

植物生态学报 ›› 2019, Vol. 43 ›› Issue (4): 374-382.DOI: 10.17521/cjpe.2019.0048
所属专题: 菌根真菌
• 研究论文 • 上一篇
收稿日期:2019-09-05
修回日期:2019-04-16
出版日期:2019-04-20
发布日期:2019-05-30
通讯作者:
高江云
基金资助:
FAN Zi-Teng,WU Yu-Ling,WANG Xin-Ju,LI Tai-Qiang,GAO Jiang-Yun( )
)
Received:2019-09-05
Revised:2019-04-16
Online:2019-04-20
Published:2019-05-30
Contact:
GAO Jiang-Yun
Supported by:摘要:
石斛属(Dendrobium)植物在种子共生萌发过程中与真菌有着较为专一的共生关系, 为探讨这种共生关系在种间杂交后代上的进化和适应, 深入理解兰科植物和真菌共生关系的形成机制, 该研究利用能有效促进铁皮石斛(Dendrobium officinale)和齿瓣石斛(D. devonianum)种子萌发形成幼苗, 并具有较强专一性的胶膜菌属(Tulasnella)真菌SSCDO-5和瘤菌根菌属(Epulorhiza)真菌FDd1, 开展真菌对铁皮石斛和D. tortile种间杂交种子萌发效应的研究。结果表明, 在真菌与种子共生培养68天时, SSCDO-5菌株和FDd1菌株都能有效地促进杂交种子形成原球茎和幼苗, 两个接菌处理之间无显著差异, 来源于铁皮石斛的SSCDO-5菌株不但没有表现出优势, 反而在杂交石斛幼苗形成率上低于来源于齿瓣石斛的FDd1菌株(SSCDO-5: (22.13 ± 6.62)%; FDd1: (29.53 ± 5.51)%); SSCDO-5菌株和铁皮石斛在幼苗形成和发育阶段的共生专一性并没有在杂交后代上得到遗传或表现, 或者说是杂交打破了这种专一性的共生关系, 使得杂交后代能够和不同的真菌建立新的共生关系。该结果不支持关于共生真菌专一性是石斛属植物杂交后代形成的重要限制因素的假设, 推测石斛属植物在幼苗分化和发育阶段与真菌这种专一性的共生关系是在适应特定生态环境的过程中形成和建立的。
范紫腾, 毋钰灵, 王新菊, 李太强, 高江云. 共生真菌对兰科植物种间杂交后代种子萌发的效应. 植物生态学报, 2019, 43(4): 374-382. DOI: 10.17521/cjpe.2019.0048
FAN Zi-Teng, WU Yu-Ling, WANG Xin-Ju, LI Tai-Qiang, GAO Jiang-Yun. Effects of symbiotic fungi on seed germination of interspecific hybrid progenies in Orchidaceae. Chinese Journal of Plant Ecology, 2019, 43(4): 374-382. DOI: 10.17521/cjpe.2019.0048

图1 三种石斛在不同处理、不同时期种子萌发及原球茎和幼苗形成情况。A, 铁皮石斛种子和SSCDO-5菌株共生培养30天时萌发的种子和形成的原球茎。B, 齿瓣石斛种子和FDd1菌株共生培养58天时形成的幼苗。C, 杂交石斛种子和FDd1菌株共生培养68天时的幼苗情况。D, 杂交石斛种子和SSCDO-5菌株共生培养68天时的幼苗情况。
Fig. 1 Seed germination, protocorm and seedling formation of three Dendrobium species under different treatments at different stages. A, Germinating seed and formed protocorm of D. officinale after 30 days incubation with SSCDO-5 strain. B, A seeding of D. devonianum after 58 days incubation with FDd1 strain. C, Seedings of hybrid after 68 days incubation with FDd1 strain. D, Seedings of hybrid after 68 days incubation with SSCDO-5 strain.
