

植物生态学报 ›› 2025, Vol. 49 ›› Issue (11): 1934-1943.DOI: 10.17521/cjpe.2024.0228 cstr: 32100.14.cjpe.2024.0228
甄玉琪, 邓晨茜, 鲍梦林, 臧纱纱, 严芳, 吴红艳*( )
)
收稿日期:2024-07-12
接受日期:2024-10-09
出版日期:2025-11-20
发布日期:2025-11-20
通讯作者:
*吴红艳(sdwuhongyan@126.com)基金资助:
ZHEN Yu-Qi, DENG Chen-Xi, BAO Meng-Lin, ZANG Sha-Sha, YAN Fang, WU Hong-Yan*( )
)
Received:2024-07-12
Accepted:2024-10-09
Online:2025-11-20
Published:2025-11-20
Supported by:摘要: 硅藻是海洋浮游植物的重要组成部分, 其光合产量达到海洋初级生产力的40%以上。阳光紫外辐射(UVR)增强和海水升温的耦合作用将会影响硅藻的光合作用, 进而影响其初级生产贡献。该研究主要探索升温对硅藻响应UVR的光合生理调控, 以进一步了解海洋环境变化对硅藻光合作用的影响。将威氏海链藻(Thalassiosira weissflogii)在18和24 ℃下培养, 并置于高可见光(光合有效辐射(PAR) 400-700 nm)和UVR (PAR+UVR, 280-700 nm)下, 以监测其光系统II (PSII)功能变化和相关生理响应。结果表明: PAR和PAR+UVR均对威氏海链藻PSII最大光化学效率(Fv/Fm)产生抑制作用, UVR存在下PSII光失活速率常数(Kpi)显著上升, 但升温时, UVR下威氏海链藻细胞具有与低温下相近的PSII修复速率常数与失活速率常数比值(Krec/Kpi)。对PSII蛋白亚基周转的分析表明, 可见光下升温使威氏海链藻细胞维持较高的D2蛋白(PsbD)库, 而在UVR下升温则协同促进受损D1蛋白(PsbA)的快速清除。此外, 升温条件下威氏海链藻细胞维持较高的超氧化物歧化酶和过氧化氢酶活性, 各处理下仅诱导产生较低水平的非光化学淬灭。该研究结果表明, 威氏海链藻在升温时可通过调节PSII修复循环以促进其光合性能对抗UVR的抑制效应。
甄玉琪, 邓晨茜, 鲍梦林, 臧纱纱, 严芳, 吴红艳. 海水升温及紫外辐射对威氏海链藻光合特性的影响. 植物生态学报, 2025, 49(11): 1934-1943. DOI: 10.17521/cjpe.2024.0228
ZHEN Yu-Qi, DENG Chen-Xi, BAO Meng-Lin, ZANG Sha-Sha, YAN Fang, WU Hong-Yan. Effects of ocean warming and ultraviolet radiation on the photosynthetic characteristics of Thalassiosira weissflogii. Chinese Journal of Plant Ecology, 2025, 49(11): 1934-1943. DOI: 10.17521/cjpe.2024.0228
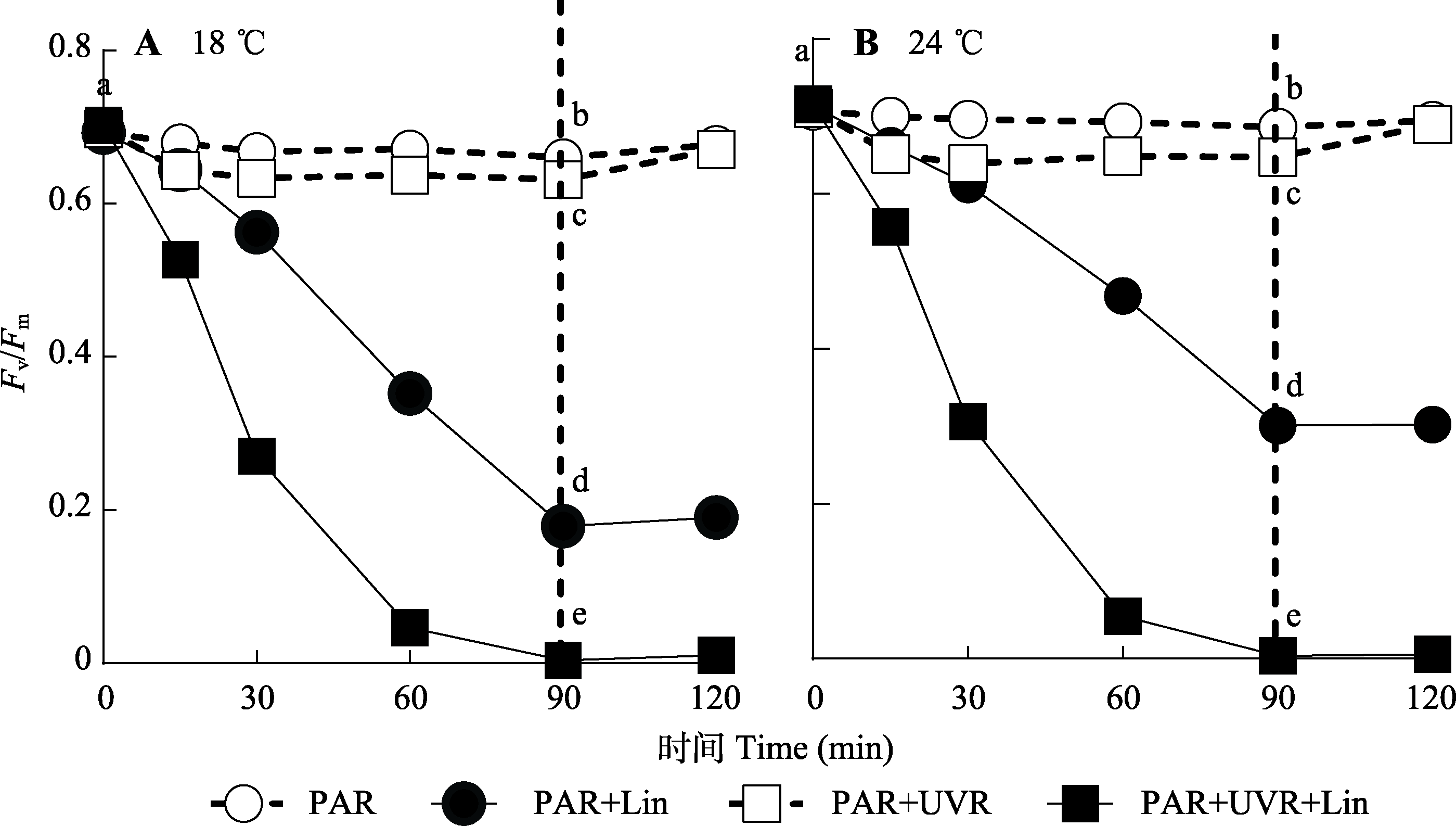
