

植物生态学报 ›› 2020, Vol. 44 ›› Issue (3): 192-204.DOI: 10.17521/cjpe.2019.0304
收稿日期:2019-11-07
接受日期:2020-02-01
出版日期:2020-03-20
发布日期:2020-03-26
通讯作者:
朱师丹 ORCID:0000-0002-9228-368X
基金资助:
SONG Hui-Qing,NI Ming-Yuan,ZHU Shi-Dan( )
)
Received:2019-11-07
Accepted:2020-02-01
Online:2020-03-20
Published:2020-03-26
Contact:
Shi-Dan ZHU ORCID:0000-0002-9228-368X
Supported by:摘要:
木质藤本是热带森林的重要组成部分, 显著影响森林的结构和功能。已有研究发现木质藤本与乔木的水力结构存在显著差异: 木质藤本的缠绕或攀缘茎细小, 但其木质部具有粗大的长导管, 输水效率高, 抗栓塞能力低。为降低基因型差异对比较结果的影响, 该研究选取热带崖豆藤属(Millettia)和买麻藤属(Gnetum)的乔木和木质藤本, 比较同属内不同生长型植物的水力和光合性状的差异, 分析水分传导效率与抗栓塞能力之间以及水力与光合性状之间的相关关系。结果发现: (1)崖豆藤属植物水力性状的种间差异大, 与生活型和需光性有关。耐阴的木质藤本反而具有较低的水分传导效率和较高的抗栓塞能力。(2)买麻藤属植物是裸子植物较为进化的类群(具有导管和阔叶), 其乔木的水分传导效率很低, 但是其木质藤本的水分传导效率高于其他阳生性的被子植物。(3)不论乔木还是木质藤本, 水分传导的有效性与安全性在枝条和叶片水平上均没有显著的权衡关系。(4)与同属乔木相比, 木质藤本的叶片较枝条的抗栓塞能力更强, 在旱季具有更高的最大净光合速率和气孔导度, 支持了木质藤本的“旱季生长优势假说”。该研究揭示了热带木质藤本水力性状的多样性和重要性, 为阐明环境变化对这一重要植物类群的影响, 需要对它们的水力特征进行更广泛的研究。
宋慧清, 倪鸣源, 朱师丹. 乔木与木质藤本的水力与光合性状的差异: 以热带森林崖豆藤属和买麻藤属为例. 植物生态学报, 2020, 44(3): 192-204. DOI: 10.17521/cjpe.2019.0304
SONG Hui-Qing, NI Ming-Yuan, ZHU Shi-Dan. Hydraulic and photosynthetic characteristics differ between co-generic tree and liana species: a case study of Millettia and Gnetum in tropical forest. Chinese Journal of Plant Ecology, 2020, 44(3): 192-204. DOI: 10.17521/cjpe.2019.0304
| 物种 Species | 生活型 Life form | 缩写 Abbreviation | 最大导管长度 Maximum vessel length (m) | 原生生境分布 Native habitat of adult in the forest |
|---|---|---|---|---|
| 崖豆藤属(豆科) Millettia (Fabaceae) | ||||
| 思茅崖豆 Millettia leptobotrya | 乔木 Tree | M. lep | 0.69 | 山坡疏林或常绿阔叶林中, 海拔300-1 000 m Open forest on slopes or evergreen broad-leaved forest; 300-1 000 m a.s.l. |
| 红河崖豆 Millettia cubittii | 乔木 Tree | M. cub | 0.49 | 河边的林地或路边, 海拔300-1 000 m Riparian forest or roadside; 300-1 000 m a.s.l. |
| 变色鸡血藤 Millettia versicolor | 乔木 Tree | M. ver | 引种于非洲南部热带次生林、稀树草原 Tropical secondary forests and savannas of southern Africa | |