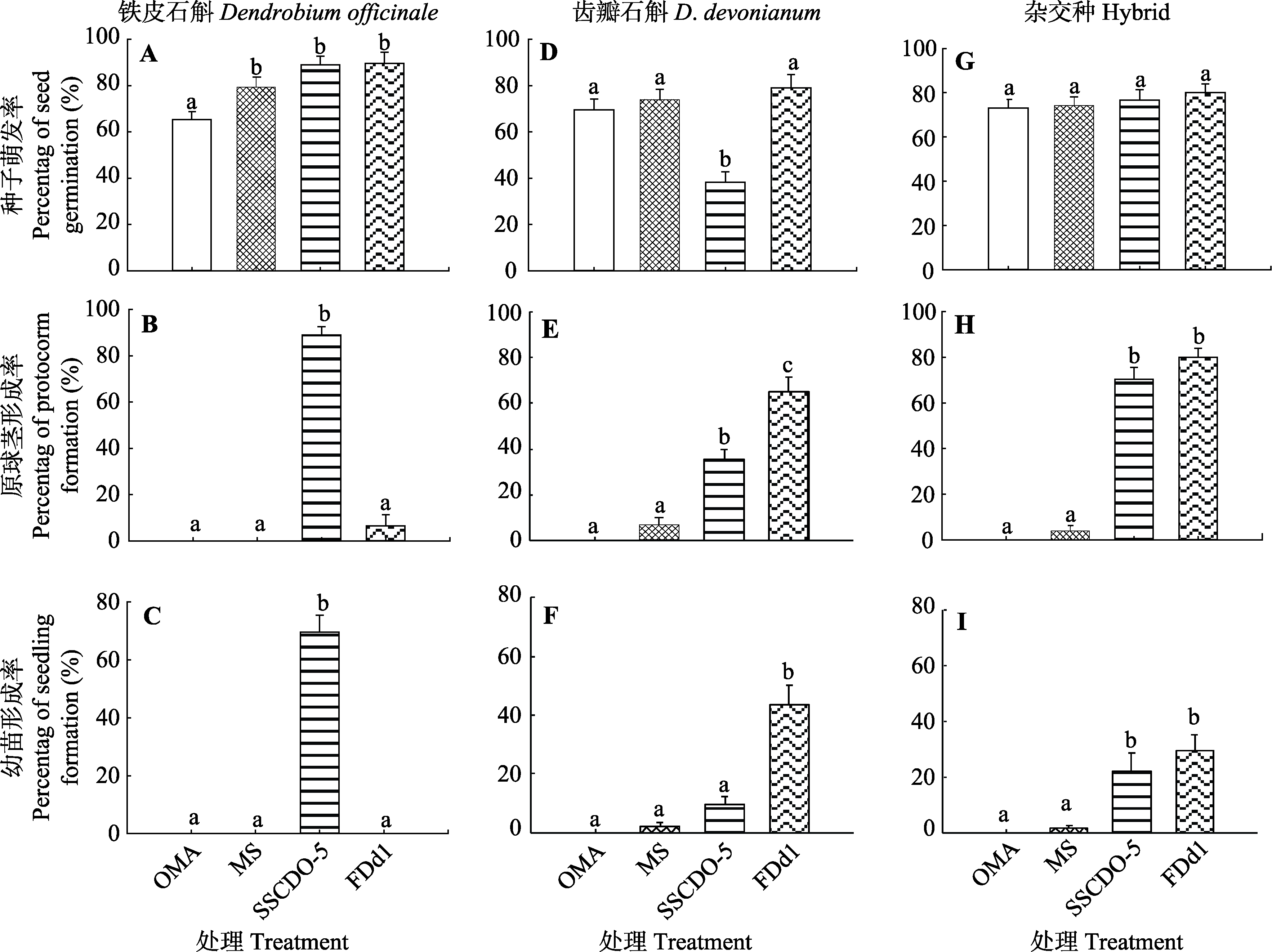
图2 三种石斛不同处理下种子萌发率(A、D、G)、原球茎形成率(B、E、H)和幼苗形成率(C、F、I)(平均值±标准误差)。柱子上方不同小写字母表示同一阶段不同处理之间差异显著(α = 0.05)。OMA, 不接菌的燕麦琼脂培养基作为营养缺乏对照处理; MS, 不接菌的MS培养基作为营养丰富对照处理; SSCDO-5, 燕麦培养基上接种SSCDO-5菌株处理; FDd1, 燕麦培养基上接种FDd1菌株处理菌的处理。
Fig. 2 Percentage of seed germination (A, D, G), protocorm (B, E, H) and seedling (C, F, I) formation of different treatments in three Dendrobium species (mean ± SE). Different lowercase letters above the bars represent significant differences between different treatments at the same stage (α = 0.05). OMA, oatmeal agar medium without fungal inoculation was used as nutrient-poor control treatment; MS, MS medium without fungal inoculation was used as nutrient-rich control treatment; SSCDO-5, treatment of fungal inoculation with SSCDO-5 strain on OMA medium; FDd1, treatment of fungal inoculation with FDd1 strain on OMA medium.