图1 18 ℃和24 ℃条件下, 威氏海链藻光系统II最大光化学效率(Fv/Fm)对光辐射的响应(平均值±标准误, n = 4)。藻细胞在PAR和PAR+UVR下照射90 min, 并在低光下恢复30 min。虚线表示高光暴露与恢复期的划分。Lin, 林肯霉素; PAR, 光合有效辐射; UVR, 紫外线辐射。不同小写字母表示处理间存在显著差异(p < 0.05)。
Fig. 1 Maximum photochemical yield (Fv/Fm) changes in Thalassiosira weissflogii cells grown at 18 °C or 24 °C treated without or with lincomycin (+Lin) when exposed to PAR or PAR + UVR for 90 min and low growth light for 30 min (mean ± SE, n = 4). The division between high light exposure and recover period was indicated by the dotted line. PAR, photosynthetically active radiation; UVR, ultraviolent radiation. Different lowercase letters indicate significant differences between treatments (p < 0.05).
| 温度 Temperature | 处理 Treatment | Krec | Kpi | Krec/Kpi |
|---|---|---|---|---|
| 18 ℃ | PAR | 0.005 459 ± 0.000 656 a | 0.000 189 ± 0.000 013 b | 28.9 |
| PAR+UVR | 0.005 403 ± 0.000 146 a | 0.000 521 ± 0.000 036 a | 10.4 | |
| 24 ℃ | PAR | 0.006 946 ± 0.001 114 a | 0.000 129 ± 0.000 008 c | 53.8 |
| PAR+UVR | 0.004 947 ± 0.000 247 b | 0.000 487 ± 0.000 003 a | 10.2 |
表1 不同处理下威氏海链藻光系统II (PSII)修复速率常数(Krec, s-1)和光失活速率常数(Kpi, s-1)及Krec与Kpi的比值(平均值±标准误, n = 4)
Table 1 Rate constant for photosystem II (PSII) repair (Krec, s-1) and photoinactivation (Kpi, s-1) and the ratio of Krec to Kpi for various treatments in Thalassiosira weissflogii (mean ± SE, n = 4)
| 温度 Temperature | 处理 Treatment | Krec | Kpi | Krec/Kpi |
|---|---|---|---|---|