| 厚果崖豆藤 Millettia pachycarpa | 藤本 Liana | M. pac | 1.50 | 山坡疏林、阔叶林内或路边, 海拔100-2 000 m Open forest on slopes, broad-leaved forest or roadside; 100-2 000 m a.s.l. |
| 香花崖豆藤 Millettia dielsiana | 藤本 Liana | M. die | 0.82 | 山坡杂木林与灌丛中, 海拔300-2 500 m Tree-shrub mixed forest on slopes; 300-2 500 m a.s.l. |
| 海南崖豆藤 Millettia pachyloba | 藤本 Liana | M. pab | 沟谷常绿阔叶林中, 海拔1 500 m以下 Evergreen broad-leaved forests in valleys; below 1 500 m a.s.l. | |
| 买麻藤属(买麻藤科) Gnetum (Gnetaceae) | ||||
| 灌状买麻藤 Gnetum gnemon | 乔木 Tree | G. gne | 湿润的常绿次生森林下, 海拔1 600-2 000 m Moist evergreen secondary forests; 1 600-2 000 m a.s.l. | |
| 少苞买麻藤 Gnetum brunonianum | 乔木 Tree | G. bru | 1.14 | 海拔350 m的阔叶林下 Broad-leaved forest; below 350 m a.s.l. |
| 小叶买麻藤 Gnetum parvifolium | 藤本 Liana | G. par | 2.00 | 海拔较低的干燥平地或湿润谷地的森林,海拔100-1 000m Dry flat or moist valleys forests at lower altitude; 100-1 000 m a.s.l. |
表1 9种植物的生活型、名称缩写、最大导管长度以及生态学特性描述
Table 1 Life form, species abbreviations, maximum vessel length and ecological descriptions for the nine woody species
| 物种 Species | 生活型 Life form | 缩写 Abbreviation | 最大导管长度 Maximum vessel length (m) | 原生生境分布 Native habitat of adult in the forest |
|---|---|---|---|---|
| 崖豆藤属(豆科) Millettia (Fabaceae) | ||||
| 思茅崖豆 Millettia leptobotrya | 乔木 Tree | M. lep | 0.69 | 山坡疏林或常绿阔叶林中, 海拔300-1 000 m Open forest on slopes or evergreen broad-leaved forest; 300-1 000 m a.s.l. |
| 红河崖豆 Millettia cubittii | 乔木 Tree | M. cub | 0.49 | 河边的林地或路边, 海拔300-1 000 m Riparian forest or roadside; 300-1 000 m a.s.l. |
| 变色鸡血藤 Millettia versicolor | 乔木 Tree | M. ver | 引种于非洲南部热带次生林、稀树草原 Tropical secondary forests and savannas of southern Africa | |
| 厚果崖豆藤 Millettia pachycarpa | 藤本 Liana | M. pac | 1.50 | 山坡疏林、阔叶林内或路边, 海拔100-2 000 m Open forest on slopes, broad-leaved forest or roadside; 100-2 000 m a.s.l. |