| 多重比较 Multiple comparisons | 铁皮石斛 D. officinale | 齿瓣石斛 D. devonianum | 杂交种 Hybrid | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 种子萌发率 Seed germination rate | 原球茎形成率 Protocorms formation rate | 幼苗率 Seedlings formation rate | 种子萌发率 Seed germination rate | 原球茎形成率 Protocorms formation rate | 幼苗率 Seedlings formation rate | 种子萌发率 Seed germination rate | 原球茎形成率 Protocorms formation rate | 幼苗率 Seedlings formation rate | |
| OMA-MS | -0.139 3 ± 0.058 2* | 0.000 0 ± 0.042 7 | 0.000 0 ± 0.053 4 | -0.044 4 ± 0.071 1 | -0.065 5 ± 0.063 9 | -0.021 4 ± 0.051 6 | -0.012 0 ± 0.058 1 | -0.039 3 ± 0.053 0 | -0.017 2 ± 0.064 4 |
| OMA-SSCDO-5 | -0.235 3 ± 0.057 7*** | -0.888 8 ± 0.042 3*** | -0.696 6 ± 0.052 9*** | 0.312 1 ± 0.069 5*** | -0.354 8 ± 0.062 4*** | -0.096 8 ± 0.050 4 | -0.036 9 ± 0.058 1 | -0.704 0 ± 0.053 0*** | -0.221 3 ± 0.064 4** |
| OMA-FDd1 | -0.242 3 ± 0.058 2*** | -0.065 5 ± 0.042 7 | 0.065 5 ± 0.053 4 | -0.094 8 ± 0.070 5 | -0.651 7 ± 0.063 3*** | -0.436 8 ± 0.051 2*** | -0.070 1 ± 0.059 1 | -0.800 4 ± 0.053 9*** | -0.295 3 ± 0.065 5*** |
| MS-SSCDO-5 | -0.096 0 ± 0.058 2 | -0.888 8 ± 0.042 7*** | -0.696 6 ± 0.053 4*** | 0.356 5 ± 0.068 1*** | -0.289 4 ± 0.061 1*** | -0.075 3 ± 0.049 4 | -0.024 9 ± 0.055 3 | -0.664 8 ± 0.050 4*** | -0.204 0 ± 0.061 3** |
| MS- FDd1 | -0.102 9 ± 0.058 7 | -0.006 6 ± 0.043 1 | 0.065 5 ± 0.053 8 | -0.050 4 ± 0.069 2 | -0.586 2 ± 0.062 1*** | -0.415 4 ± 0.050 2*** | -0.058 1 ± 0.056 3 | -0.761 1 ± 0.0513 4*** | -0.278 0 ± 0.062 4*** |
| SSCDO-5- FDd1 | -0.007 0 ± 0.058 2 | 0.823 3 ± 0.042 7*** | 0.631 0 ± 0.053 4*** | -0.406 9 ± 0.067 5*** | -0.296 9 ± 0.060 6*** | -0.340 0 ± 0.049 0*** | -0.033 2 ± 0.056 3 | -0.096 4 ± 0.051 3 | -0.074 0 ± 0.062 4 |
表1 不同处理三种石斛种子萌发率、原球茎形成率和幼苗形成率的多重比较(均值差±标准误差)
Table 1 Multiple comparisons on seed germination rate, protocorms formation rate and seedlings formation rate of different treatments for three Dendrobium species (mean ± SE)
| 多重比较 Multiple comparisons | 铁皮石斛 D. officinale | 齿瓣石斛 D. devonianum | 杂交种 Hybrid | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 种子萌发率 Seed germination rate | 原球茎形成率 Protocorms formation rate | 幼苗率 Seedlings formation rate | 种子萌发率 Seed germination rate | 原球茎形成率 Protocorms formation rate | 幼苗率 Seedlings formation rate | 种子萌发率 Seed germination rate | 原球茎形成率 Protocorms formation rate | 幼苗率 Seedlings formation rate | |
| OMA-MS | -0.139 3 ± 0.058 2* | 0.000 0 ± 0.042 7 | 0.000 0 ± 0.053 4 | -0.044 4 ± 0.071 1 | -0.065 5 ± 0.063 9 | -0.021 4 ± 0.051 6 | -0.012 0 ± 0.058 1 | -0.039 3 ± 0.053 0 | -0.017 2 ± 0.064 4 |
| OMA-SSCDO-5 | -0.235 3 ± 0.057 7*** | -0.888 8 ± 0.042 3*** | -0.696 6 ± 0.052 9*** | 0.312 1 ± 0.069 5*** | -0.354 8 ± 0.062 4*** | -0.096 8 ± 0.050 4 | -0.036 9 ± 0.058 1 | -0.704 0 ± 0.053 0*** | -0.221 3 ± 0.064 4** |