| 18 ℃ | PAR | 0.005 459 ± 0.000 656 a | 0.000 189 ± 0.000 013 b | 28.9 |
| PAR+UVR | 0.005 403 ± 0.000 146 a | 0.000 521 ± 0.000 036 a | 10.4 | |
| 24 ℃ | PAR | 0.006 946 ± 0.001 114 a | 0.000 129 ± 0.000 008 c | 53.8 |
| PAR+UVR | 0.004 947 ± 0.000 247 b | 0.000 487 ± 0.000 003 a | 10.2 |
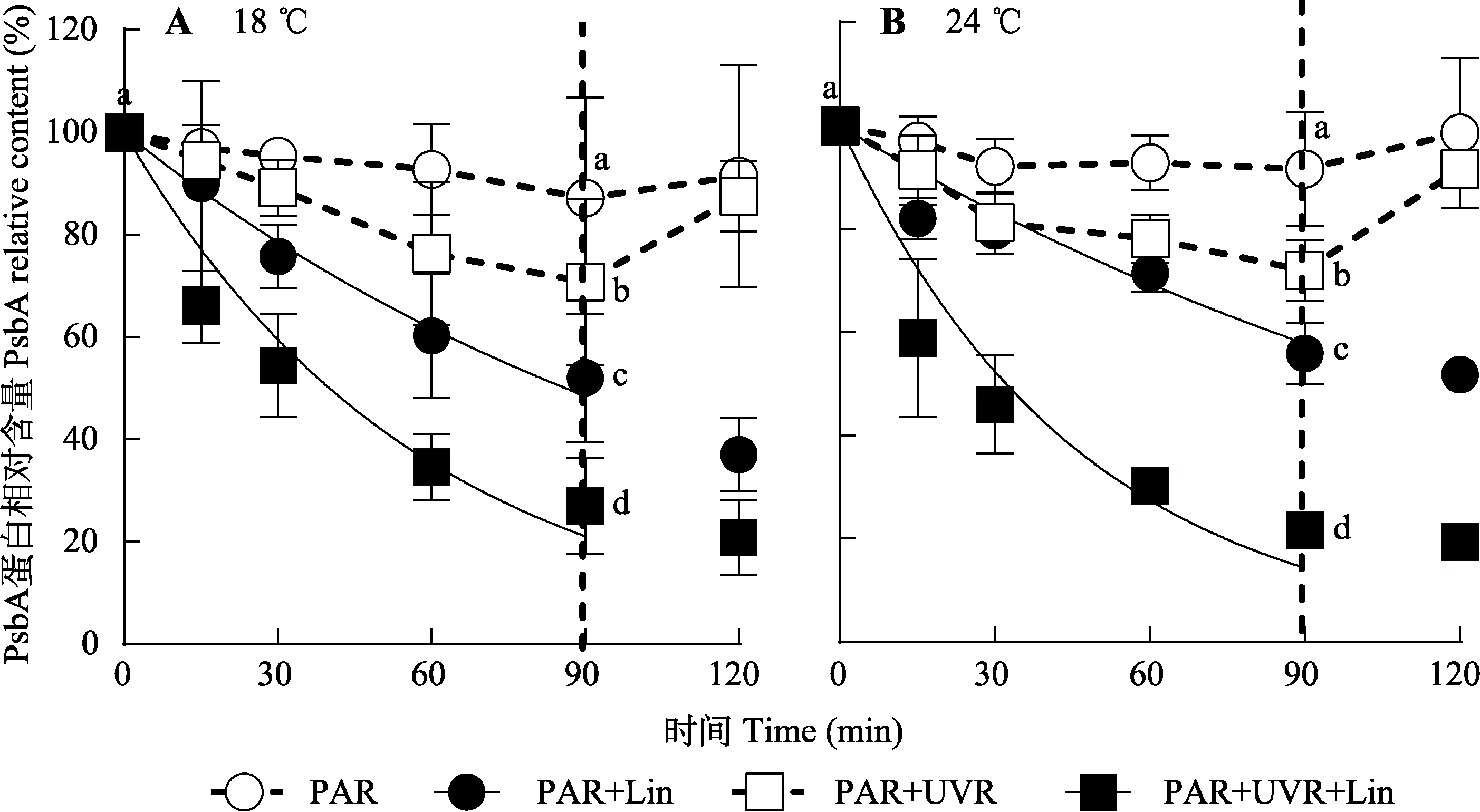
图2 18 ℃和24 ℃条件下光辐射对威氏海链藻PsbA蛋白相对含量的影响(平均值±标准误, n = 4)。藻细胞在PAR和PAR+ UVR下照射90 min, 并在低光下恢复30 min。虚线表示高光暴露与恢复期的划分。Lin, 林肯霉素; PAR, 光合有效辐射; UVR, 紫外线辐射。不同小写字母表示不同处理下存在显著差异(p < 0.05)。
Fig. 2 PsbA relative content changes in Thalassiosira weissflogii cells grown at 18 °C or 24 °C treated without or with lincomycin (+Lin) when exposed to PAR or PAR + UVR for 90 min and low growth light for 30 min (mean ± SE, n = 4). The division between high light exposure and recover period was indicated by the dotted line. PAR, photosynthetically active radiation; UVR, ultraviolet radiation. Different lowercase letters indicate significant differences under different treatments (p < 0.05).
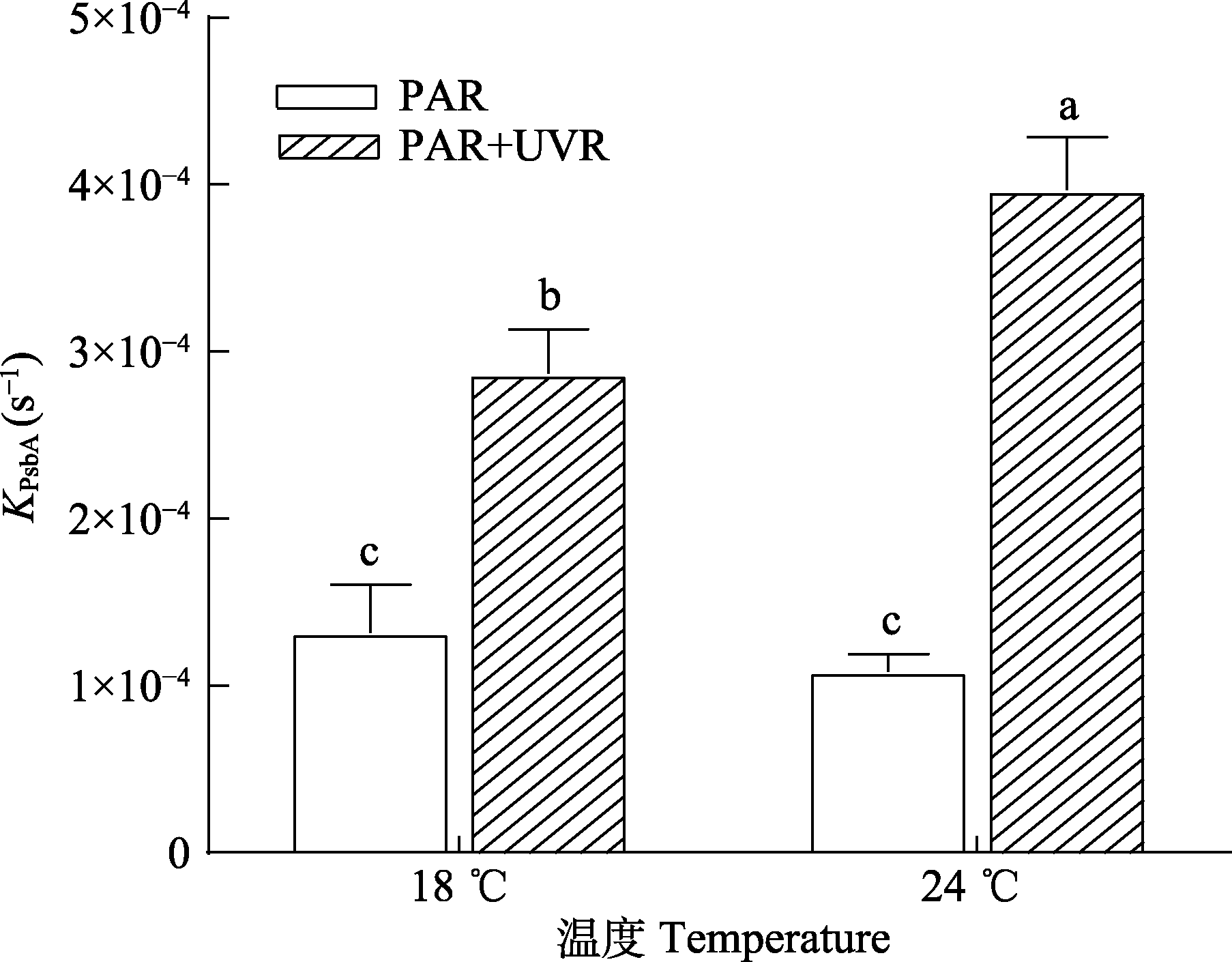
图3 18 ℃和24 ℃下威氏海链藻在不同辐射处理下PsbA清除速率常数(KPsbA)的变化(平均值±标准误, n = 4)。PAR, 光合有效辐射; UVR, 紫外辐射。不同小写字母表示不同处理间存在显著差异(p < 0.05)。
Fig. 3 Removal rate constant of PsbA (KPsbA) in Thalassiosira weissflogii grown at 18 °C or 24 °C (mean ± SE, n = 4). PAR, photosynthetically active radiation; UVR, ultraviolet radiation. Different lowercase letters indicate significant differences under different treatments (p < 0.05).