| 香花崖豆藤 Millettia dielsiana | 藤本 Liana | M. die | 0.82 | 山坡杂木林与灌丛中, 海拔300-2 500 m Tree-shrub mixed forest on slopes; 300-2 500 m a.s.l. |
| 海南崖豆藤 Millettia pachyloba | 藤本 Liana | M. pab | 沟谷常绿阔叶林中, 海拔1 500 m以下 Evergreen broad-leaved forests in valleys; below 1 500 m a.s.l. | |
| 买麻藤属(买麻藤科) Gnetum (Gnetaceae) | ||||
| 灌状买麻藤 Gnetum gnemon | 乔木 Tree | G. gne | 湿润的常绿次生森林下, 海拔1 600-2 000 m Moist evergreen secondary forests; 1 600-2 000 m a.s.l. | |
| 少苞买麻藤 Gnetum brunonianum | 乔木 Tree | G. bru | 1.14 | 海拔350 m的阔叶林下 Broad-leaved forest; below 350 m a.s.l. |
| 小叶买麻藤 Gnetum parvifolium | 藤本 Liana | G. par | 2.00 | 海拔较低的干燥平地或湿润谷地的森林,海拔100-1 000m Dry flat or moist valleys forests at lower altitude; 100-1 000 m a.s.l. |
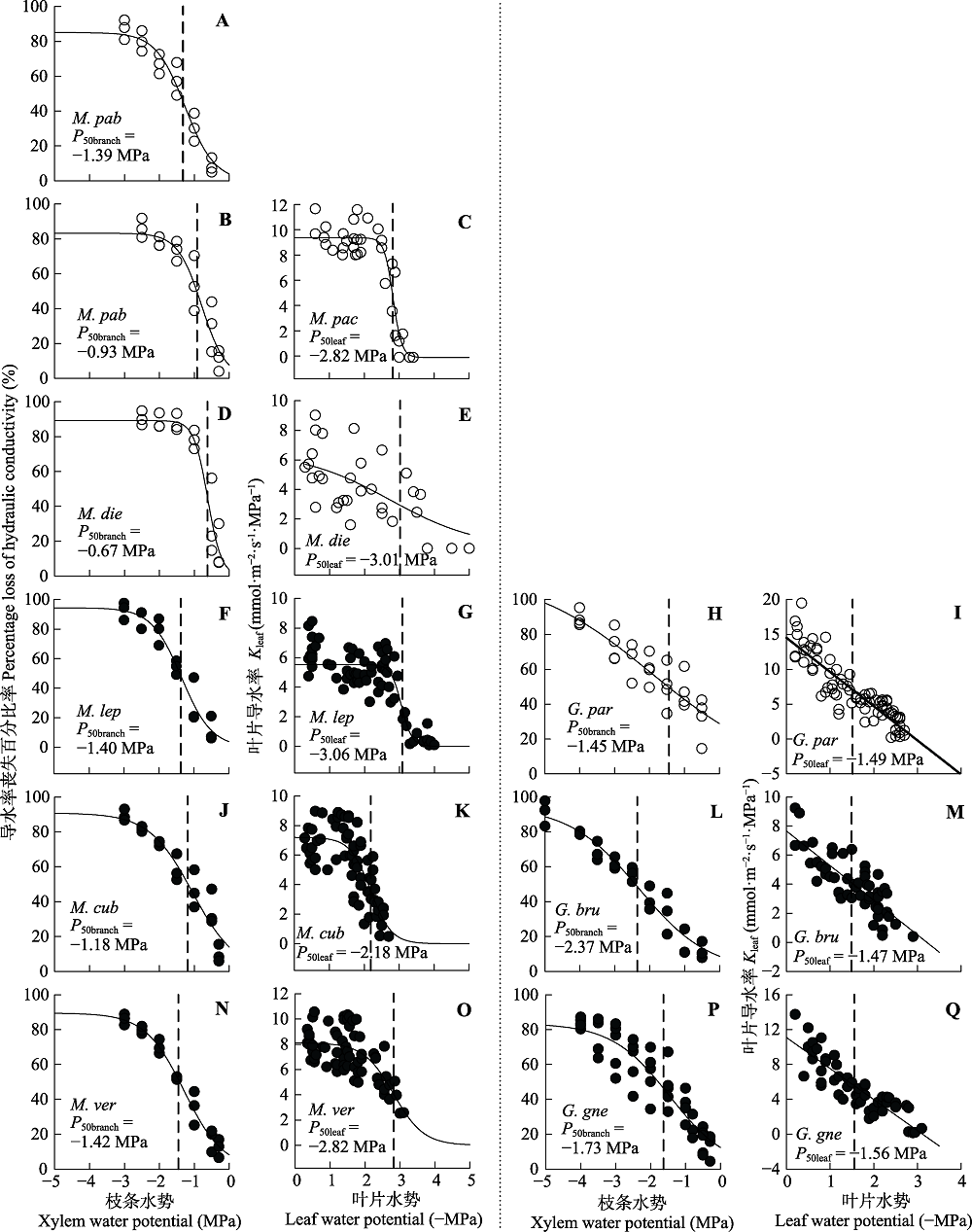
图1 崖豆藤属(左)与买麻藤属(右)木本植物的枝条和叶片脆弱性曲线。实心和空心圆分别代表乔木和木质藤本。树种名称缩写见表1。垂直虚线表示枝条与叶片导水率损失50%时的水势值(P50branch和P50leaf)。
Fig. 1 Branch and leaf vulnerability curves of the woody species in Millettia (left) and Gnetum (right). Filled and open circles indicate tree and liana species, respectively. Species abbreviations are shown in Table 1. Water potential at 50% loss of branch hydraulic conductivity (P50branch) and leaf hydraulic conductance (P50leaf) are indicated by vertical dashed lines. Kleaf, leaf hydraulic conductivity.
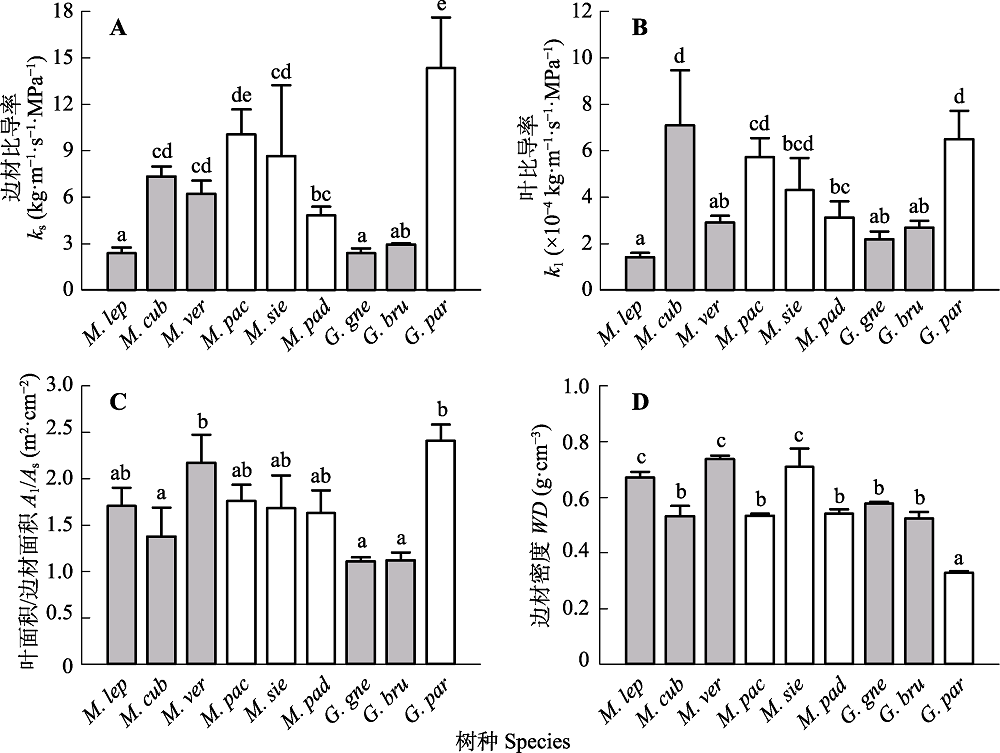
图2 崖豆藤属与买麻藤属9种植物的边材比导率(ks)、叶比导率(kl)、叶面积/边材面积(Al/As)和边材密度(WD)的比较(平均值+标准误差)。实心和空心柱分别表示乔木和木质藤本。不同字母表示各指标之间有显著性差异(p < 0.05)。物种名称的缩写见表1。
Fig. 2 Comparison in branch hydraulic traits among the nine woody species in Millettia and Gnetum (mean + SE). ks, sapwood specific hydraulic conductivity; kl, leaf specific hydraulic conductivity; Al/As, leaf area/sapwood area ratio; WD, sapwood density. Filled and open bars indicate tree and liana species, respectively. Different letters indicate significant difference at p < 0.05. Species abbreviations are shown in Table 1.