| OMA-FDd1 | -0.242 3 ± 0.058 2*** | -0.065 5 ± 0.042 7 | 0.065 5 ± 0.053 4 | -0.094 8 ± 0.070 5 | -0.651 7 ± 0.063 3*** | -0.436 8 ± 0.051 2*** | -0.070 1 ± 0.059 1 | -0.800 4 ± 0.053 9*** | -0.295 3 ± 0.065 5*** |
| MS-SSCDO-5 | -0.096 0 ± 0.058 2 | -0.888 8 ± 0.042 7*** | -0.696 6 ± 0.053 4*** | 0.356 5 ± 0.068 1*** | -0.289 4 ± 0.061 1*** | -0.075 3 ± 0.049 4 | -0.024 9 ± 0.055 3 | -0.664 8 ± 0.050 4*** | -0.204 0 ± 0.061 3** |
| MS- FDd1 | -0.102 9 ± 0.058 7 | -0.006 6 ± 0.043 1 | 0.065 5 ± 0.053 8 | -0.050 4 ± 0.069 2 | -0.586 2 ± 0.062 1*** | -0.415 4 ± 0.050 2*** | -0.058 1 ± 0.056 3 | -0.761 1 ± 0.0513 4*** | -0.278 0 ± 0.062 4*** |
| SSCDO-5- FDd1 | -0.007 0 ± 0.058 2 | 0.823 3 ± 0.042 7*** | 0.631 0 ± 0.053 4*** | -0.406 9 ± 0.067 5*** | -0.296 9 ± 0.060 6*** | -0.340 0 ± 0.049 0*** | -0.033 2 ± 0.056 3 | -0.096 4 ± 0.051 3 | -0.074 0 ± 0.062 4 |
| [1] | Aceto S, Caputo P, Cozzolino S, Gaudio L, Moretti A ( 1999). Phylogeny and evolution of Orchis and allied genera based on ITS DNA variation: Morphological gaps and molecular continuity. Molecular Phylogenetics & Evolution, 13, 67-76. |
| [2] | Arditti J ( 1967). Factors affecting the germination of orchid seeds. The Botanical Review, 33, 1-97. |
| [3] | Arditti J, Ghani AKA ( 2000). Numerical and physical properties of orchid seeds and their biological implications. New Phytologist, 145, 367-421. |
| [4] | Bidartondo MI, Read DJ (2008). Fungal specificity bottlenecks during orchid germination and development. Molecular Ecology, 17, 3707-3716. |
| [5] | Brundrett MC, Scade A, Batty AL, Dixon KW, Sivasithamparam K ( 2003). Development of in situ and ex situ seed baiting techniques to detect mycorrhizal fungi from terrestrial orchid habitats. Mycological Research, 107, 1210-1220. |
| [6] | Chase MW, Cameron KM, Freudenstein JV, Pridgeon AM, Salazar G, Berg C, Schuiteman A ( 2015). An updated classification of Orchidaceae. Botanical Journal of the Linnean Society, 177, 151-174. |
| [7] | Coyne JA, Orr HA ( 2004). Speciation. Sinauer Associates, Sunderland, USA. |
| [8] | Cozzolino S, Widmer A ( 2005). Orchid diversity: An evolutionary consequence of deception? Trends in Ecology & Evolution, 20, 487-494. |
| [9] | Dafni A, Ivri Y (1979). Pollination ecology of, and hybridization between,Orchis coriophora L. and O. collina Sol. ex Russ.(Orchidaceae) in Israel. New Phytologist, 83, 181-187. |
| [10] | Dearnaley JDW, Martos F, Selosse MA (2012). Orchid mycorrhizas: Molecular ecology, physiology, evolution and conservation aspects. In: Hock B ed. Fungal Associations. 2nd edn. Springer, Berlin,Germany. 207-230. |