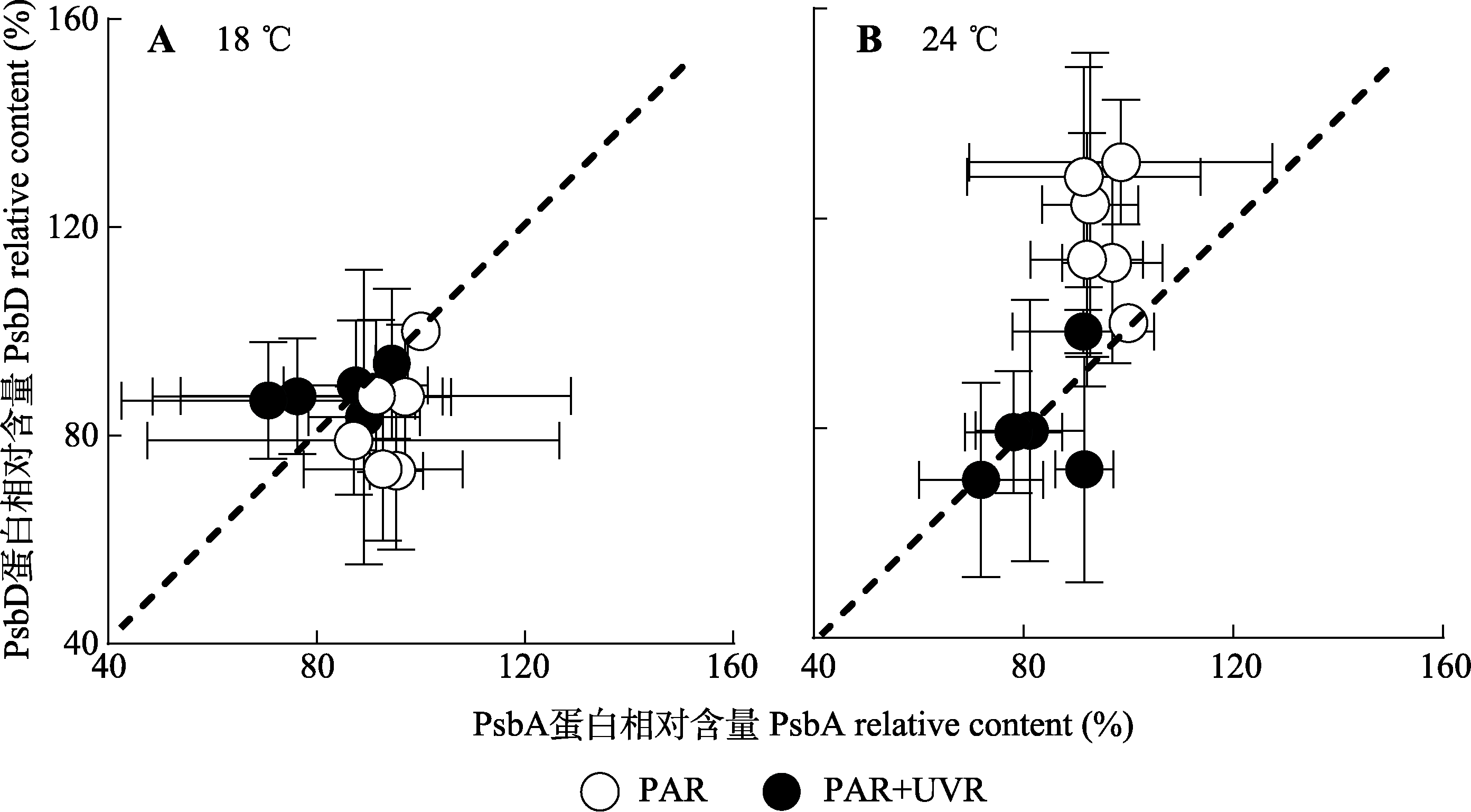
图4 18 ℃和24 ℃条件下PAR及PAR+UVR对威氏海链藻的PsbA和PsbD蛋白相对含量的影响(平均值±标准误, n = 3)。PAR, 光合有效辐射; UVR, 紫外辐射。
Fig. 4 PsbA relative content versus PsbD relative content in Thalassiosira weissflogii grown at 18 °C or 24 °C exposed to PAR and PAR + UVR (mean ± SE, n = 3). PAR, photosynthetically active radiation; UVR, ultraviolet radiation.