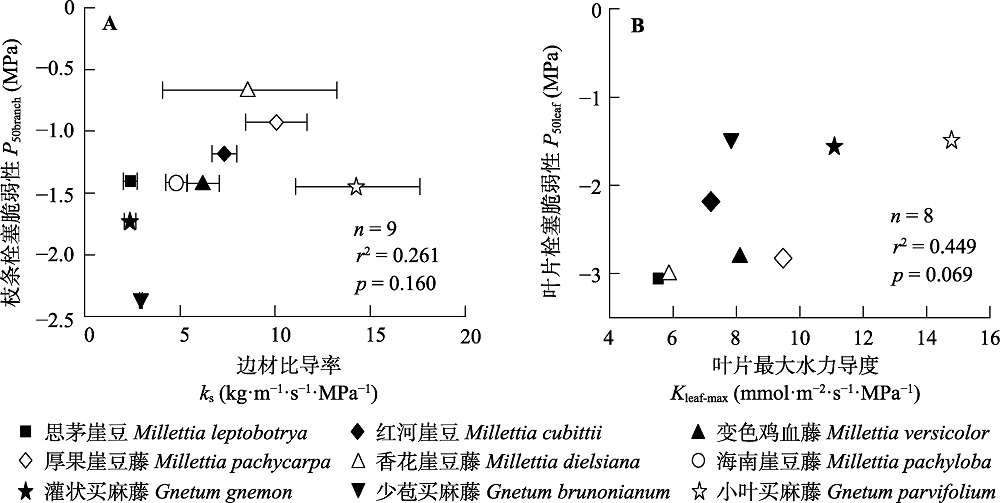
图3 植物水分传导有效性与安全性之间的相关关系。A, 枝条木质部抗栓塞能力(P50branch)与边材比导率(ks)之间的相关关系; B, 叶片抗栓塞能力(P50leaf)与叶片最大导水率(Kleaf-max)之间的相关关系。误差线为标准误差。
Fig. 3 Relationship between sapwood specific hydraulic conductivity (ks) and vulnerability to cavitation in branches (A), and relationship between maximum hydraulic leaf conductance (Kleaf-max) and vulnerability to cavitation in leaves (B). P50branch, xylem water potential at 50% loss of branch hydraulic conductivity; P50leaf, leaf water potential at 50% loss of leaf hydraulic conductance. Error bars are standard errors.
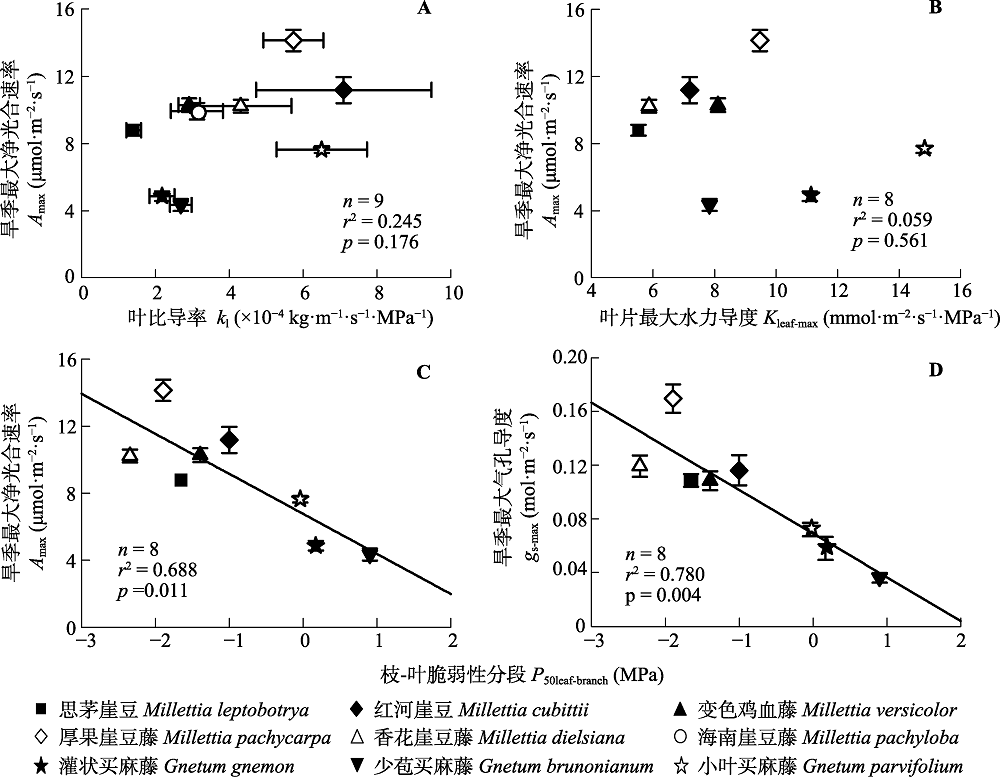
图4 水力性状与旱季叶片气体交换速率之间的相关关系。A、B, 旱季最大净光合速率(Amax)与叶比导率(kl)和叶片水力导度(Kleaf-max)之间的相关性; C、D, 枝-叶脆弱性分段(P50leaf-branch)与旱季最大净光合速率(Amax)和气孔导度(gs-max)之间的相关关系。误差线为标准误差。
Fig. 4 Relationships between maximum net photosynthetic rates during the dry season (Amax) and leaf specific hydraulic conductivity (kl)(A), or maximum leaf hydraulic conductivity (Kleaf-max)(B), and relationships between the difference in P50 between leaves and branches (P50leaf-branch) and Amax (C) or maximum stomatal conductance during the dry season (gs-max)(D). Error bars are standard errors.
| [1] |
Allen CD, MacAlady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EHT, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Semerci A, Cobb N (2010). A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. Forest Ecology and Management, 259, 660-684.