| [11] | Dearnaley JWD, Perotto S, Selosse MA (2016). Structure and development of orchid mycorrhizas. In: Martin F ed. Molecular Mycorrhizal Symbiosis. Springer, Berlin. 63-86. |
| [12] | Gao JY, Liu Q, Yu DL (2014). Orchids of Xishuangbanna: Diversity and Conservation. China Forestry Publishing House, Beijing. 15-31. |
| [ 高江云, 刘强, 余东莉 (2014). 西双版纳的兰科植物: 多样性和保护. 中国林业出版社, 北京. 15-31.] | |
| [13] | Hegarty MJ, Hiscock SJ ( 2005). Hybrid speciation in plants: New insights from molecular studies. New Phytologist, 165, 411-423. |
| [14] | Hollick PS, Taylor RJ, McComb JA, Dixon KW ( 2005). If orchid mycorrhizal fungi are so specific, how do natural hybrids cope? Selbyana, 26, 159-170. |
| [15] | Huang H, Zi XM, Lin H, Gao JY ( 2018). Host-specificity of symbiotic mycorrhizal fungi for enhancing seed germination, protocorm formation and seedling development of over-collected medicinal orchid,Dendrobium devonianum. Journal of Microbiology, 56, 42-48. |
| [16] | Kauth P, Dutra D, Johnson T (2008). Techniques and applications of in vitro orchid seed germination. In: Teixeira da Silva JA ed. Floriculture, Ornamental and Plant Biotechnology: Advances and Topical Issues. Global Science Books, Iselworth,UK. 375-391. |
| [17] | Lee YI ( 2007). The asymbiotic seed germination of six Paphiopedilum species in relation to the time of seed collection and seed pretreatment. Acta Horticulturae, 755, 381-386. |
| [18] | Liu HX, Luo YB, Liu H ( 2010). Studies of mycorrhizal fungi of Chinese orchids and their role in orchid conservation in China―A review. The Botanic Review, 76, 241-262. |
| [19] | Long B, Niemiera AX, Cheng ZY, Long CL ( 2010). In vitro propagation of four threatened Paphiopedilum species (Orchidaceae). Plant Cell Tissue and Organ Culture, 101, 151-162. |
| [20] | Mallet J ( 2005). Hybridization as an invasion of the genome. Trends in Ecology & Evolution, 20, 229-237. |
| [21] | Martos F, Francios M, Pailler T, Kottke I, Cédric G, Marc- André Selosse ( 2012). The role of epiphytism in architecture and evolutionary constraint within mycorrhizal networks of tropical orchids. Molecular Ecology, 21, 5098-5109. |
| [22] | Masuhara G, Katsuya K (1994). In situ and in vitro specificity between Rhizoctonia spp. and Spiranthes sinensis(Persoon) Ames. var. amoena 127, 711-718. |
| [23] | McCormick MK, Jacquemyn H ( 2014). What constrains the distribution of orchid populations? New Phytologist, 202, 392-400. |
| [24] | McKendrick SL, Leake JR, Read DJ ( 2000). Symbiotic germination and development of myco-heterotrophic plants in nature: Transfer of carbon from ectomycorrhizal Salix repens and Betula pendula to the orchid Corallorhiza trifida through shared hyphal connections. New Phytologist, 145, 539-548. |