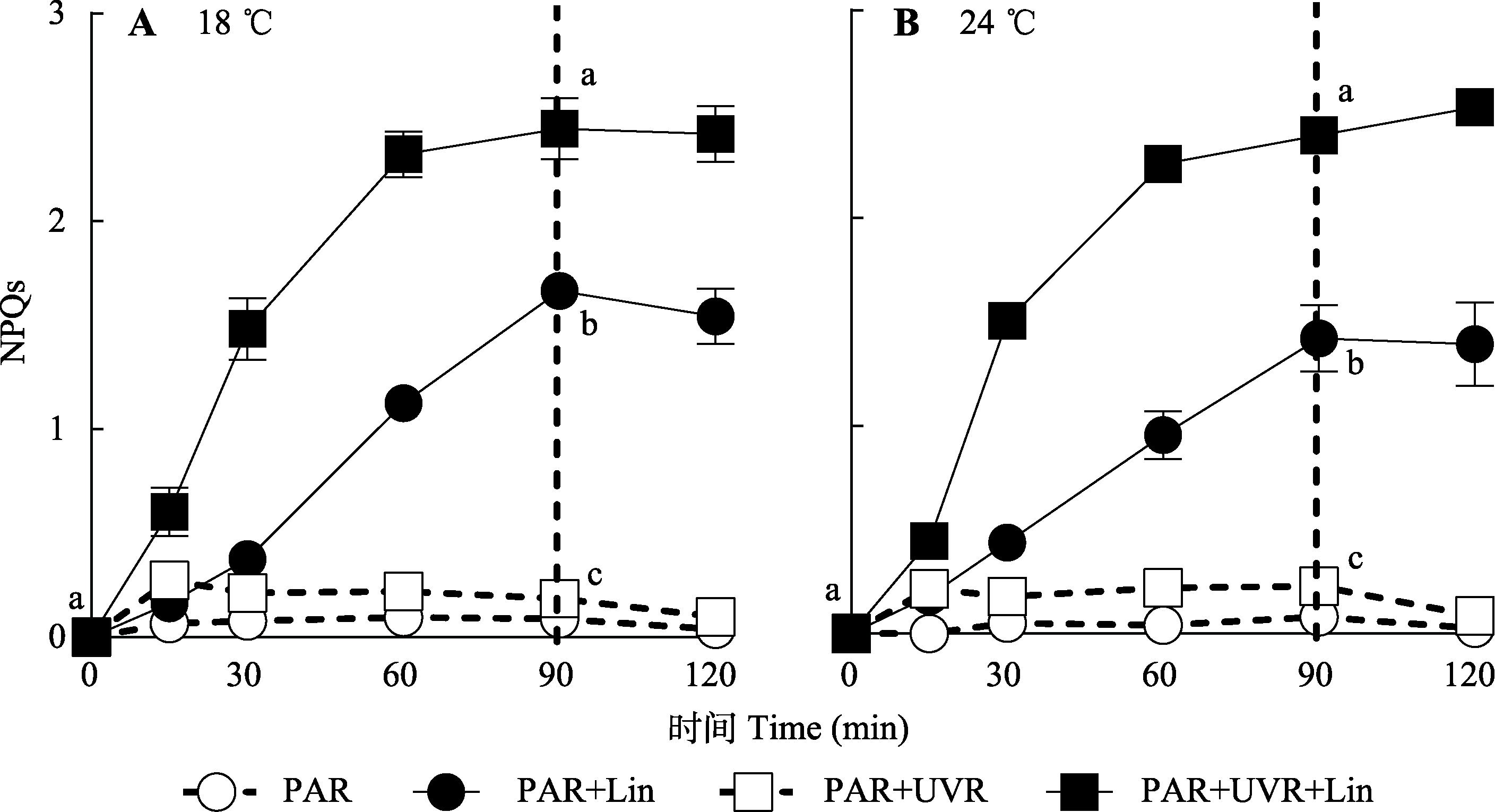
图5 18 ℃和24 ℃条件下光辐射对威氏海链藻非光化学淬灭(NPQs)的影响(平均值±标准误, n = 4)。藻细胞在PAR和PAR+ UVR下照射90 min, 并在低光下恢复30 min。虚线表示高光暴露与恢复期的划分。Lin, 林肯霉素; PAR, 光合有效辐射; UVR, 紫外线辐射。不同小写字母表示不同处理间存在显著差异(p < 0.05)。
Fig. 5 Non-photochemical quenching (NPQs) changes in Thalassiosira weissflogii cells grown at 18 °C or 24 °C treated without or with lincomycin (+Lin) when exposed to PAR or PAR + UVR for 90 min and low growth light for 30 min (mean ± SE, n = 4). The division between high light exposure and recover period was indicated by the dotted line. PAR, photosynthetically active radiation; UVR, ultraviolet radiation. Different lowercase letters indicate significant differences under different treatments (p < 0.05).
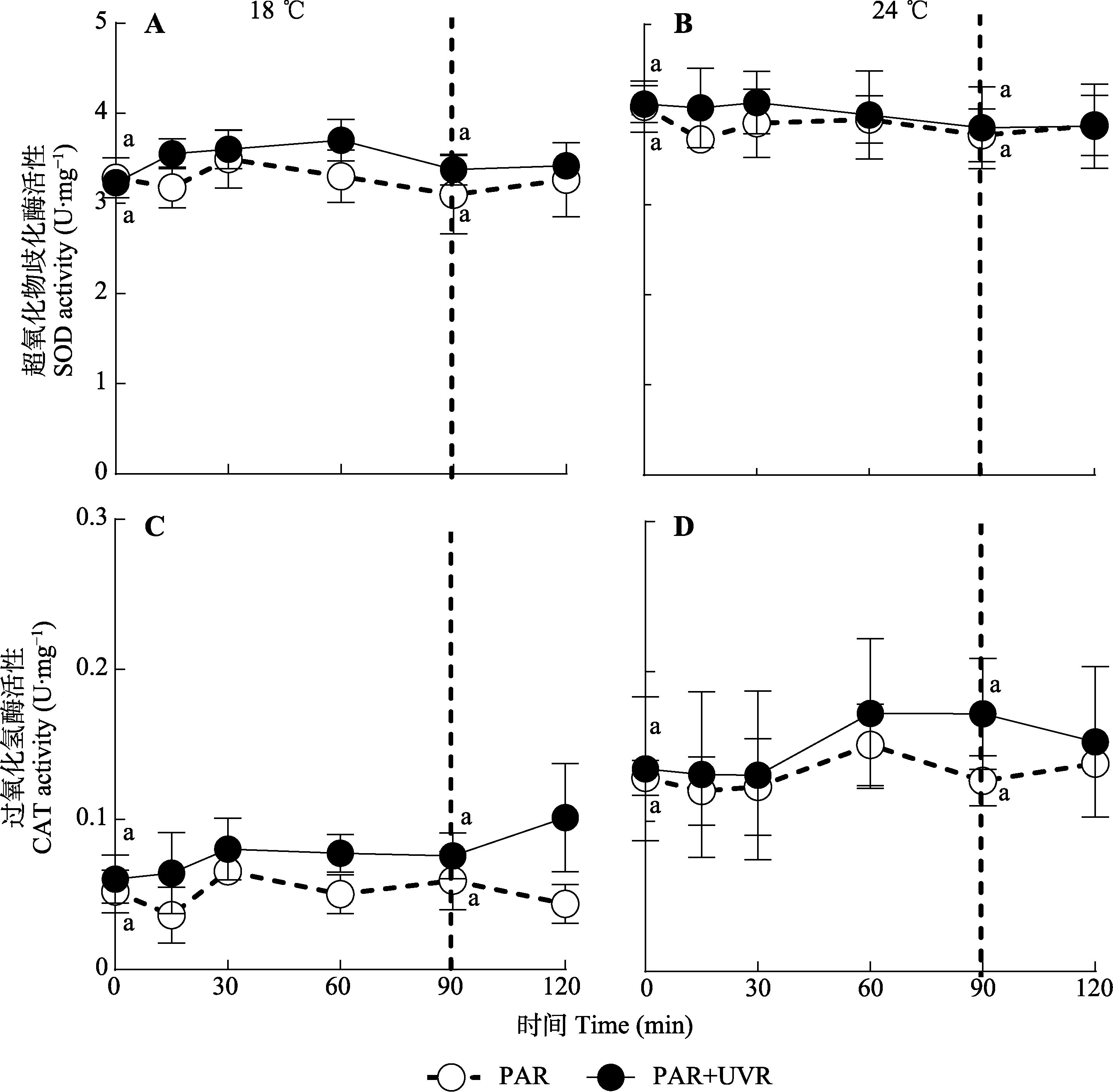
图6 18 ℃ (A、C)和24 ℃ (B、D)条件下光辐射对威氏海链藻超氧化物歧化酶(SOD)和过氧化氢酶(CAT)活性的影响(平均值±标准误, n = 4)。藻细胞在PAR和PAR+UVR下照射90 min, 并在低光下恢复30 min。虚线表示高光暴露与恢复期的划分。PAR, 光合有效辐射; UVR, 紫外线辐射。相同小写字母表示不同处理间不存在显著性差异(p ≥ 0.05)。
Fig. 6 Superoxide dismutase (SOD) and catalase (CAT) activity changes in Thalassiosira weissflogii cells grown at 18 °C (A, C) or 24 °C (B, D) when exposed to PAR or PAR + UVR for 90 min and low growth light for 30 min (mean ± SE, n = 4). The division between high light exposure and recover period was indicated by the dotted line. PAR, photosynthetically active radiation; UVR, ultraviolet radiation. Same lowercase letters indicate no significant differences under different treatments (p ≥ 0.05).
| [1] |
Campbell D, Eriksson MJ, Öquist G, Gustafsson P, Clarke AK (1998). The cyanobacterium Synechococcus resists UV-B by exchanging photosystem II reaction center D1 proteins. Proceedings of the National Academy of Sciences of the United States of America, 95, 364-369.
DOI PMID |
| [2] |
Campbell DA, Tyystjärvi E (2012). Parameterization of photosystem II photoinactivation and repair. Biochimica et Biophysica Acta, 1817, 258-265.
DOI PMID |
| [3] |
Cao L, Bi D, Fan W, Xu J, Beardall J, Gao K, Wu Y (2024). Warming exacerbates the impacts of ultraviolet radiation in temperate diatoms but alleviates the effect on polar species. Photochemistry and Photobiology, 100, 491-498.