DOI URL |
| [2] | Angyalossy V, Pace MR, Lima AC (2014). Liana anatomy: a broad perspective on structural evolution of the vascular system//Schnitzer SA, Bongers F, Burnham RJ, Putz FE. Ecology of Lianas. John Wiley & Sons, Chichester, UK. 253-287. |
| [3] |
Bittencourt PRL, Pereira L, Oliveira RS (2016). On xylem hydraulic efficiencies, wood space-use and the safety-efficiency tradeoff. New Phytologist, 211, 1152-1155.
DOI URL |
| [4] |
Blackman CJ, Brodribb TJ, Jordan GJ (2010). Leaf hydraulic vulnerability is related to conduit dimensions and drought resistance across a diverse range of woody angiosperms. New Phytologist, 188, 1113-1123.
DOI URL |
| [5] |
Brodribb TJ, Bowman DJMS, Nichols S, Delzon S, Burlett R (2010). Xylem function and growth rate interact to determine recovery rates after exposure to extreme water deficit. New Phytologist, 188, 533-542.
DOI URL |
| [6] | Brodribb TJ, Feild TS (2000). Stem hydraulic supply is linked to leaf photosynthetic capacity: evidence from New Caledonian and Tasmanian rainforests. Plant, Cell & Environment, 23, 1381-1388. |
| [7] |
Brodribb TJ, Holbrook NM (2003). Stomatal closure during leaf dehydration, correlation with other leaf physiological traits. Plant Physiology, 132, 2166-2173.
DOI URL |
| [8] |
Brodribb TJ, Holbrook NM (2004). Stomatal protection against hydraulic failure: a comparison of coexisting ferns and angiosperms. New Phytologist, 162, 663-670.
DOI URL |
| [9] | Brodribb TJ, Holbrook NM (2006). Declining hydraulic efficiency as transpiring leaves desiccate: two types of response. Plant, Cell & Environment, 29, 2205-2215. |
| [10] |
Bucci SJ, Scholz FG, Campanello PI, Montti L, Jimenez- Castillo M, Rockwell FA, Manna LL, Guerra P, Bernal PL, Troncoso O, Enricci J, Holbrook MN, Goldstein G (2012). Hydraulic differences along the water transport system of South American Nothofagus species: Do leaves protect the stem functionality? Tree Physiology, 32, 880-893.
DOI URL |
| [11] |
Buckley TN, John GP, Scoffoni C, Sack L (2015). How does leaf anatomy influence water transport outside the xylem? Plant Physiology, 168, 1616-1635.
DOI URL |
| [12] |
Cai ZQ, Schnitzer SA, Bongers F (2009). Seasonal differences in leaf-level physiology give lianas a competitive advantage over trees in a tropical seasonal forest. Oecologia, 161, 25-33.
DOI URL |
| [13] |
Campbell MJ, Edwards W, Magrach A, Alamgir M, Porolak G, Mohandass D, Laurance WF (2018). Edge disturbance drives liana abundance increase and alteration of Liana- host tree interactions in tropical forest fragments. Ecology and Evolution, 8, 4237-4251.
DOI URL |
| [14] | Celis G, Avalos G (2013). Acclimation of seedlings of Gnetum leyboldii Tul.(Gnetaceae) to light changes in a tropical rain forest. Revista De Biologia Tropical, 61, 1859-1868. |
| [15] |
Chen YJ, Cao KF, Schnitzer SA, Fan ZX, Zhang JL, Bongers F (2015). Water-use advantage for lianas over trees in tropical seasonal forests. New Phytologist, 205, 128-136.
DOI URL |
| [16] | Choat B, Drayton WM, Brodersen C, Mattthews MA, Shackel KA, Wada H, McElrone AJ (2010). Measurement of vulnerability to water stress-induced cavitation in grapevine: a comparison of four techniques applied to a long-vesseled species. Plant, Cell & Environment, 33, 1502-1512. |
| [17] |
Choat B, Jansen S, Brodribb TJ, Cochard H, Delzon S, Bhaskar R, Bucci SJ, Feild TS, Gleason SM, Hacke UG, Jacobsen AL, Lens F, Maherali H, Martínez-Vilalta J, Mayr S, Mencuccini M, Mitchell PJ, Nardini A, Pittermann J, Pratt RB, Sperry JS, Westoby M, Wright IJ, Zanne AE (2012). Global convergence in the vulnerability of forests to drought. Nature, 491, 752-755.
DOI URL |
| [18] |
Cochard H, Cruiziat P, Tyree MT (1992). Use of positive pressures to establish vulnerability curves: further support for the air-seeding hypothesis and implications for pressure- volume analysis. Plant Physiology, 100, 205-209.
DOI URL |
| [19] |
Collins CG, Wright SJ, Wurzburger N (2016). Root and leaf traits reflect distinct resource acquisition strategies in tropical lianas and trees. Oecologia, 180, 1037-1047.
DOI URL |
| [20] |
Cruiziat P, Cochard H, Améglio T (2002). Hydraulic architecture of trees: main concepts and results. Annals of Forest Science, 59, 723-752.
DOI URL |
| [21] | de Guzman ME, Santiago LS, Schnitzer SA, Álvarez-Cansino L (2017). Trade-offs between water transport capacity and drought resistance in neotropical canopy liana and tree species. Tree Physiology, 37, 1404-1414. |
| [22] |
Deng N, Hou C, Liu CX, Li MH, Bartish I, Tian YX, Chen W, Du CJ, Jiang ZP, Shi SQ (2019). Significance of photosynthetic characters in the evolution of Asian Gnetum(Gnetales). Frontiers in Plant Science, 10, 39. DOI: 10.3389/fpls.2019.00039.