| [25] | Meng YY, Shao SC, Liu SJ, Gao JY ( 2019). Do the fungi associated with roots of adult plants support seed germination? A case study on Dendrobium exile(Orchidaceae). Global Ecology and Conservation, 17, e00582. DOI: 10.1016/ j.gecco.2019.e00582. |
| [26] | Nontachaiyapoom S, Sasirat S, Manoch L ( 2010). Isolation and identification of Rhizoctonia-like fungi from roots of three orchid genera, Paphiopedilum, Dendrobium, and Cymbidium, collected in Chiang Rai and Chiang Mai provinces of Thailand. Mycorrhiza, 20, 459-471. |
| [27] | Ramsay MM, Stewart J ( 1998). Re-establishment of the lady’s slipper orchid (Cypripedium Calceolus L.) in Britain. Botanical Journal of the Linnean Society, 126, 173-181. |
| [28] | Rasmussen HN, Dixon KW, Jersakova J, Tesitelova T ( 2015). Germination and seedling establishment in orchids: A complex of requirements. Annals of Botany, 116, 391-402. |
| [29] | Roberts DL (2003). Pollination biology: The role of sexual reproduction in orchid conservation. In: Dixon KW, Kell SP, Barrett RL, Cribb PJ eds. Orchid Conservation. Natural History Publications, Kota Kinabalu, Sabah. 113-136. |
| [30] | Schatz B ( 2006). Fine scale distribution of pollinator explains the occurrence of the natural orchid hybrid × Orchis bergonii. Ecoscience, 13, 111-118. |
| [31] | Schatz B, Geoffroy A, Dainat B, Bessiere JM, Buatois B, Hossaert-Mckey M, Selosse MA ( 2010). A case study of modified interactions with symbionts in a hybrid mediterranean orchid. American Journal of Botany, 97, 1278-1288. |
| [32] | Sheng CL, Li YY, Gao JY ( 2012). Ex situ symbiotic seed germination, isolation and identification of effective symbiotic fungus in Cymbidium mannii(Orchidaceae). Chinese Journal of Plant Ecology, 36, 859-869. |
| [ 盛春玲, 李勇毅, 高江云 ( 2012). 硬叶兰种子的迁地共生萌发及有效共生真菌的分离和鉴定. 植物生态学报, 36, 859-869.] | |
| [33] | Stewart SL, Kane ME ( 2007). Symbiotic seed germination and evidence for in vitro mycobiont specificity in Spiranthes brevilabris(Orchidaceae) and its implications for species-level conservation. In Vitro Cellular & Developmental Biology-Plant, 43, 178-186. |
| [34] | Stimart DP, Ascher PD ( 1981). In vitro germination of Paphiopedilum seed on a completely defined medium. Scientia Horticulturae, 14, 165-170. |
| [35] | Swarts ND, Dixon KW ( 2009). Terrestrial orchid conservation in the age of extinction. Annals of Botany, 104, 543-556. |
| [36] | Vujanovic V, St-Arnaud M, Barabe D, Thibeault G ( 2000). Viability testing of orchid seed and the promotion of colouration and germination. Annals of Botany, 86, 79-86. |
| [37] | Whitehead MR, Peakall R ( 2014). Pollinator specificity drives strong pre-pollination reproductive isolation in sympatric sexually deceptive orchids. Evolution, 68, 1561-1575. |