DOI URL |
| [4] |
Cross WF, Hood JM, Benstead JP, Huryn AD, Nelson D (2015). Interactions between temperature and nutrients across levels of ecological organization. Global Change Biology, 21, 1025-1040.
DOI PMID |
| [5] |
Dobretsov S, Coutinho R, Rittschof D, Salta M, Ragazzola F, Hellio C (2019). The oceans are changing: impact of ocean warming and acidification on biofouling communities. Biofouling, 35, 585-595.
DOI PMID |
| [6] |
Ebenhöh O, Spelberg S (2018). The importance of the photosynthetic Gibbs effect in the elucidation of the Calvin-Benson-Bassham cycle. Biochemical Society Transactions, 46, 131-140.
DOI PMID |
| [7] |
Edelman M, Mattoo AK (2008). D1-protein dynamics in photosystem II: the lingering enigma. Photosynthesis Research, 98, 609-620.
DOI PMID |
| [8] |
Gao G, Shi Q, Xu Z, Xu J, Campbell DA, Wu H (2018). Global warming interacts with ocean acidification to alter PSII function and protection in the diatom Thalassiosira weissflogii. Environmental and Experimental Botany, 147, 95-103.
DOI URL |
| [9] |
Gao K, Yu H, Brown MT (2007). Solar PAR and UV radiation affects the physiology and morphology of the Cyanobacterium anabaena sp. PCC 7120. Journal of Photochemistry and Photobiology B: Biology, 89, 117-124.
DOI URL |
| [10] | Gao KS, Beardall J, Häder DP, Hall-Spencer JM, Gao G, Hutchins DA (2019). Effects of ocean acidification on marine photosynthetic organisms under the concurrent influences of warming, UV radiation, and deoxygenation. Frontiers in Marine Science, 6, 322. DOI: 10.3389/fmars.2019.00322. |
| [11] | Gattuso JP, Magnan A, Billé R, Cheung WWL, Howes EL, Joos F, Allemand D, Bopp L, Cooley SR, Eakin CM, Hoegh-Guldberg O, Kelly RP, Pörtner HO, Rogers AD, Baxter JM, et al. (2015). Contrasting futures for ocean and society from different anthropogenic CO2 emissions scenarios. Science, 349, aac4722. DOI: 10.1126/science.aac4722. |
| [12] |
Goss R, Lepetit B (2015). Biodiversity of NPQ. Journal of Plant Physiology, 172, 13-32.