DOI URL |
| [23] |
Doyle JA, Donoghue MJ (1986). Seed plant phylogeny and the origin of angiosperms: an experimental cladistic approach. The Botanical Review, 52, 321-431.
DOI URL |
| [24] | Editorial Committee of Flora of China, Chinese Academy of Sciences (1993). Flora Reipublicae Popularis Sinicae. Science Press, Beijing. |
| [ 中国科学院中国植物志编辑委员会(1993). 中国植物志. 科学出版社, 北京.] | |
| [25] |
Ennajeh M, Simões F, Khemira H, Cochard H (2011). How reliable is the double-ended pressure sleeve technique for assessing xylem vulnerability to cavitation in woody angiosperms? Physiologia Plantarum, 142, 205-210.
DOI URL |
| [26] | Ewers FW, Rosell JA, Olson ME (2015). Lianas as structural parasites//Hacke U. Functional and Ecological Xylem Anatomy. Springer,Cham. 163-188. |
| [27] |
Feild TS, Balun L (2008). Xylem hydraulic and photosynthetic function of Gnetum(Gnetales) species from Papua New Guinea. New Phytologist, 177, 665-675.
DOI URL |
| [28] |
Fu PL, Jiang YJ, Wang AY, Brodribb TJ, Zhang JL, Zhu SD, Cao KF (2012). Stem hydraulic traits and leaf water-stress tolerance are co-ordinated with the leaf phenology of angiosperm trees in an Asian tropical dry karst forest. Annals of Botany, 110, 189-199.
DOI URL |
| [29] |
Gleason SM, Westoby M, Jansen S, Choat B, Hacke UG, Pratt RB, Bhaskar R, Brodribb TJ, Bucci SJ, Cao KF, Cochard H, Delzon S, Domec JC, Fan ZX, Feild TS, Jacobsen AL, Johnson DM, Lens F, Maherali H, Martínez-Vilalta J, Mayr S, McCulloh KA, Mencuccini M, Mitchell PJ, Morris H, Nardini A, Pittermann J, Plavcová L, Schreiber SG, Sperry JS, Wright IJ, Zanne AE (2016). Weak tradeoff between xylem safety and xylem-specific hydraulic efficiency across the world’s woody plant species. New Phytologist, 209, 123-136.
DOI URL |
| [30] | Isnard S, Feild TS (2014). The evolution of angiosperm lianescence: a perspective from xylem structure-function //Schnitzer SA, Bongers F, Burnham RJ, Putz FE. Ecology of Lianas. John Wiley & Sons,Chichester,UK. 221-238. |
| [31] |
Isnard S, Silk WK (2009). Moving with climbing plants from Charles Darwin’s time into the 21st century. American Journal of Botany, 96, 1205-1221.
DOI URL |
| [32] | Kotowska MM, Hertel D, Rajab YA, Barus H, Schuldt B (2015). Patterns in hydraulic architecture from roots to branches in six tropical tree species from cacao agroforestry and their relation to wood density and stem growth. Frontiers in Plant Science, 6, 191. DOI: 10.3389/fpls.2015.00191. |
| [33] | Li YH, Pei SJ, Xu ZF (1996). List of Palnt in Xishuangbanna. Yunnan Nationality Press, Kunming. |
| [ 李延辉, 裴盛基, 许再富 (1996). 西双版纳高等植物名录. 云南民族出版社, 昆明.] | |
| [34] |
Ma RY, Zhang JL, Cavaleri MA, Sterck F, Strijk JS, Cao KF (2015). Convergent evolution towards high net carbon gain efficiency contributes to the shade tolerance of palms (Arecaceae). PLOS ONE, 10, e0140384. DOI: 10.1371/journal.pone.0140384.
DOI URL |
| [35] |
Maréchaux I, Bartlett MK, Iribar A, Sack L, Chave J (2017). Stronger seasonal adjustment in leaf turgor loss point in lianas than trees in an Amazonian forest. Biology Letters, 13, 20160819. DOI: 10.1098/rsbl.2016.0819.
DOI URL |
| [36] |
Markesteijn L, Poorter L, Bongers F, Paz H, Sack L (2011). Hydraulics and life history of tropical dry forest tree species: coordination of species’ drought and shade tolerance. New Phytologist, 191, 480-495.
DOI URL |
| [37] | McCulloh KA, Johnson DM, Meinzer FC, Woodruff DR (2014). The dynamic pipeline: hydraulic capacitance and xylem hydraulic safety in four tall conifer species. Plant, Cell & Environment, 37, 1171-1183. |
| [38] |
Meinzer FC, McCulloh KA, Lachenbruch B, Woodruff DR, Johnson DM (2010). The blind men and the elephant: the impact of context and scale in evaluating conflicts between plant hydraulic safety and efficiency. Oecologia, 164, 287-296.
DOI URL |
| [39] |
Mencuccini M, Minunno F, Salmon Y, Martínez-Vilalta J, Hölttä T (2015). Coordination of physiological traits involved in drought-induced mortality of woody plants. New Phytologist, 208, 396-409.
DOI URL |
| [40] |
Pammenter NW, van der Willigen C (1998). A mathematical and statistical analysis of the curves illustrating vulnerability of xylem to cavitation. Tree Physiology, 18, 589-593.
