理气象因子的关联[J]. 植物生态学报, 2020, 44(12): 1224-1235. |
| [7] | 杨雪, 申俊芳, 赵念席, 高玉葆. 不同基因型羊草数量性状的可塑性及遗传分化[J]. 植物生态学报, 2017, 41(3): 359-368. |
| [8] | 周晓旋, 蔡玲玲, 傅梅萍, 洪礼伟, 沈英嘉, 李庆顺. 红树植物胎生现象研究进展[J]. 植物生态学报, 2016, 40(12): 1328-1343. |
| [9] | 李西良,侯向阳,吴新宏,萨茹拉,纪磊,陈海军,刘志英,丁勇. 草甸草原羊草茎叶功能性状对长期过度放牧的可塑性响应[J]. 植物生态学报, 2014, 38(5): 440-451. |
| [10] | 王意锟, 金爱武, 朱强根, 邱永华, 季新良, 张四海. 施肥对毛竹种群不同年龄分株间胸径大小关系的影响[J]. 植物生态学报, 2014, 38(3): 289-297. |
| [11] | 彭一可, 罗芳丽, 李红丽, 于飞海. 根状茎型植物扁秆荆三棱对土壤养分异质性尺度和对比度的生长响应[J]. 植物生态学报, 2013, 37(4): 335-343. |
| [12] | 罗维成, 曾凡江, 刘波, 宋聪, 彭守兰, Stefan K. ARNDT. 疏叶骆驼刺母株与子株间的水分整合[J]. 植物生态学报, 2013, 37(2): 164-172. |
| [13] | 葛俊, 邢福. 克隆植物对种间竞争的适应策略[J]. 植物生态学报, 2012, 36(6): 587-596. |
| [14] | 覃凤飞,李强,崔棹茗,李洪萍,杨智然. 越冬期遮阴条件下3个不同秋眠型紫花苜蓿品种叶片解剖结构与其光生态适应性[J]. 植物生态学报, 2012, 36(4): 333-345. |
| [15] | 朱志红, 席博, 李英年, 臧岳铭, 王文娟, 刘建秀, 郭华. 高寒草甸不同生境粗喙薹草补偿生长研究[J]. 植物生态学报, 2010, 34(3): 348-358. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2026 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
![]()