DOI PMID |
| [13] | Häder DP, Gao KS (2015). Interactions of anthropogenic stress factors on marine phytoplankton. Frontiers in Environmental Science, 3, 1-14. |
| [14] |
Halac SR, Villafañe VE, Gonçalves RJ, Helbling EW (2014). Photochemical responses of three marine phytoplankton species exposed to ultraviolet radiation and increased temperature: role of photoprotective mechanisms. Journal of Photochemistry and Photobiology B: Biology, 141, 217-227.
DOI URL |
| [15] |
Halac SR, Villafañe VE, Helbling EW (2010). Temperature benefits the photosynthetic performance of the diatoms Chaetoceros gracilis and Thalassiosira weissflogii when exposed to UVR. Journal of Photochemistry and photobiology B:Biology, 101, 196-205.
DOI URL |
| [16] |
Helbling EW, Buma AGJ, Boelen P, van der Strate HJ, Giordanino MVF, Villafañe VE (2011). Increase in Rubisco activity and gene expression due to elevated temperature partially counteracts ultraviolet radiation- induced photoinhibition in the marine diatom Thalassiosira weissflogii. Limnology and Oceanography, 56, 1330-1342.
DOI URL |
| [17] |
Jin P, Wan JF, Zhou YY, Gao KS, Beardall J, Lin JM, Huang JL, Lu YC, Liang SM, Wang KQ, Ma ZL, Xia JR (2022). Increased genetic diversity loss and genetic differentiation in a model marine diatom adapted to ocean warming compared to high CO2. The ISME Journal, 16, 2587-2598.
DOI URL |
| [18] |
Kok B (1956). On the inhibition of photosynthesis by intense light. Biochimica et Biophysica Acta, 21, 234-244.
PMID |
| [19] |
Komenda J, Sobotka R, Nixon PJ (2012). Assembling and maintaining the photosystem II complex in chloroplasts and cyanobacteria. Current Opinion in Plant Biology, 15, 245-251.
DOI PMID |
| [20] |
Kós PB, Deák Z, Cheregi O, Vass I (2008). Differential regulation of psbA and psbD gene expression, and the role of the different D 1 protein copies in the Cyanobacterium thermosynechococcus elongatus BP-1. Biochimica et Biophysica Acta - Bioenergetics, 1777, 74-83.
DOI URL |
| [21] | Krause GH, Jahns P (2007). Non-photochemical energy dissipation determined by chlorophyll fluorescence quenching: characterization and function// Papageorgiou GC, Govindjee. Chlorophyll a Fluorescence: A Signature of Photosynthesis. Springer, Dordrecht. 463-495. |
| [22] |
Lavaud J, Six C, Campbell DA (2016). Photosystem II repair in marine diatoms with contrasting photophysiologies. Photosynthesis Research, 127, 189-199.
DOI PMID |
| [23] | Malviya S, Scalco E, Audic S, Vincent F, Veluchamy A, Poulain J, Wincker P, Iudicone D, de Vargas C, Bittner L, Zingone A, Bowler C (2016). Insights into global diatom distribution and diversity in the world’s ocean. Proceedings of the National Academy of Sciences of the United States of America, 113, E1516-E1525. |
| [24] |
Murata N, Takahashi S, Nishiyama Y, Allakhverdiev SI (2007). Photoinhibition of photosystem II under environmental stress. Biochimica et Biophysica Acta-Bioenergetics, 1767, 414-421.
DOI PMID |
| [25] |
Noyma NP, Mesquita MCB, Roland F, Marinho MM, Huszar VLM, Lürling M (2021). Increasing temperature counteracts the negative effect of UV radiation on growth and photosynthetic efficiency of Microcystis aeruginosa and Raphidiopsis raciborskii. Photochemistry and Photobiology, 97, 753-762.
DOI URL |
| [26] |
Sippola K, Aro EM (2000). Expression of psbA genes is regulated at multiple levels in the Cyanobacterium synechococcus sp. PCC 7942. Photochemistry and Photobiology, 71, 706-714.
PMID |
| [27] | Takahashi S, Whitney S, Itoh S, Maruyama T, Badger M (2008). Heat stress causes inhibition of the de novo synthesis of antenna proteins and photobleaching in cultured Symbiodinium. Proceedings of the National Academy of Sciences of the United States of America, 105, 4203-4208. |
| [28] |
Tong SY, Hutchins DA, Gao KS (2019). Physiological and biochemical responses of Emiliania huxleyi to ocean acidification and warming are modulated by UV radiation. Biogeosciences, 16, 561-572.
DOI URL |
| [29] |
Toseland A, Daines SJ, Clark JR, Kirkham A, Strauss J, Uhlig C, Lenton TM, Valentin K, Pearson GA, Moulton V, Mock T (2013). The impact of temperature on marine phytoplankton resource allocation and metabolism. Nature Climate Change, 3, 979-984.
DOI |
| [30] | Williamson CE, Neale PJ, Hylander S, Rose KC, Figueroa FL, Robinson SA, Häder DP, Wä SÅ, Worrest RC (2019). The interactive effects of stratospheric ozone depletion, UV radiation, and climate change on aquatic ecosystems. Photochemical & Photobiological Sciences, 18, 717-746. |
| [31] | Wong CY, Teoh ML, Phang SM, Lim PE, Beardall J (2015). Interactive effects of temperature and UV radiation on photosynthesis of Chlorella strains from polar, temperate and tropical environments: differential impacts on damage and repair. PLoS ONE, 10, e0139469. DOI: 10.1371/journal.pone.0139469. |
| [32] |
Wu HY, Cockshutt AM, McCarthy A, Campbell DA (2011). Distinctive photosystem II photoinactivation and protein dynamics in marine diatoms. Plant Physiology, 156, 2184-2195.