DOI URL |
| [41] |
Pivovaroff AL, Pasquini SC, de Guzman ME, Alstad KP, Stemke JS, Santiago LS (2016). Multiple strategies for drought survival among woody plant species. Functional Ecology, 30, 517-526.
DOI URL |
| [42] |
Putz FE (1984). The natural history of lianas on barro Colorado Island, Panama. Ecology, 65, 1713-1724.
DOI URL |
| [43] |
Rodríguez-Ronderos ME, Bohrer G, Sanchez-Azofeifa A, Powers JS, Schnitzer SA (2016). Contribution of lianas to plant area index and canopy structure in a Panamanian forest. Ecology, 97, 3271-3277.
DOI URL |
| [44] |
Sack L, Frole K (2006). Leaf structural diversity is related to hydraulic capacity in tropical rain forest trees. Ecology, 87, 483-491.
DOI URL |
| [45] |
Sack L, Holbrook NM (2006). Leaf hydraulics. Annual Review of Plant Biology, 57, 361-381.
DOI URL |
| [46] |
Santiago LS, de Guzman ME, Baraloto C, Vogenberg JE, Brodie M, Hérault B, Fortunel C, Bonal D (2018). Coordination and trade-offs among hydraulic safety, efficiency and drought avoidance traits in Amazonian rainforest canopy tree species. New Phytologist, 218, 1015-1024.
DOI URL |
| [47] |
Santiago LS, Goldstein G, Meinzer FC, Fisher JB, MacHado K, Woodruff D, Jones T (2004). Leaf photosynthetic traits scale with hydraulic conductivity and wood density in Panamanian forest canopy trees. Oecologia, 140, 543-550.
DOI URL |
| [48] | Santiago LS, Pasquini SC, de Guzman ME (2014). Physiological implications of the liana growth form//Schnitzer SA, Bongers F, Burnham RJ, Putz FE. Ecology of Lianas. John Wiley & Sons, Chichester, UK. 288-298. |
| [49] | Santiago LS, Wright SJ (2007). Leaf functional traits of tropical forest plants in relation to growth form. Functional Ecology, 21, 19-27. |
| [50] |
Schnitzer SA (2005). A mechanistic explanation for global patterns of liana abundance and distribution. The American Naturalist, 166, 262-276.
DOI URL |
| [51] |
Schnitzer SA, Bongers F (2011). Increasing liana abundance and biomass in tropical forests: emerging patterns and putative mechanisms. Ecology Letters, 14, 397-406.
DOI URL |
| [52] |
Schnitzer SA, Dalling JW, Carson WP (2000). The impact of lianas on tree regeneration in tropical forest canopy gaps: evidence for an alternative pathway of gap-phase regeneration. Journal of Ecology, 88, 655-666.
DOI URL |
| [53] |
Schnitzer SA, van der Heijden GMF (2019). Lianas have a seasonal growth advantage over co-occurring trees. Ecology, 100, e02655. DOI: 10.1002/ecy.2655.
DOI URL |
| [54] |
Schulte PJ, Hinckley TM (1985). A comparison of pressure- volume curve data analysis techniques. Journal of Experimental Botany, 36, 1590-1602.
DOI URL |
| [55] |
Scoffoni C, Albuquerque C, Brodersen CR, Townes SV, John GP, Bartlett MK, Buckley TN, McElrone AJ, Sack L (2017a). Outside-xylem vulnerability, not xylem embolism, controls leaf hydraulic decline during dehydration. Plant Physiology, 173, 1197-1210.
DOI URL |
| [56] |
Scoffoni C, Albuquerque C, Brodersen CR, Townes SV, John GP, Cochard H, Buckley TN, McElrone AJ, Sack L (2017b). Leaf vein xylem conduit diameter influences susceptibility to embolism and hydraulic decline. New Phytologist, 213, 1076-1092.
DOI URL |
| [57] |
Scoffoni C, Vuong C, Diep S, Cochard H, Sack L (2014). Leaf shrinkage with dehydration: coordination with hydraulic vulnerability and drought tolerance. Plant Physiology, 164, 1772-1788.
DOI URL |
| [58] |
Smith-Martin CM, Skelton RP, Johnson KM, Lucani C, Brodribb TJ (2020). Lack of vulnerability segmentation among woody species in a diverse dry sclerophyll woodland community. Functional Ecology, 34, 777-787.
DOI URL |
| [59] | Sperry JS, Donnelly JR, Tyree MT (1988). A method for measuring hydraulic conductivity and embolism in xylem. Plant, Cell & Environment, 11, 35-40. |
| [60] |
Sperry JS, Saliendra NZ (1994). Intra- and inter-plant variation in xylem cavitation in Betula occidentalis. Plant, Cell & Environment, 17, 1233-1241.
DOI URL |
| [61] |
Tomlinson PB, Fisher JB (2005). Development of nonlignified fibers in leaves of Gnetum gnemon(Gnetales). American Journal of Botany, 92, 383-389.
DOI URL |
| [62] |
Trifiló P, Raimondo F, Savi T, Lo Gullo MA, Nardini A (2016). The contribution of vascular and Extra-vascular water pathways to drought-induced decline of leaf hydraulic conductance. Journal of Experimental Botany, 67, 5029-5039.
DOI URL |
| [63] |
Tyree MT, Ewers FW (1991). The hydraulic architecture of trees and other woody plants. New Phytologist, 119, 345-360.