| [38] | Yamazaki J, Miyoshi K ( 2006). In vitro asymbiotic germination of immature seed and formation of protocorm by Cephalanthera falcata(Orchidaceae). Annals of Botany, 98, 1197-1206. |
| [39] | Zelmer CD, Cuthbertson L, Currah RS ( 1996). Fungi associated with terrestrial orchid mycorrhizas, seeds and protocorms. Mycoscience, 37, 439-448. |
| [40] | Zettler LW, Piskin KA, Stewart SL, Hartsock JJ, Bowles ML, Bell TJ ( 2005). Protocorm mycobionts of the federally threatened eastern prairie fringed orchid,Platanthera leucophaea(Nutt.) Lindley, and a technique to prompt leaf elongation in seedlings. Studies in Mycology, 53, 163-171. |
| [41] | Zhou X, Gao JY ( 2016). Highly compatible Epa-01 strain promotes seed germination and protocorm development of Papilionanthe teres(Orchidaceae). Plant Cell Tissue and Organ Culture, 125, 479-493. |
| [42] | Zi XM, Sheng CL, Goodale UM, Shao SC, Gao JY ( 2014). In situ seed baiting to isolate germination-enhancing fungi for an epiphytic orchid, Dendrobium aphyllum(Orchidaceae). Mycorrhiza, 24, 487-499. |
| [43] | Zotz G ( 2013). The systematic distribution of vascular epiphytes—A critical update. Botanical Journal of the Linnean Society, 171, 453-481. |
| [1] | 徐子怡 金光泽. 阔叶红松林不同菌根类型幼苗细根功能性状的变异与权衡[J]. 植物生态学报, 2024, 48(5): 612-622. |
| [2] | 陈科宇 邢森 唐玉 孙佳慧 任世杰 张静 纪宝明. 不同草地型土壤丛枝菌根真菌群落特征及其驱动因素[J]. 植物生态学报, 2024, 48(5): 660-674. |
| [3] | 胡蝶 蒋欣琪 戴志聪 陈戴一 张雨 祁珊珊 杜道林. 丛枝菌根真菌提高入侵杂草南美蟛蜞菊对除草剂的耐受性[J]. 植物生态学报, 2024, 48(5): 651-659. |
| [4] | 陈保冬, 付伟, 伍松林, 朱永官. 菌根真菌在陆地生态系统碳循环中的作用[J]. 植物生态学报, 2024, 48(1): 1-20. |
| [5] | 任悦, 高广磊, 丁国栋, 张英, 赵珮杉, 柳叶. 不同生长期樟子松外生菌根真菌群落物种组成及其驱动因素[J]. 植物生态学报, 2023, 47(9): 1298-1309. |
| [6] | 胡同欣, 李蓓, 李光新, 任玥霄, 丁海磊, 孙龙. 火烧黑碳对生长季兴安落叶松林外生菌根真菌群落物种组成的影响[J]. 植物生态学报, 2023, 47(6): 792-803. |
| [7] | 杨佳绒, 戴冬, 陈俊芳, 吴宪, 刘啸林, 刘宇. 丛枝菌根真菌多样性对植物群落构建和稀有种维持的研究进展[J]. 植物生态学报, 2023, 47(6): 745-755. |
| [8] | 何斐, 李川, Faisal SHAH, 卢谢敏, 王莹, 王梦, 阮佳, 魏梦琳, 马星光, 王卓, 姜浩. 丛枝菌根菌丝桥介导刺槐-魔芋间碳转运和磷吸收[J]. 植物生态学报, 2023, 47(6): 782-791. |
| [9] | 赵榕江, 陈焘, 董丽佳, 郭辉, 马海鲲, 宋旭, 王明刚, 薛伟, 杨强. 植物-土壤反馈及其在生态学中的研究进展[J]. 植物生态学报, 2023, 47(10): 1333-1355. |
| [10] | 张慧, 曾文静, 龚新桃, 马泽清. 亚热带典型树种根毛特征及其与共生真菌的关系[J]. 植物生态学报, 2023, 47(1): 88-100. |
| [11] | 谢伟, 郝志鹏, 张莘, 陈保冬. 丛枝菌根网络介导的植物间信号交流研究进展及展望[J]. 植物生态学报, 2022, 46(5): 493-515. |
| [12] | 单婷婷, 陈彤垚, 陈晓梅, 郭顺星, 王爱荣. 菌根真菌与兰科植物氮营养关系的研究进展[J]. 植物生态学报, 2022, 46(5): 516-528. |
| [13] | 马炬峰, 辛敏, 徐陈超, 祝琬莹, 毛传澡, 陈欣, 程磊. 丛枝菌根真菌与氮添加对不同根形态基因型水稻氮吸收的影响[J]. 植物生态学报, 2021, 45(7): 728-737. |
| [14] | 左永令, 杨小波, 李东海, 吴二焕, 杨宁, 李龙, 张培春, 陈琳, 李晨笛. 环境因子对海南岛野生兰科植物物种组成与分布格局的影响[J]. 植物生态学报, 2021, 45(12): 1341-1349. |
| [15] | 庞芳, 夏维康, 何敏, 祁珊珊, 戴志聪, 杜道林. 固氮菌缓解氮限制环境中丛枝菌根真菌对加拿大一枝黄花的营养竞争[J]. 植物生态学报, 2020, 44(7): 782-790. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2022 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19