DOI PMID |
| [33] |
Wu XX, Zhu ZW, Li X, Zha DS (2012). Effects of cytokinin on photosynthetic gas exchange, chlorophyll fluorescence parameters and antioxidative system in seedlings of eggplant (Solanum melongena L.) under salinity stress. Acta Physiologiae Plantarum, 34, 2105-2114.
DOI URL |
| [34] |
Wu YP, Zhang MJ, Li ZZ, Xu JT, Beardall J (2020). Differential responses of growth and photochemical performance of marine diatoms to ocean warming and high light irradiance. Photochemistry and Photobiology, 96, 1074-1082.
DOI PMID |
| [35] | Xu ZG, Yang SD, Li MZ, Bao ML, Wu HY (2023). Warming modulates the photosynthetic performance of Thalassiosira pseudonana in response to UV radiation. Frontiers in Microbiology, 14, 1284792. DOI: 10.3389/fmicb.2023.1284792. |
| [36] |
Ye YT, Shi DL (2020). Effects of global change on key processes of primary production in marine ecosystems. Chinese Journal of Plant Ecology, 44, 575-582.
DOI URL |
|
[叶幼亭, 史大林 (2020). 全球变化对海洋生态系统初级生产关键过程的影响. 植物生态学报, 44, 575-582.]
DOI |
|
| [37] |
Zang SS, Yan F, Yu DD, Song JJ, Wang L, Xu ZG, Wu HY (2022). Reduced salinity interacts with ultraviolet radiation to alter photosystem II function in diatom Skeletonema costatum. Journal of Oceanology and Limnology, 40, 1615-1627.
DOI |
| [38] | Zhang PY, Tang XX, Cai HJ, Yu J, Yang Z (2005). Effects of UV-B radiation on protein and nucleic acid synthesis in three marine red-tide microalgae. Acta Phytoecologica Sinica, 29, 405-409. |
| [张培玉, 唐学玺, 蔡恒江, 于娟, 杨震 (2005). 3种海洋赤潮微藻蛋白质和核酸合成对UV-B辐射增强的响应. 植物生态学报, 29, 405-409.] |
| [1] | 闫小红, 傅英健, 胡文海. 亚热带地区3种常绿阔叶植物光系统II功能对冬季短暂升温的响应[J]. 植物生态学报, 2025, 49(2): 331-342. |
| [2] | 李伟斌, 张红霞, 张玉书, 陈妮娜. 昼夜不对称增温对长白山阔叶红松林碳汇能力的影响[J]. 植物生态学报, 2023, 47(9): 1225-1233. |
| [3] | 蒋海港, 曾云鸿, 唐华欣, 刘伟, 李杰林, 何国华, 秦海燕, 王丽超, 姚银安. 三种藓类植物固碳耗水节律调节作用[J]. 植物生态学报, 2023, 47(7): 988-997. |
| [4] | 刘海燕, 臧纱纱, 张春霞, 左进城, 阮祚禧, 吴红艳. 磷饥饿下硅藻光系统II光化学反应及其对高光强的响应[J]. 植物生态学报, 2023, 47(12): 1718-1727. |
| [5] | 吴霖升, 张永光, 章钊颖, 张小康, 吴云飞. 日光诱导叶绿素荧光遥感及其在陆地生态系统监测中的应用[J]. 植物生态学报, 2022, 46(10): 1167-1199. |
| [6] | 靳川, 李鑫豪, 蒋燕, 徐铭泽, 田赟, 刘鹏, 贾昕, 查天山. 黑沙蒿光合能量分配组分在生长季的相对变化与调控机制[J]. 植物生态学报, 2021, 45(8): 870-879. |
| [7] | 武洪敏, 双升普, 张金燕, 寸竹, 孟珍贵, 李龙根, 沙本才, 陈军文. 短期生长环境光强骤增导致典型阴生植物三七光系统受损的机制[J]. 植物生态学报, 2021, 45(4): 404-419. |
| [8] | 叶子飘, 于冯, 安婷, 王复标, 康华靖. 植物气孔导度对CO2响应模型的构建[J]. 植物生态学报, 2021, 45(4): 420-428. |
| [9] | 李景, 王欣, 王振华, 王斌, 王成章, 邓美凤, 刘玲莉. 臭氧和气溶胶复合污染对杨树叶片光合作用的影响[J]. 植物生态学报, 2020, 44(8): 854-863. |
| [10] | 李旭, 吴婷, 程严, 谭钠丹, 蒋芬, 刘世忠, 褚国伟, 孟泽, 刘菊秀. 南亚热带常绿阔叶林4个树种对增温的生理生态适应能力比较[J]. 植物生态学报, 2020, 44(12): 1203-1214. |
| [11] | 刘校铭, 杨晓芳, 王璇, 张守仁. 暖温带落叶阔叶林辽东栎和五角枫生长和光合生理生态特征对模拟氮沉降的响应[J]. 植物生态学报, 2019, 43(3): 197-207. |
| [12] | 李鑫豪, 闫慧娟, 卫腾宙, 周文君, 贾昕, 查天山. 油蒿资源利用效率在生长季的相对变化及对环境因子的响应[J]. 植物生态学报, 2019, 43(10): 889-898. |
| [13] | 张娜, 朱阳春, 李志强, 卢信, 范如芹, 刘丽珠, 童非, 陈静, 穆春生, 张振华. 淹水和干旱生境下铅对芦苇生长、生物量分配和光合作用的影响[J]. 植物生态学报, 2018, 42(2): 229-239. |
| [14] | 韩吉梅, 张旺锋, 熊栋梁, 张亚黎. 植物光合作用叶肉导度及主要限制因素研究进展[J]. 植物生态学报, 2017, 41(8): 914-924. |
| [15] | 蔡建国, 韦孟琪, 章毅, 魏云龙. 遮阴对绣球光合特性和叶绿素荧光参数的影响[J]. 植物生态学报, 2017, 41(5): 570-576. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2026 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19
![]()