DOI URL |
| [64] |
van der Sande MT, Poorter L, Schnitzer SA, Engelbrecht BMJ, Markesteijn L (2019). The hydraulic efficiency-safety trade-off differs between lianas and trees. Ecology, 100, e02666. DOI: 10.1002/ecy.2666.
DOI URL |
| [65] |
Villagra M, Campanello PI, Bucci SJ, Goldstein G (2013). Functional relationships between leaf hydraulics and leaf economic traits in response to nutrient addition in subtropical tree species. Tree Physiology, 33, 1308-1318.
DOI URL |
| [66] | Yin PX, Cai J (2018). New possible mechanisms of embolism formation when measuring vulnerability curves by air injection in a pressure sleeve. Plant, Cell & Environment, 41, 1361-1368. |
| [67] |
Zhang L, Chen YJ, Ma KP, Bongers F, Sterck FJ (2019). Fully exposed canopy tree and liana branches in a tropical forest differ in mechanical traits but are similar in hydraulic traits. Tree Physiology, 39, 1713-1724.
DOI URL |
| [68] |
Zhu SD, Cao KF (2009). Hydraulic properties and photosynthetic rates in co-occurring lianas and trees in a seasonal tropical rainforest in southwestern China. Plant Ecology, 204, 295-304.
DOI URL |
| [69] |
Zhu SD, Chen YJ, Cao KF, Ye Q (2015). Interspecific variation in branch and leaf traits among three Syzygium tree species from different successional tropical forests. Functional Plant Biology, 42, 423-432.
DOI URL |
| [70] |
Zhu SD, Chen YJ, Fu PL, Cao KF (2017). Different hydraulic traits of woody plants from tropical forests with contrasting soil water availability. Tree Physiology, 37, 1469-1477.
DOI URL |
| [71] |
Zhu SD, Liu H, Xu QY, Cao KF, Ye Q (2016). Are leaves more vulnerable to cavitation than branches? Functional Ecology, 30, 1740-1744.
DOI URL |
| [72] | Zimmerman MH, Brown CL (1971). Trees: Structure and Function. Springer-Verlag, New York. |
| [1] | 冯旭飞, 雷长英, 张玉洁, 向导, 杨明凤, 张旺锋, 张亚黎. 棉花花铃期叶片氮分配对光合氮利用效率的影响[J]. 植物生态学报, 2023, 47(11): 1600-1610. |
| [2] | 和璐璐, 张萱, 章毓文, 王晓霞, 刘亚栋, 刘岩, 范子莹, 何远洋, 席本野, 段劼. 辽东山区不同坡向长白落叶松人工林树冠特征与林木生长关系[J]. 植物生态学报, 2023, 47(11): 1523-1539. |
| [3] | 黄冬柳, 项伟, 李忠国, 朱师丹. 南亚热带10种造林树种的水力结构和水力安全[J]. 植物生态学报, 2022, 46(5): 602-612. |
| [4] | 林雍, 陈智, 杨萌, 陈世苹, 高艳红, 刘冉, 郝彦宾, 辛晓平, 周莉, 于贵瑞. 中国干旱半干旱区生态系统光合参数的时空变异及其影响因素[J]. 植物生态学报, 2022, 46(12): 1461-1472. |
| [5] | 李豪, 马如玉, 强波, 贺聪, 韩路, 王海珍. 胡杨当年生小枝茎构型对展叶效率的影响[J]. 植物生态学报, 2021, 45(11): 1251-1262. |
| [6] | 朱启林, 向蕊, 汤利, 龙光强. 间作对氮调控玉米光合速率和光合氮利用效率的影响[J]. 植物生态学报, 2018, 42(6): 672-680. |
| [7] | 朱良军, 李宗善, 王晓春. 树轮木质部解剖特征及其与环境变化的关系[J]. 植物生态学报, 2017, 41(2): 238-251. |
| [8] | 李志民, 王传宽, 罗丹丹. 兴安落叶松叶水力与光合性状的变异性和相关性[J]. 植物生态学报, 2017, 41(11): 1140-1148. |
| [9] | 金鹰, 王传宽. 九种不同材性的温带树种叶水力性状及其权衡关系[J]. 植物生态学报, 2016, 40(7): 702-710. |
| [10] | 赵文赛, 孙永林, 刘西平. 干旱-复水-再干旱处理对玉米光合能力和生长的影响[J]. 植物生态学报, 2016, 40(6): 594-603. |
| [11] | 赵延涛, 许洺山, 张志浩, 周刘丽, 张晴晴, 宋彦君, 阎恩荣. 浙江天童常绿阔叶林不同演替阶段木本植物的水力结构特征[J]. 植物生态学报, 2016, 40(2): 116-126. |
| [12] | 司晓林, 王文银, 高小刚, 徐当会. 氮硅添加对高寒草甸垂穗披碱草叶片全氮含量及净光合速率的影响[J]. 植物生态学报, 2016, 40(12): 1238-1244. |
| [13] | 唐海萍, 薛海丽, 房飞. 叶片和群落尺度净光合速率关系的探讨[J]. 植物生态学报, 2015, 39(9): 924-931. |
| [14] | 宋清华, 赵成章, 史元春, 杜晶, 王继伟, 陈静. 不同坡向甘肃臭草根系分叉数和连接长度的权衡关系[J]. 植物生态学报, 2015, 39(6): 577-585. |
| [15] | 杨浩, 罗亚晨. 糙隐子草功能性状对氮添加和干旱的响应[J]. 植物生态学报, 2015, 39(1): 32-42. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2026 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
![]()